Abstract
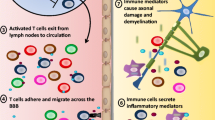
Immune cells mediate critical inflammatory and neurodegenerative processes in the CNS in individuals with multiple sclerosis (MS). In MS, activated microglia, border-associated macrophages and monocyte-derived macrophages in the CNS can encounter T cells that have infiltrated the brain parenchyma from the circulation. Although microglia and T cells both contribute to normal CNS development and homeostasis, evidence suggests that the meeting of activated microglia and macrophages with encephalitogenic T cells exacerbates their capacity to inflict injury. This crosstalk involves many cell-surface molecules, cytokines and neurotoxic factors. In this Review, we summarize the mechanisms and consequences of T cell–microglia interactions as identified with in vitro experiments and animal models, and discuss the challenges that arise when translating this preclinical knowledge to MS in humans. We also consider therapeutic approaches to MS of which the mechanisms involve prevention or modulation of T cell and microglia responses and their interactions.
Key points
-
Microglia and T cells help to maintain homeostasis in the CNS.
-
Activated microglia and macrophages in the CNS interact with T cells via cell-to-cell contact and cytokine-mediated communication to promote neuroinflammation in an experimental autoimmune encephalomyelitis (EAE) model of multiple sclerosis (MS).
-
Monocytes and monocyte-derived cells add heterogeneity to the CNS macrophage population in EAE and MS and could also interact with T cells to regulate disease pathology.
-
Several challenges are involved in translating T cell–microglia interactions identified in vitro or in animal models to MS, so these findings should be considered carefully when generalizing to the human disease.
-
The mechanisms of existing and potential MS therapeutics involve interference with the interactions and functions of T cells and microglia, but the contribution of these effects is difficult to confirm.



Similar content being viewed by others
References
Engelhardt, B., Vajkoczy, P. & Weller, R. O. The movers and shapers in immune privilege of the CNS. Nat. Immunol. 18, 123–131 (2017).
Louveau, A. et al. Structural and functional features of central nervous system lymphatic vessels. Nature 523, 337–341 (2015).
Louveau, A. et al. CNS lymphatic drainage and neuroinflammation are regulated by meningeal lymphatic vasculature. Nat. Neurosci. 21, 1380–1391 (2018).
Ransohoff, R. M. How neuroinflammation contributes to neurodegeneration. Science. 353, 777–783 (2016).
Friese, M. A., Schattling, B. & Fugger, L. Mechanisms of neurodegeneration and axonal dysfunction in multiple sclerosis. Nat. Rev. Neurol. 10, 225–238 (2014).
Perry, V. H., Nicoll, J. A. R. & Holmes, C. Microglia in neurodegenerative disease. Nat. Rev. Neurol. 6, 193–201 (2010).
González, H. & Pacheco, R. T-cell-mediated regulation of neuroinflammation involved in neurodegenerative diseases. J. Neuroinflammation 11, 201 (2014).
Dendrou, C. A., Fugger, L. & Friese, M. A. Immunopathology of multiple sclerosis. Nat. Rev. Immunol. 15, 545–558 (2015).
Lassmann, H., Brück, W. & Lucchinetti, C. F. The immunopathology of multiple sclerosis: an overview. Brain Pathol. 17, 210–218 (2007).
Marik, C., Felts, P. A., Bauer, J., Lassmann, H. & Smith, K. J. Lesion genesis in a subset of patients with multiple sclerosis: a role for innate immunity? Brain 130, 2800–2815 (2007).
Schirmer, L. et al. Substantial early, but nonprogressive neuronal loss in multiple sclerosis (MS) spinal cord. Ann. Neurol. 66, 698–704 (2009).
’t Hart, B. A., Gran, B. & Weissert, R. EAE: imperfect but useful models of multiple sclerosis. Trends Mol. Med. 17, 119–125 (2011).
Ransohoff, R. M. Animal models of multiple sclerosis: the good, the bad and the bottom line. Nat. Neurosci. 15, 1074–1077 (2012).
Stromnes, I. M. & Goverman, J. M. Active induction of experimental allergic encephalomyelitis. Nat. Protoc. 1, 1810–1819 (2006).
De Rosbo, N. K. et al. Reactivity to myelin antigens in multiple sclerosis. Peripheral blood lymphocytes respond predominantly to myelin oligodendrocyte glycoprotein. J. Clin. Invest. 92, 2602–2608 (1993).
Becher, B., Spath, S. & Goverman, J. Cytokine networks in neuroinflammation. Nat. Rev. Immunol. 17, 49–59 (2017).
Kallaur, A. P. et al. Cytokine profile in relapsing-remitting multiple sclerosis patients and the association between progression and activity of the disease. Mol. Med. Rep. 7, 1010–1020 (2013).
Khaibullin, T. et al. Elevated levels of proinflammatory cytokines in cerebrospinal fluid of multiple sclerosis patients. Front. Immunol. 8, 531 (2017).
Maimone, D., Gregory, S., Arnason, B. G. W. & Reder, A. T. Cytokine levels in the cerebrospinal fluid and serum of patients with multiple sclerosis. J. Neuroimmunol. 32, 67–74 (1991).
Saxena, A., Martin-Blondel, G., Mars, L. T. & Liblau, R. S. Role of CD8 T cell subsets in the pathogenesis of multiple sclerosis. FEBS Lett. 585, 3758–3763 (2011).
Lucchinetti, C. F. et al. Inflammatory cortical demyelination in early multiple sclerosis. N. Engl. J. Med. 365, 2188–2197 (2011).
