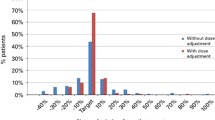
Summary:
A retrospective population pharmacokinetic (PPK) analysis was performed in 24 pediatric patients (PEDS) (0.45–16.7 years old) receiving i.v. busulfan/cyclophosphamide (i.v. Bu/Cy 4) regimen prior to allogeneic hematopoietic stem cell transplantation. I.V. Bu doses were given as a 2-hour infusion every 6 h over 4 days. Initial dosing of i.v. Bu was 1 mg/kg for children ⩽4 years old and 0.8 mg/kg for patients >4 years old. Bu plasma concentrations at doses 1, 9 and 13 were analyzed through a multivariate NONMEM analysis. A close log-linear relationship between body weight (BW) and i.v. Bu clearance was demonstrated with no further age-dependency or gender effect. The interpatient coefficient of variation (CV) in Bu clearance significantly decreased from 56% (covariate-free model) to 19% (BW covariate model) and reproducible i.v. Bu exposure between doses was illustrated (intraindividual CV=9%). Based on the PPK model, a novel Bu dosing regimen (ie: doses in mg/kg adjusted to discrete weight categories) for a better AUC targeting was developed by simulation on 1000 patients. Age-based dosing was demonstrated not to be clinically relevant with i.v. Bu. Use of the new BW-based dosing appears to be more appropriate for the PEDS.




Similar content being viewed by others
References
Michel G, Gluckman E, Esperoou-Bourdeau H et al. Allogeneic bone marrow transplantation for children with acute myeloblastic leukaemia in first complete remission: impact of conditioning regimen without total body irradiation – a report from the Société Française de Greffe de Moelle. J Clin Oncol 1994; 12: 1217–1222.
Hassan M, Ljungman P, Bolme P et al. Busulfan bioavailability. Blood 1994; 84: 2144–2150.
Vassal G, Koscielny S, Challine D et al. Busulfan disposition and hepatic veno-occlusive disease in children undergoing bone marrow transplantation. Cancer Chemother Pharmacol 1996; 37: 247–253.
Pawlowska AB, Blazar BR, Angelucci E et al. Relationship of plasma pharmacokinetics of high dose oral busulphan to the outcome of allogeneic bone marrow transplantation in children with thalassaemia. Blood 1996; 88 (Suppl. 1): 457a.
Shaw PJ, Scharping CE, Brian RJ, Earl JW . Busulphan pharmacokinetics using a single daily high dose regimen in children with acute leukaemia. Blood 1994; 84: 2357–2362.
Hassan M, Fasth A, Gerritsen B et al. Busulfan kinetics and limited sampling model in children with leukemia and inherited disorders. Bone Marrow Transplant 1996; 18: 843–850.
Hassan M, Oberg G, Bekassy AN et al. Pharmacokinetics of high-dose busulphan in relation to age and chronopharmacology. Cancer Chermother Pharmacol 1991; 28: 130–134.
Vassal G . Pharmacologically-guided dose adjustment of busulfan in high-dose chemotherapy regimens: rationale and pitfalls (review). Anticancer Res 1994; 14: 2363–2370.
Tran HAT, Madden T, Petropoulos D et al. Individualizing high-dose oral busulfan: prospective dose adjustment in a pediatric population undergoing allogeneic stem cell transplantation for advanced hematologic malignancies. Bone Marrow Transplant 2000; 26: 463–470.
Krivoy N, Hoffer E, Elhasid R et al. Therapeutic monitoring of busulfan in pediatric bone marrow transplantation. Pediatr Hematol Oncol 2002; 19: 31–37.
Slattery JT, Sanders JE, Buckner CD et al. Graft-rejection and toxicity following bone marrow transplantation in relation to busulfan pharmacokinetics. Bone Marrow Transplant 1995; 16: 31–42.
Dix SP, Wingard JR, Mullins RE et al. Association of busulfan area under the curve with veno-occlusive disease following BMT. Bone Marrow Transplant 1996; 17: 225–230.
Chattergoon DS, Saunders EF, Klein J et al. An improved limited sampling method for individualised busulphan dosing in bone marrow transplantation in children. Bone Marrow Transplant 1997; 20: 347–354.
Slattery JT, Risler LJ . Therapeutic monitoring of busulfan in hematopoietic stem cell transplantation. Ther Drug Monit 1998; 20: 543–554.
Grochow LB . Busulfan disposition: the role of therapeutic monitoring in bone marrow transplantation induction regimens. Semin Oncol 1993; 20: 18–25.
De Leve LD . Cellular target of cyclophosphamide toxicity in the murine liver: role of glutathione and site of metabolic activation. Hepatology 1996; 24: 830–837.
McDonald GB, Ren S, Bouvier ME et al. Venoocclusive disease of the liver and cyclophosphamide pharmacokinetics: a prospective study in marrow transplant patients (abstract). Hepatology 1999; 17: 314A.
