Abstract
Objective
To determine if prophylactic indomethacin (PINDO) decreases serious pulmonary hemorrhages in infants <28 weeks.
Study design
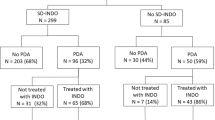
Intention-to-treat analysis of 615 consecutively admitted infants during four alternating protocol-driven epochs of PINDO or expectant patent ductus arteriosus (PDA) management.
Results
41/615 (6.7%) developed serious pulmonary hemorrhage at 2 (1, 3) days (median (IQR)). In unadjusted and adjusted multivariable models, infants born in a PINDO epoch had significantly lower incidences of pulmonary hemorrhage and pulmonary hemorrhage or death before 7 days. There were less moderate/large PDA during PINDO epochs. The associations between PINDO and pulmonary hemorrhage and pulmonary hemorrhage/death were no longer significant when presence of a PDA was included in the analyses. There was no apparent association between PINDO epochs and the incidence of serious intraventricular hemorrhages.
Conclusion
Even though PINDO no longer appears to affect the incidence of sIVH it still is associated with a lower incidence of pulmonary hemorrhage.
Similar content being viewed by others
Data availability
The datasets generated and/or analyzed during the current study are available from the corresponding author on reasonable request.
References
Cole VA, Normand IC, Reynolds EO, Rivers RP. Pathogenesis of hemorrhagic pulmonary edema and massive pulmonary hemorrhage in the newborn. Pediatrics. 1973;51:175–87.
Ferreira CH, Carmona F, Martinez FE. Prevalence, risk factors and outcomes associated with pulmonary hemorrhage in newborns. J Pediatr. 2014;90:316–22.
Chen YY, Wang HP, Lin SM, Chang JT, Hsieh KS, Huang FK, et al. Pulmonary hemorrhage in very low-birthweight infants: risk factors and management. Pediatr Int. 2012;54:743–7.
Rakza T, Magnenant E, Klosowski S, Tourneux P, Bachiri A, Storme L. Early hemodynamic consequences of patent ductus arteriosus in preterm infants with intrauterine growth restriction. J Pediatr. 2007;151:624–8.
Alpan G, Clyman RI. Cardiovascular effects of surfactant replacement with special reference to the patent ductus arteriosus. In: Robertson B, Taeusch HW, editors. Surfactant therapy for lung disease: lung biology in health and disease. 84. New York: Marcel Dekker, Inc.; 1995. p. 531–45.
Perez Fontan JJ, Clyman RI, Mauray F, Heymann MA, Roman C. Respiratory effects of a patent ductus arteriosus in premature newborn lambs. J Appl Physiol. 1987;63:2315–24.
Alpan G, Scheerer R, Bland RD, Clyman R. Patent ductus arteriosus increases lung fluid filtration in preterm lambs. Pediatr Res. 1991;30:616–21.
Lewis AB, Heymann MA, Rudolph AM. Gestational changes in pulmonary vascular responses in fetal lambs in utero. Circ Res. 1976;39:536–41.
Al Faleh K, Smyth J, Roberts R, Solimano A, Asztalos E, Schmidt B. Prevention and 18-month outcome of serious pulmonary hemorrhage in extremely low birth weight infants: results from the triall of indomethacin prophylaxis in preterms. Pediatrics. 2008;121:e233–8.
Kluckow M, Jeffery M, Gill A, Evans N. A randomised placebo-controlled trial of early treatment of the patent ductus arteriosus. Arch Dis Child Fetal Neonatal Ed. 2014;99:F99–104.
Aranda JV, Clyman R, Cox B, Van Overmeire B, Wozniak P, Sosenko I, et al. A randomized, double-blind, placebo-controlled trial on intravenous ibuprofen L-lysine for the early closure of nonsymptomatic patent ductus arteriosus within 72 h of birth in extremely low-birth-weight infants. Am J Perinatol. 2009;26:235–45.
Roze JC, Cambonie G, Le Thuaut A, Debillon T, Ligi I, Gascoin G, et al. Effect of early targeted treatment of ductus arteriosus with ibuprofen on survival without cerebral Palsy at 2 Years in infants with extreme prematurity: a randomized clinical trial. J Pediatr. 2021;233:33–42.e2.
Kappico JM, Cayabyab R, Ebrahimi M, Uzunyan MY, Barton L, Siassi B, et al. Pulmonary hemorrhage in extremely low birth weight infants: significance of the size of left to right shunting through a valve incompetent patent foramen ovale. J Perinatol. 2022;42:1233–7.
Bussmann N, Breatnach C, Levy PT, McCallion N, Franklin O, El-Khuffash A. Early diastolic dysfunction and respiratory morbidity in premature infants: an observational study. J Perinatol. 2018;38:1205–11.
West JB, Mathieu-Costello O. Stress failure of pulmonary capillaries: role in lung and heart disease. Lancet. 1992;340:762–7.
Scholl JE, Yanowitz TD. Pulmonary hemorrhage in very low birth weight infants: a case-control analysis. J Pediatr. 2015;166:1083–4.