Machado-Santos, J. et al. The compartmentalized inflammatory response in the multiple sclerosis brain is composed of tissue-resident CD8+ T lymphocytes and B cells. Brain 141, 2066–2082 (2018).
van Nierop, G. P. et al. Phenotypic and functional characterization of T cells in white matter lesions of multiple sclerosis patients. Acta Neuropathol. 134, 383–401 (2017).
Huseby, E. S. et al. A pathogenic role for myelin-specific CD8+ T cells in a model for multiple sclerosis. J. Exp. Med. 194, 669–676 (2001).
Sun, D. et al. Myelin antigen-specific CD8+ T cells are encephalitogenic and produce severe disease in C57BL/6 mice. J. Immunol. 166, 7579–7587 (2001).
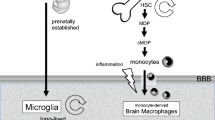
Goldmann, T. et al. Origin, fate and dynamics of macrophages at central nervous system interfaces. Nat. Immunol. 17, 797–805 (2016).
Elmore, M. R. P. et al. Colony-stimulating factor 1 receptor signaling is necessary for microglia viability, unmasking a microglia progenitor cell in the adult brain. Neuron 82, 380–397 (2014).
Ginhoux, F. et al. Fate mapping analysis reveals that adult microglia derive from primitive macrophages. Science 330, 841–845 (2010). This is one of the first studies to show that the origin and turnover of resident microglia in the CNS of mice are distinct from those of the mononuclear phagocyte system.
Wang, Y. et al. IL-34 is a tissue-restricted ligand of CSF1R required for the development of Langerhans cells and microglia. Nat. Immunol. 13, 753–760 (2012).
Qin, Y. et al. A milieu molecule for TGF-β required for microglia function in the nervous system. Cell 174, 156–171 (2018).
Datta, M. et al. Histone deacetylases 1 and 2 regulate microglia function during development, homeostasis, and neurodegeneration in a context-dependent manner. Immunity 48, 514–529 (2018).
Varol, D. et al. Dicer deficiency differentially impacts microglia of the developing and adult brain. Immunity 46, 1030–1044 (2017).
Tay, T. L. et al. A new fate mapping system reveals context-dependent random or clonal expansion of microglia. Nat. Neurosci. 20, 793–803 (2017).
Jordão, M. J. C. et al. Single-cell profiling identifies myeloid cell subsets with distinct fates during neuroinflammation. Science 363, eaat7554 (2019). In this study, single-cell RNA sequencing shows the heterogeneity and function of different myeloid cell populations in the CNS in the EAE model of MS.
Giordana, M. T. et al. Reactive cell proliferation and microglia following injury to the rat brain. Neuropathol. Appl. Neurobiol. 20, 163–174 (1994).
Bellver-Landete, V. et al. Microglia are an essential component of the neuroprotective scar that forms after spinal cord injury. Nat. Commun. 10, 518 (2019).
Li, Q. & Barres, B. A. Microglia and macrophages in brain homeostasis and disease. Nat. Rev. Immunol. 18, 225–242 (2018).
Menassa, D. A. & Gomez-Nicola, D. Microglial dynamics during human brain development. Front. Immunol. 9, 1014 (2018).
Prinz, M., Erny, D. & Hagemeyer, N. Ontogeny and homeostasis of CNS myeloid cells. Nat. Immunol. 18, 385–392 (2017).
Van Hove, H. et al. A single-cell atlas of mouse brain macrophages reveals unique transcriptional identities shaped by ontogeny and tissue environment. Nat. Neurosci. 22, 1021–1035 (2019).
Huang, Y. et al. Repopulated microglia are solely derived from the proliferation of residual microglia after acute depletion. Nat. Neurosci. 21, 530–540 (2018).
Ajami, B., Bennett, J. L., Krieger, C., Tetzlaff, W. & Rossi, F. M. V. Local self-renewal can sustain CNS microglia maintenance and function throughout adult life. Nat. Neurosci. 10, 1538–1543 (2007).
Cronk, J. C. et al. Peripherally derived macrophages can engraft the brain independent of irradiation and maintain an identity distinct from microglia. J. Exp. Med. 215, 1627–1647 (2018).
Waisman, A., Ginhoux, F., Greter, M. & Bruttger, J. Homeostasis of microglia in the adult brain: review of novel microglia depletion systems. Trends Immunol. 36, 625–636 (2015).
Bruttger, J. et al. Genetic cell ablation reveals clusters of local self-renewing microglia in the mammalian central nervous system. Immunity 43, 92–106 (2015).
Varvel, N. H. et al. Microglial repopulation model reveals a robust homeostatic process for replacing CNS myeloid cells. Proc. Natl. Acad. Sci. USA. 109, 18150–18155 (2012).
Ueno, M. et al. Layer V cortical neurons require microglial support for survival during postnatal development. Nat. Neurosci. 16, 543–551 (2013).
Madry, C. et al. Microglial ramification, surveillance, and interleukin-1β release are regulated by the two-pore domain K+ channel THIK-1. Neuron 97, 299–312 (2018).
Schafer, D. P. et al. Microglia sculpt postnatal neural circuits in an activity and complement-dependent manner. Neuron 74, 691–705 (2012).
Parkhurst, C. N. et al. Microglia promote learning-dependent synapse formation through brain-derived neurotrophic factor. Cell 155, 1596–1609 (2013).
Wlodarczyk, A. et al. A novel microglial subset plays a key role in myelinogenesis in developing brain. EMBO J. 36, 3292–3308 (2017).
Pridans, C. et al. Pleiotropic impacts of macrophage and microglial deficiency on development in rats with targeted mutation of the Csf1r locus. J. Immunol. 201, 2683–2699 (2018).
Rubino, S. J. et al. Acute microglia ablation induces neurodegeneration in the somatosensory system. Nat. Commun. 9, 4578 (2018).