Bolinger AM, Zangwill AB, Slattery JT et al. Target dose adjustment of busulfan in pediatric patients undergoing bone marrow transplantation. Bone Marrow Transplant 2001; 28: 1013–1018.
McCune JS, Gooley T, Gibbs JP et al. Busulfan concentration and graft rejection in pediatric patients undergoing hematopoietic stem cell transplantation. Bone Marrow Transplant 2002; 30: 167–173.
Grochow LB, Krivit W, Whitley CB, Blazar B . Busulfan disposition in children. Blood 1990; 75: 1723–1727.
Regazzi MB, Locatelli F, Buggia I et al. Disposition of high-dose busulfan in pediatric patients undergoing bone marrow transplantation. Clin Pharmacol Ther 1993; 54: 45–52.
Vassal G, Fischer A, Challine D et al. Busulfan disposition below the age of three: alteration in children with lysosomal storage disease. Blood 1993; 3: 1030–1034.
Gibbs JP, Murray G, Risler L et al. Age-dependent tetrahydrothiophenium ion formation in young children and adults receiving high-dose busulfan. Cancer Res 1997; 57: 5509–5516.
Gibbs JP, Liacouras CA, Baldassano RN, Slattery JT . Up-regulation of glutathione S-transferase activity in enterocytes of young children. Drug Metab Dispos 1999; 27: 1466–1469.
Vassal G, Deroussent A, Challine D et al. Is 600 mg/m2 the appropriate dosage of busulfan in children undergoing bone marrow transplantation? Blood 1992; 9: 2475–2479.
Yeager AM, Wagner JE, Graham ML et al. Optimization of busulfan dosage in children undergoing bone marrow transplantation: a pharmacokinetic study of dose escalation. Blood 1992; 9: 2425–2428.
Bleyzac N, Souillet G, Magron P et al. Improved clinical outcome of pediatric bone marrow recipients using a test dose and Bayesian pharmacokinetic individualization of busulfan dosage regimens. Bone Marrow Transplant 2001; 28: 743–751.
Beal SL, Sheiner LB . NONMEM User's Guide. University of California, San Francisco, 1998.
Halford NHG . A size standard for pharmacokinetics. Clin Pharmacokinetics 1996; 30: 329–332.
Parke V, Halford NHG, Charles BG . A procedure for generating bootstrap samples for the validation of non linear mixed-effects population models. Comput Methods Program Biomed 1999; 59: 19–29.
Boyd E . Surface Area of the Human Body. University of Minnesota Press: Mineapolis, 1935.
Adersson BS, Kashyap A, Gian V et al. Conditioning therapy with intravenous busulfan and cyclophosphamide (IV BuCy2) for hematologic malignancies prior to allogeneic stem cell transplantation: a phase II study. Biol Blood Marrow Transplant 2003; 8: 145–154.
Schuler US, Renner UD, Kroschinsky F et al. Intravenous busulphan for conditioning before autologous or allogeneic human blood stem cell transplantation. Br J Haematol 2001; 114: 944–950.
Hassan M, Nilsson C, Hassan Z et al. A phase II trial of liposomal busulphan as an intravenous myeloablative agent prior to stem cell transplantation: 500 mg/m2 as a optimal total dose for conditioning. Bone Marrow Transplant 2002; 30: 833–841.
Sharkey I, Boddy AV, Mycroft J et al. Body surface area estimation in children using weight alone: application in paediatric oncology. Br J Cancer 2001; 85: 23–28.
DuBois D, DuBois EF . A formula to estimate the approximate surface area if height and weight be known. Arch Intern Med 1916; 17: 863–871.
George SL, Gehan EA . Methods for measurement of body surface area. J Pediatr 1979; 94: 342–343.
Haycock GB, Schwarz GJ . Geometric method for measuring body surface area. A height–weight formula validated in infants. J Paediatr 1978; 93: 62–66.
Mosteller RD . Simplified calculation of body surface area. N Eng J Med 1987; 317: 1098.
Sendroy J, Cechini LP . Determination of human body surface area from height and weight. J Appl Physiol 1952; 7: 1–12.
Briars GL, Bailey BJR . Surface area estimation: pocket calculator v nomogram. Arch Dis Childhood 1994; 70: 246–247.
Dupuis LL, Najdova M, Saunders EF . Retrospective appraisal of busulfan dose adjustment in children. Bone Marrow Transplant 2000; 26: 1143–1147.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Nguyen, L., Fuller, D., Lennon, S. et al. I.V. busulfan in pediatrics: a novel dosing to improve safety/efficacy for hematopoietic progenitor cell transplantation recipients. Bone Marrow Transplant 33, 979–987 (2004). https://doi.org/10.1038/sj.bmt.1704446
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/sj.bmt.1704446
- Springer Nature Limited