Hagemeyer, N. et al. Microglia contribute to normal myelinogenesis and to oligodendrocyte progenitor maintenance during adulthood. Acta Neuropathol. 134, 441–458 (2017).
Wendeln, A. C. et al. Innate immune memory in the brain shapes neurological disease hallmarks. Nature 556, 332–338 (2018).
Banks, W. A. et al. Lipopolysaccharide-induced blood-brain barrier disruption: roles of cyclooxygenase, oxidative stress, neuroinflammation, and elements of the neurovascular unit. J. Neuroinflammation 12, 223 (2015).
Hammond, T. R. et al. Single-cell RNA sequencing of microglia throughout the mouse lifespan and in the injured brain reveals complex cell-state changes. Immunity 50, 253–271 (2019).
Li, Q. et al. Developmental heterogeneity of microglia and brain myeloid cells revealed by deep single-cell RNA sequencing. Neuron 101, 207–223 (2019).
Mrdjen, D. et al. High-dimensional single-cell mapping of central nervous system immune cells reveals distinct myeloid subsets in health, aging, and disease. Immunity 48, 380–395 (2018). In this study, mass cytometry was used to compare the heterogeneity and function of myeloid cell subsets in the CNS during health and neurodegenerative disease.
Ajami, B. et al. Single-cell mass cytometry reveals distinct populations of brain myeloid cells in mouse neuroinflammation and neurodegeneration models. Nat. Neurosci. 21, 541–551 (2018).
Siffrin, V. et al. Differential immune cell dynamics in the CNS cause CD4 T cell compartmentalization. Brain 132, 1247–1258 (2009).
Kivisäkk, P. et al. Human cerebrospinal fluid central memory CD4+ T cells: evidence for trafficking through choroid plexus and meninges via P-selectin. Proc. Natl. Acad. Sci. USA. 100, 8389–8394 (2003). This study identifies memory T cells in the cerebrospinal fluid of patients with MS and provides evidence for a potential mechanism of T cell trafficking into the brain.
Fritzsching, B. et al. Cutting edge: in contrast to effector T cells, CD4+CD25+FoxP3+ regulatory T cells are highly susceptible to CD95 ligand- but not to TCR-mediated cell death. J. Immunol. 175, 32–36 (2014).
Wolf, S. A. et al. CD4-positive T lymphocytes provide a neuroimmunological link in the control of adult hippocampal neurogenesis. J. Immunol. 182, 3979–3984 (2009).
Ziv, Y. et al. Immune cells contribute to the maintenance of neurogenesis and spatial learning abilities in adulthood. Nat. Neurosci. 9, 268–275 (2006).
Kipnis, J. et al. Neuroprotective autoimmunity: naturally occurring CD4+CD25+ regulatory T cells suppress the ability to withstand injury to the central nervous system. Proc. Natl. Acad. Sci. USA. 99, 15620–15625 (2002).
Kunis, G., Baruch, K., Miller, O. & Schwartz, M. Immunization with a myelin-derived antigen activates the brain’s choroid plexus for recruitment of immunoregulatory cells to the CNS and attenuates disease progression in a mouse model of ALS. J. Neurosci. 35, 6381–6393 (2015).
Hvilsted Nielsen, H., Toft-Hansen, H., Lambertsen, K. L., Owens, T. & Finsen, B. Stimulation of adult oligodendrogenesis by myelin-specific T cells. Am. J. Pathol. 179, 2028–2041 (2011).
Kieback, E. et al. Thymus-derived regulatory T cells are positively selected on natural self-antigen through cognate interactions of high functional avidity. Immunity 44, 1114–1126 (2016).
Xie, L., Choudhury, G. R., Winters, A., Yang, S. H. & Jin, K. Cerebral regulatory T cells restrain microglia/macrophage-mediated inflammatory responses via IL-10. Eur. J. Immunol. 45, 180–191 (2015).
Ito, M. et al. Brain regulatory T cells suppress astrogliosis and potentiate neurological recovery. Nat. 565, 246–250 (2019).
Venken, K. et al. Natural naive CD4+CD25+CD127low regulatory T cell (Treg) development and function are disturbed in multiple sclerosis patients: recovery of memory Treg homeostasis during disease progression. J. Immunol. 180, 6411–6420 (2008).
Fritzsching, B. et al. Intracerebral human regulatory T cells: analysis of CD4+CD25+FOXP3+ T cells in brain lesions and cerebrospinal fluid of multiple sclerosis patients. PLoS One 18, e17988 (2011).
Kerfoot, S. M. & Kubes, P. Overlapping roles of P-selectin and 4 integrin to recruit leukocytes to the central nervous system in experimental autoimmune encephalomyelitis. J. Immunol. 169, 1000–1006 (2002).
Bartholomäus, I. et al. Effector T cell interactions with meningeal vascular structures in nascent autoimmune CNS lesions. Nature 462, 94–98 (2009).
Kawakami, N. et al. Live imaging of effector cell trafficking and autoantigen recognition within the unfolding autoimmune encephalomyelitis lesion. J. Exp. Med. 201, 1805–1814 (2005). Together with Bartholomäus et al. (2009), this paper uses real-time imaging to demonstrate live interactions and movement of T cells with CNS myeloid cells at the blood–brain barrier and in CNS lesions in the EAE model of MS.
Vajkoczy, P., Laschinger, M. & Engelhardt, B. α4-integrin-VCAM-1 binding mediates G protein-independent capture of encephalitogenic T cell blasts to CNS white matter microvessels. J. Clin. Invest. 108, 557–565 (2001).
Greter, M. et al. Dendritic cells permit immune invasion of the CNS in an animal model of multiple sclerosis. Nat. Med. 11, 328–334 (2005).
Mundt, S. et al. Conventional DCs sample and present myelin antigens in the healthy CNS and allow parenchymal T cell entry to initiate neuroinflammation. Sci. Immunol. 4, eaau8380 (2019). This study demonstrates the importance of MHCII-expressing conventional dendritic cells as APCs in the EAE model of MS.
Stephenson, E. L. et al. Chondroitin sulfate proteoglycans as novel drivers of leucocyte infiltration in multiple sclerosis. Brain 141, 1094–1110 (2018).
Reboldi, A. et al. C-C chemokine receptor 6-regulated entry of TH-17 cells into the CNS through the choroid plexus is required for the initiation of EAE. Nat. Immunol. 10, 514–523 (2009).
Haider, L. et al. Oxidative damage in multiple sclerosis lesions. Brain 134, 1914–1924 (2011).
Kuhlmann, T. Acute axonal damage in multiple sclerosis is most extensive in early disease stages and decreases over time. Brain 125, 2202–2212 (2002).
Androdias, G. et al. Meningeal T cells associate with diffuse axonal loss in multiple sclerosis spinal cords. Ann. Neurol. 68, 465–476 (2010).
Heneka, M. T., Golenbock, D. T. & Latz, E. Innate immunity in Alzheimer’s disease. Nat. Immunol. 27, 220–222 (2015).
Wang, Y. et al. TREM2 lipid sensing sustains the microglial response in an Alzheimer’s disease model. Cell 160, 1061–1071 (2015).
García-Vallejo, J. J. et al. CNS myelin induces regulatory functions of DC-SIGN-expressing, antigen-presenting cells via cognate interaction with MOG. J. Exp. Med. 211, 1465–1483 (2014).
Duan, Y., Sahley, C. L. & Muller, K. J. Ac. ATP and NO dually control migration of microglia to nerve lesions. Dev. Neurobiol. 69, 60–72 (2009).
Windhagen, A. et al. Expression of costimulatory molecules B7-1 (CD80), B7-2 (CD86), and interleukin 12 cytokine in multiple sclerosis lesions. J. Exp. Med. 182, 1985–1996 (1995).
Gerritse, K. et al. CD40-CD40 ligand interactions in experimental allergic encephalomyelitis and multiple sclerosis. Proc. Natl. Acad. Sci. USA. 93, 2499–2504 (1996).
Zrzavy, T. et al. Loss of ‘homeostatic’ microglia and patterns of their activation in active multiple sclerosis. Brain 140, 1900–1913 (2017).
Kopf, M., Schneider, C. & Nobs, S. P. The development and function of lung-resident macrophages and dendritic cells. Nat. Immunol. 16, 36–44 (2015).
Cho, K. W. et al. An MHC II-dependent activation loop between adipose tissue macrophages and CD4+ T cells controls obesity-induced inflammation. Cell Rep. 9, 605–617 (2014).
Schreiber, H. a et al. Intestinal monocytes and macrophages are required for T cell polarization in response to Citrobacter rodentium. J. Exp. Med. 210, 2025–2039 (2013).
Mack, C. L., Vanderlugt-Castaneda, C. L., Neville, K. L. & Miller, S. D. Microglia are activated to become competent antigen presenting and effector cells in the inflammatory environment of the Theiler’s virus model of multiple sclerosis. J. Neuroimmunol. 144, 68–79 (2003).
Re, F. et al. Granulocyte-macrophage colony-stimulating factor induces an expression program in neonatal microglia that primes them for antigen presentation. J. Immunol. 169, 2264–2273 (2014).
Ponomarev, E. D. et al. GM-CSF production by autoreactive T cells is required for the activation of microglial cells and the onset of experimental autoimmune encephalomyelitis. J. Immunol. 178, 39–48 (2007).
Komuczki, J. et al. Fate-mapping of GM-CSF expression identifies a discrete subset of inflammation-driving T helper cells regulated by cytokines IL-23 and IL-1β. Immunity 50, 1289–1304 (2019). This study uses fate mapping to show that CSF-2-producing T H cells in the CNS are critical for the pathogenesis of EAE.
Choi, S. S., Lee, H. J., Lim, I., Satoh, J. I. & Kim, S. U. Human astrocytes: secretome profiles of cytokines and chemokines. PLoS One 9, e92325 (2014).
Lee, S. C., Liu, W., Brosnan, C. F. & Dickson, D. W. GM-CSF promotes proliferation of human fetal and adult microglia in primary cultures. Glia 12, 309–318 (1994).
Esen, N. & Kielian, T. Effects of low dose GM-CSF on microglial inflammatory profiles to diverse pathogen-associated molecular patterns (PAMPs). J. Neuroinflammation 4, 10 (2007).
Matyszak, M. K. et al. Microglia induce myelin basic protein-specific T cell anergy or T cell activation, according to their state of activation. Eur. J. Immunol. 29, 3063–3076 (1999).
Siffrin, V. et al. In vivo imaging of partially reversible TH17 cell-induced neuronal dysfunction in the course of encephalomyelitis. Immunity 33, 424–436 (2010).
Sosa, R. A., Murphey, C., Ji, N., Cardona, A. E. & Forsthuber, T. G. The kinetics of myelin antigen uptake by myeloid cells in the central nervous system during experimental autoimmune encephalomyelitis. J. Immunol. 191, 5848–5857 (2013).
Wolf, Y. et al. Microglial MHC class II is dispensable for experimental autoimmune encephalomyelitis and cuprizone-induced demyelination. Eur. J. Immunol. 48, 1308–1318 (2018).
Grewal, I. S. & Flavell, R. A. The role of CD40 ligand in costimulation and T-cell activation. Immunol. Rev. 153, 85–106 (1996).
Howland, K. C., Ausubel, L. J., London, C. A. & Abbas, A. K. The roles of CD28 and CD40 ligand in T cell activation and tolerance. J. Immunol. 164, 4465–4470 (2000).
Ponomarev, E. D., Shriver, L. P. & Dittel, B. N. CD40 expression by microglial cells is required for their completion of a two-step activation process during central nervous system autoimmune inflammation. J. Immunol. 176, 1402–1410 (2006).
Keren-Shaul, H. et al. A unique microglia type associated with restricting development of Alzheimer’s disease. Cell 169, 1276–1290 (2017).
Korin, B. et al. High-dimensional, single-cell characterization of the brain’s immune compartment. Nat. Neurosci. 20, 1300–1309 (2017).
Wlodarczyk, A., Løbner, M., Cédile, O. & Owens, T. Comparison of microglia and infiltrating CD11c+ cells as antigen presenting cells for T cell proliferation and cytokine response. J. Neuroinflammation 11, 1–9 (2014).
Ebner, F. et al. Microglial activation milieu controls regulatory T cell responses. J. Immunol. 191, 5594–5602 (2013).
Krasemann, S. et al. The TREM2-APOE pathway drives the transcriptional phenotype of dysfunctional microglia in neurodegenerative diseases. Immunity 47, 566–581 (2017).
Neniskyte, U. & Brown, G. C. Lactadherin/MFG-E8 is essential for microglia-mediated neuronal loss and phagoptosis induced by amyloid β. J. Neurochem. 126, 312–317 (2013).
Brown, G. C. & Vilalta, A. How microglia kill neurons. Brain Res. 1628, 288–297 (2015).
Nikić, I. et al. A reversible form of axon damage in experimental autoimmune encephalomyelitis and multiple sclerosis. Nat. Med. 17, 495–499 (2011). This study shows that macrophages and microglia mediate a reversible focal axonal degeneration in EAE and potentially in MS.
Huang, Y. et al. Glutaminase dysregulation in HIV-1-infected human microglia mediates neurotoxicity: relevant to HIV-1-associated neurocognitive disorders. J. Neurosci. 31, 15195–15204 (2011).
Gan, L. et al. Identification of cathepsin B as a mediator of neuronal death induced by Abeta-activated microglial cells using a functional genomics approach. J. Biol. Chem. 279, 5565–5572 (2004).
Chabot, S., Williams, G. & Yong, V. W. Microglial production of TNF-α is induced by activated T lymphocytes involvement of VLA-4 and inhibition by interferonβ-1b. J. Clin. Invest. 100, 604–612 (1997). This study shows that T cell interactions with microglia via adhesion molecules can increase inflammatory cytokine production.
Ye, S. M. & Johnson, R. W. Increased interleukin-6 expression by microglia from brain of aged mice. J. Neuroimmunol. 93, 139–148 (1999).
Gimsa, U., Peter, S. V., Lehmann, K., Bechmann, I. & Nitsch, R. Axonal damage induced by invading T cells in organotypic central nervous system tissue in vitro: involvement of microglial cells. Brain Pathol. 10, 365–377 (2000).
Heppner, F. L. et al. Experimental autoimmune encephalomyelitis repressed by microglial paralysis. Nat. Med. 11, 146–152 (2005).
Wimmer, I. et al. Microglia pre-activation and neurodegeneration precipitate neuroinflammation without exacerbating tissue injury in experimental autoimmune encephalomyelitis. Acta Neuropathol. Commmun. 9, 1–13 (2019).
Murphy, A. C., Lalor, S. J., Lynch, M. A. & Mills, K. H. Infiltration of Th1 and Th17 cells and activation of microglia in the CNS during the course of experimental autoimmune encephalomyelitis. Brain Behav Immun 24, 641–651 (2010).
Grebing, M. et al. Myelin-specific T cells induce interleukin-1beta expression in lesion-reactive microglial-like cells in zones of axonal degeneration. Glia 64, 407–424 (2016).
Prajeeth, C. K. et al. Effector molecules released by Th1 but not Th17 cells drive an M1 response in microglia. Brain. Behav. Immun. 37, 248–259 (2014).
Codarri, L. et al. RORγ3t drives production of the cytokine GM-CSF in helper T cells, which is essential for the effector phase of autoimmune neuroinflammation. Nat. Immunol. 12, 560–567 (2011).
Croxford, A. L. et al. The cytokine GM-CSF drives the inflammatory signature of CCR2+ monocytes and licenses autoimmunity. Immunity 43, 502–514 (2015).
Galli, E. et al. GM-CSF and CXCR4 define a T helper cell signature in multiple sclerosis. Nat. Med. 25, 1290–1300 (2019). This study uses mass cytometry to show that CSF-2-producing T cells are enriched in the peripheral blood and in the CNS of patients with relapsing–remitting MS.
Becher, B., Durell, B. G. & Noelle, R. J. IL-23 produced by CNS-resident cells controls T cell encephalitogenicity during the effector phase of experimental autoimmune encephalomyelitis. J. Clin. Invest. 112, 1186–1191 (2003).
Guadagno, J., Swan, P., Shaikh, R. & Cregan, S. P. Microglia-derived IL-1β triggers p53-mediated cell cycle arrest and apoptosis in neural precursor cells. Cell Death Dis. 6, e1179 (2015).
Davies, C. A. et al. The progression and topographic distribution of interleukin-1β expression after permanent middle cerebral artery occlusion in the rat. J. Cereb. Blood Flow Metab. 19, 87–98 (1999).
O’Sullivan, B. J. et al. IL-1 breaks tolerance through expansion of CD25+ effector T cells. J. Immunol. 176, 7278–7287 (2014).
Ben-Sasson, S. Z. et al. IL-1 acts directly on CD4 T cells to enhance their antigen-driven expansion and differentiation. Proc. Natl. Acad. Sci. USA. 106, 7119–7124 (2009).
Shinohara, M. L., Kim, J.-H., Garcia, V. A. & Cantor, H. Engagement of the type I interferon receptor on dendritic cells inhibits T helper 17 cell development: role of intracellular osteopontin. Immunity 29, 68–78 (2008).
Hur, E. M. et al. Osteopontin-induced relapse and progression of autoimmune brain disease through enhanced survival of activated T cells. Nat. Immunol. 8, 74–83 (2007).
Derkow, K., Krüger, C., Dembny, P. & Lehnardt, S. Microglia induce neurotoxic IL-17+ γδ T cells dependent on TLR2, TLR4, and TLR9 activation. PLoS One 10, e0135898 (2015).
Williams, K., Dooley, N., Ulvestad, E., Becher, B. & Antel, J. P. IL-10 production by adult human derived microglial cells. Neurochem. Int. 29, 55–64 (1996).
Chabot, S., Williams, G., Hamilton, M., Sutherland, G. & Yong, V. W. Mechanisms of IL-10 production in human microglia-T cell interaction. J. Immunol. 162, 6819–6828 (1999).
Kataoka, A., Tozaki-Saitoh, H., Koga, Y., Tsuda, M. & Inoue, K. Activation of P2X7 receptors induces CCL3 production in microglial cells through transcription factor NFAT. J. Neurochem. 108, 115–125 (2009).
Kohno, H., Maeda, T., Perusek, L., Pearlman, E. & Maeda, A. CCL3 production by microglial cells modulates disease severity in murine models of retinal degeneration. J. Immunol. 192, 3816–3827 (2014).
Mildner, A. et al. CCR2+Ly-6Chi monocytes are crucial for the effector phase of autoimmunity in the central nervous system. Brain 132, 2487–2500 (2009).
King, I. L., Dickendesher, T. L. & Segal, B. M. Circulating Ly-6C+ myeloid precursors migrate to the CNS and play a pathogenic role during autoimmune demyelinating disease. Blood 113, 3190–3197 (2009).
Locatelli, G. et al. Mononuclear phagocytes locally specify and adapt their phenotype in a multiple sclerosis model. Nat. Neurosci. 21, 1196–1208 (2018).
Ajami, B., Bennett, J. L., Krieger, C., McNagny, K. M. & Rossi, F. M. V. Infiltrating monocytes trigger EAE progression, but do not contribute to the resident microglia pool. Nat. Neurosci. 14, 1142–1150 (2011). This article demonstrates that monocytes do not differentiate into tissue-resident microglia after EAE in mice.
Lagumersindez-Denis, N. et al. Differential contribution of immune effector mechanisms to cortical demyelination in multiple sclerosis. Acta Neuropathol. 134, 15–34 (2017).
Prins, M. et al. Discrepancy in CCL2 and CCR2 expression in white versus grey matter hippocampal lesions of multiple sclerosis patients. Acta Neuropathol. Commun. 2, 98 (2014).
Yamasaki, R. et al. Differential roles of microglia and monocytes in the inflamed central nervous system. J. Exp. Med. 211, 1533–1549 (2014). Together with Ajami et al. (2011), this study shows that monocytes and monocyte-derived cells are greater contributors to EAE pathogenesis and progression than are microglia.
Gao, H. et al. Opposing functions of microglial and macrophagic TNFR2 in the pathogenesis of experimental autoimmune encephalomyelitis. Cell Rep. 18, 198–212 (2017).
Ko, H.-J. et al. GM-CSF-responsive monocyte-derived dendritic cells are pivotal in Th17 pathogenesis. J. Immunol. 192, 2202–2209 (2014).
Miller, S. D., McMahon, E. J., Schreiner, B. & Bailey, S. L. Antigen presentation in the CNS by myeloid dendritic cells drives progression of relapsing experimental autoimmune encephalomyelitis. Ann. N. Y. Acad. Sci. 1103, 179–191 (2007).
Dal-Secco, D. et al. A dynamic spectrum of monocytes arising from the in situ reprogramming of CCR2+ monocytes at a site of sterile injury. J. Exp. Med. 212, 447–456 (2015).
Yona, S. et al. Fate mapping reveals origins and dynamics of monocytes and tissue macrophages under homeostasis. Immunity 38, 79–91 (2013).
Olson, J. K., Girvin, A. M. & Miller, S. D. Direct activation of innate and antigen-presenting functions of microglia following infection with Theiler’s virus. J. Virol. 75, 9780–9789 (2002).
Bohlen, C. J. et al. Diverse requirements for microglial survival, specification, and function revealed by defined-medium cultures. Neuron 94, 759–773 (2017).
Lassmann, H. & Bradl, M. Multiple sclerosis: experimental models and reality. Acta Neuropathol. 133, 223–244 (2017).
Gosselin, D. et al. An environment-dependent transcriptional network specifies human microglia identity. Science 356, eaal3222 (2017).
Masuda, T. et al. Spatial and temporal heterogeneity of mouse and human microglia at single-cell resolution. Nature 566, 388–392 (2019). Together with Gosselin et al. (2017), this study uses single-cell RNA sequencing to compare mouse and human microglia during health and neuroinflammation.
Zrzavy, T. et al. Dominant role of microglial and macrophage innate immune responses in human ischemic infarcts. Brain Pathol. 28, 791–805 (2018).
Buttgereit, A. et al. Sall1 is a transcriptional regulator defining microglia identity and function. Nat. Immunol. 17, 1397–1406 (2016).
Kaiser, T., & Feng, G. Tmem119-EGFP and Tmem119-CreERT2 transgenic mice for labeling and manipulating microglia. bioRxiv https://doi.org/10.1101/624825 (2019).
Ji, Q., Castelli, L. & Goverman, J. M. MHC class I-restricted myelin epitopes are cross-presented by Tip-DCs that promote determinant spreading to CD8+ T cells. Nat. Immunol. 14, 254–261 (2013).
Huber, M. et al. IL-17A secretion by CD8+ T cells supports Th17-mediated autoimmune encephalomyelitis. J. Clin. Invest. 123, 247–260 (2013).
Höftberger, R. et al. Expression of major histocompatibility complex class I molecules on the different cell types in multiple sclerosis lesions. Brain Pathol. 14, 43–50 (2004).
Malo, C. S. et al. MHC class I expression by microglia is required for generating a complete antigen-specific CD8 T cell response in the CNS. J. Immunol. 200, 99.7 (2018).
Denic, A. et al. Deletion of beta-2-microglobulin ameliorates spinal cord lesion load and promotes recovery of brainstem NAA levels in a murine model of multiple sclerosis. Brain Pathol. 22, 698–708 (2012).
Mishra, M. K. & Wee Yong, V. Myeloid cells-targets of medication in multiple sclerosis. Nat. Rev. Neurol. 12, 539–551 (2016).
Tintore, M., Vidal-Jordana, A. & Sastre-Garriga, J. Treatment of multiple sclerosis — success from bench to bedside. Nat. Rev. Neurol. 15, 53–58 (2018). Together with Mishra and Wee Yong (2016), these are excellent reviews that summarize the different therapeutics available for treating MS.
Reindl, M., Di Pauli, F., Rostásy, K. & Berger, T. The spectrum of MOG autoantibody-associated demyelinating diseases. Nat. Rev. Neurol. 9, 455–461 (2013).
Rudick, R. A. & Sandrock, A. Natalizumab: α4-integrin antagonist selective adhesion molecule inhibitors for MS. Expert. Rev. Neurother. 4, 571–580 (2004).
Brinkmann, V. et al. Fingolimod (FTY720): discovery and development of an oral drug to treat multiple sclerosis. Nat. Rev. Drug Discov. 9, 883–897 (2010).
Kappos, L. et al. Siponimod versus placebo in secondary progressive multiple sclerosis (EXPAND): a double-blind, randomised, phase 3 study. Lancet 391, 1263–1273 (2018).
Gentile, A. et al. Siponimod (BAF312) prevents synaptic neurodegeneration in experimental multiple sclerosis. J. Neuroinflammation 13, 207 (2016).
Hinkerohe, D. et al. Dexamethasone prevents LPS-induced microglial activation and astroglial impairment in an experimental bacterial meningitis co-culture model. Brain Res. 1329, 45–54 (2010).
Huo, Y., Rangarajan, P., Ling, E. A. & Dheen, S. T. Dexamethasone inhibits the Nox-dependent ROS production via suppression of MKP-1-dependent MAPK pathways in activated microglia. BMC Neurosci. 12, 49 (2011).
Chao, C. C. et al. Cytokine release from microglia: differential inhibition by pentoxifylline and dexamethasone. J. Infect. Dis. 166, 847–853 (1992).
Kiefer, R. & Kreutzberg, G. W. Effects of dexamethasone on microglial activation in vivo: selective downregulation of major histocompatibility complex class II expression in regenerating facial nucleus. J. Neuroimmunol. 34, 99–108 (1991).
Kawanokuchi, J., Mizuno, T., Kato, H., Mitsuma, N. & Suzumura, A. Effects of interferon-β on microglial functions as inflammatory and antigen presenting cells in the central nervous system. Neuropharmacol. 46, 734–742 (2004).
Teige, I., Liu, Y. & Issazadeh-Navikas, S. IFN-beta inhibits T cell activation capacity of central nervous system APCs. J. Immunol. 177, 3542–3553 (2006).
Ross, T. M. et al. Intranasal administration of interferon beta bypasses the blood-brain barrier to target the central nervous system and cervical lymph nodes: a non-invasive treatment strategy for multiple sclerosis. J. Neuroimmunol. 151, 66–77 (2004).
Schreiner, B. et al. Interferon-β enhances monocyte and dendritic cell expression of B7-H1 (PD-L1), a strong inhibitor of autologous T-cell activation: relevance for the immune modulatory effect in multiple sclerosis. J. Neuroimmunol. 155, 172–182 (2004).
Hallal-Longo, D. E. M. et al. Diminished myelin-specific T cell activation associated with increase in CTLA4 and Fas molecules in multiple sclerosis patients treated with IFN-β. J. Interf. Cytokine Res. 27, 865–873 (2007).
Havrdová, E. et al. Activity of secukinumab, an anti-IL-17A antibody, on brain lesions in RRMS: results from a randomized, proof-of-concept study. J. Neurol. 263, 1287–1295 (2016).
Van Oosten, B. W. et al. Increased MRI activity and immune activation in two multiple sclerosis patients treated with the monoclonal anti-tumor necrosis factor antibody cA2. Neurology 47, 1531–1534 (1996).
The Lenercept multiple sclerosis study group. TNF neutralization in MS: results of a randomized, placebo-controlled multicenter study. Neurology 53, 457–465 (1999).
Araki, M., Nakamura, M., Sato, W., Takahashi, Y. & Yamamura, T. Potential benefits of the anti-IL-6 receptor antibody tocilizumab in multiple sclerosis patients with high plasmablast frequency. J. Neurol. Sci. 381, 131 (2017).
Beauchemin, P. & Carruthers, R. MS arising during tocilizumab therapy for rheumatoid arthritis. Mult. Scler. 22, 254–256 (2016).
Skurkovich, S. et al. Randomized study of antibodies to IFN-γ and TNF-α in secondary progressive multiple sclerosis. Mult. Scler. 7, 277–284 (2001).
Naves, R. et al. The interdependent, overlapping, and differential roles of type I and II IFNs in the pathogenesis of experimental autoimmune encephalomyelitis. J. Immunol. 191, 2967–2977 (2013).
Arellano, G., Ottum, P. A., Reyes, L. I., Burgos, P. I. & Naves, R. Stage-specific role of interferon-gamma in experimental autoimmune encephalomyelitis and multiple sclerosis. Front. Immunol. 6, 492 (2015).
Wilms, H. et al. Dimethylfumarate inhibits microglial and astrocytic inflammation by suppressing the synthesis of nitric oxide, IL-1β, TNF-α and IL-6 in an in-vitro model of brain inflammation. J. Neuroinflammation 7, 30 (2010).
Kornberg, M. D. et al. Dimethyl fumarate targets GAPDH and aerobic glycolysis to modulate immunity. Science 360, 449–453 (2018).
Pul, R. et al. Glatiramer acetate modulates TNF-α and IL-10 secretion in microglia and promotes their phagocytic activity. J. Neuroimmune Pharmacol. 6, 381–388 (2011).
Ratchford, J. N. et al. Decreased microglial activation in MS patients treated with glatiramer acetate. J. Neurol. 259, 1199–1205 (2012).
Wostradowski, T. et al. In vitro evaluation of physiologically relevant concentrations of teriflunomide on activation and proliferation of primary rodent microglia. J. Neuroinflammation 13, 250 (2016).
Korn, T., Magnus, T., Toyka, K. & Jung, S. Modulation of effector cell functions in experimental autoimmune encephalomyelitis by leflunomide — mechanisms independent of pyrimidine depletion. J. Leukoc. Biol. 76, 950–960 (2004).
Kobayashi, K. et al. Minocycline selectively inhibits M1 polarization of microglia. Cell Death Dis. 4, e525 (2013).
Koch, M. W. et al. Hydroxychloroquine reduces microglial activity and attenuates experimental autoimmune encephalomyelitis. J. Neurol. Sci. 358, 131–137 (2015).
Giles, D. A., Duncker, P. C., Wilkinson, N. M., Washnock-Schmid, J. M. & Segal, B. M. CNS-resident classical DCs play a critical role in CNS autoimmune disease. J. Clin. Invest. 128, 5322–5334 (2018).
Agrawal, S. M., Silva, C., Tourtellotte, W. W. & Yong, V. W. EMMPRIN: a novel regulator of leukocyte transmigration into the CNS in multiple sclerosis and experimental autoimmune encephalomyelitis. J. Neurosci. 31, 669–677 (2011).
Acknowledgements
The authors’ research is supported by operating grants from the Alberta Innovates Health Solutions CRIO Team program, the MS Society of Canada and the Canadian Institutes of Health Research. Y.D. acknowledges postdoctoral fellowship support from the Alberta MS Collaboration. V.W.Y. has salary support from the Canada Research Chair (Tier 1) program.
Author information
Authors and Affiliations
Contributions
Both authors conceptualized the manuscript. Y.D. wrote the initial draft. V.W.Y. supervised, reviewed and edited the manuscript. Both authors finalized the manuscript.
Corresponding author
Ethics declarations
Competing interests
Y.D. declares no competing interests. V.W.Y. has received speaker’s and advisory board honoraria from Biogen, EMD Serono, Novartis, Roche, Sanofi-Genzyme and Teva, and educational grants from Biogen, EMD Serono, Novartis, Roche, Sanofi-Genzyme and Teva.
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Peer review information
Nature Reviews Neurology thanks R. Liblau, T. Owens and the other, anonymous, reviewer(s) for their contribution to the peer review of this work.
Glossary
- Diapedesis
-
The migration of leukocytes across the endothelial wall of the blood vessel into the tissue during inflammation.
- Homing receptors
-
Cell adhesion molecules expressed on T cells that recognize their respective ligands expressed by the target tissues or cells, and which facilitate T cell trafficking.
- Zitter strain background
-
A strain of inbred rats that carry the zitter mutation in the Atrn gene and develop neuropathological features of multiple sclerosis, including neurodegeneration, hypomyelination and microgliosis.
- γδ T cells
-
A small set of T cells with unconventional γδ T cell receptors that can recognize non-MHC-restricted antigens such as microbial metabolites, lipids and markers of cell stress.
- Parabiosis
-
An experimental technique in which the vasculature of two animals are surgically united, often used together with fate mapping mouse models to study the migration of immune cells into different tissues.
Rights and permissions
About this article
Cite this article
Dong, Y., Yong, V.W. When encephalitogenic T cells collaborate with microglia in multiple sclerosis. Nat Rev Neurol 15, 704–717 (2019). https://doi.org/10.1038/s41582-019-0253-6
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/s41582-019-0253-6
- Springer Nature Limited
This article is cited by
-
Border-associated macrophages in the central nervous system
Journal of Neuroinflammation (2024)
-
Granzyme B + CD8 + T cells with terminal differentiated effector signature determine multiple sclerosis progression
Journal of Neuroinflammation (2023)
-
Spatial transcriptomics and neurofilament light chain reveal changes in lesion patterns in murine autoimmune neuroinflammation
Journal of Neuroinflammation (2023)
-
Bruton tyrosine kinase inhibitors for multiple sclerosis
Nature Reviews Neurology (2023)
-
Gastrointestinal Tract, Microbiota and Multiple Sclerosis (MS) and the Link Between Gut Microbiota and CNS
Current Microbiology (2023)