Li J, Xia H, Ye L, Li X, Zhang Z. Exploring prediction model and survival strategies for pulmonary hemorrhage in premature infants: a single-center, retrospective study. Transl Pediatr. 2021;10:1324–32.
Mehta R, Petrova A. Intrauterine neutrophil activation is associated with pulmonary haemorrhage in preterm infants. Arch Dis Child Fetal Neonatal Ed. 2006;91:F415–8.
McCurnin D, Seidner S, Chang LY, Waleh N, Ikegami M, Petershack J, et al. Ibuprofen-induced patent ductus arteriosus closure: physiologic, histologic, and biochemical effects on the premature lung. Pediatrics. 2008;121:945–56.
Domanico RS, Waldman JD, Lester LA, McPhillips HA, Catrambone JE, Covert RF. Prophylactic indomethacin reduces the incidence of pulmonary hemorrhage and patent ductus arteriosus in surfactant treated infants < 1250 grams. Pediatr Res. 1994;35:331.
Schmidt B, Davis P, Moddemann D, Ohlsson A, Roberts RS, Saigal S, et al. Long-term effects of indomethacin prophylaxis in extremely-low-birth- weight infants. N. Engl J Med. 2001;344:1966–72.
Ment LR, Duncan CC, Ehrenkranz RA, Kleinman CS, Taylor KJW, Scott DT, et al. Randomized low-dose indomethacin trial for prevention of intraventricular hemorrhage in very low birth weight neonates. J Pediatr. 1988;112:948–55.
Ment LR, Duncan CC, Ehrenkranz RA, Kleinman CS, Pitt BR, Taylor KJW, et al. Randomized indomethacin trial for prevention of intraventricular hemorrhage in very low birth weight infants. J Pediatr. 1985;107:937–43.
Fowlie PW, Davis PG, McGuire W. Prophylactic intravenous indomethacin for preventing mortality and morbidity in preterm infants. Cochrane Database Syst Rev. 2010. https://doi.org/10.1002/14651858.CD000174.pub2.
Dahlgren N, Nilsson B, Sakabe T, Siesjo BK. The effect of indomethacin on cerebral blood flow and oxygen consumption in the rat at normal and increased carbon dioxide tensions. Acta Physiol Scand. 1981;111:475–85.
Ment LR, Stewart WB, Ardito TA, Huang E, Madri JA. Indomethacin promotes germinal matrix microvessel maturation in the newborn beagle pup. Stroke. 1992;23:1132–7.
Ment LR, Stewart WB, Scott DT, Duncan CC. Beagle puppy model of intraventricular hemorrhage: randomized indomethacin prevention trial. Neurology. 1983;33:179–84.
Ballabh P, Xu H, Hu F, Braun A, Smith K, Rivera A, et al. Angiogenic inhibition reduces germinal matrix hemorrhage. Nat Med. 2007;13:477–85.
Ballabh P. Pathogenesis and prevention of intraventricular hemorrhage. Clin Perinatol. 2014;41:47–67.
Qureshi M, Shah PS, Abdelgadir D, Ye XY, Afifi J, Yuen R, et al. Gestational age-dependent variations in effects of prophylactic indomethacin on brain injury and intestinal injury. J Pediatr. 2021;235:26–33.e2.
Chawla S, Natarajan G, Laptook AR, Chowdhury D, Bell EF, Ambalavanan N, et al. Model for severe intracranial hemorrhage and role of early indomethacin in extreme preterm infants. Pediatr Res. 2022;92:1648–56.
Kandraju H, Kanungo J, Lee KS, Daspal S, Adie MA, Dorling J, et al. Association of Co-exposure of antenatal steroid and prophylactic indomethacin with spontaneous intestinal perforation. J Pediatr. 2021;235:34–41.e1.
Miller CJ, Prusakov P, Magers J, Speaks S, Sacic H, Escobar K, et al. Effects of prophylactic indomethacin on intraventricular hemorrhage and adverse outcomes in neonatal intensive care unit. J Perinatol. 2022;42:1644–8.
Pan I, Shah PA, Singh J, Kelly KN, Bondi DS. Comparison of neonatal outcomes with and without prophylaxis with indomethacin in premature neonates. J Pediatr Pharm Ther. 2021;26:478–83.
Abdi HH, Backes CH, Ball MK, Talavera-Barber MM, Klebanoff MA, Jadcherla SR, et al. Prophylactic Indomethacin in extremely preterm infants: association with death or BPD and observed early serum creatinine levels. J Perinatol. 2021;41:749–55.
Handley SC, Passarella M, Lee HC, Lorch SA. Incidence trends and risk factor variation in severe intraventricular hemorrhage across a population based cohort. J Pediatr. 2018;200:24–9.e3.
Kramer KP, Minot K, Butler C, Haynes K, Mason A, Nguyen L, et al. Reduction of severe intraventricular hemorrhage in preterm infants: a quality improvement project. Pediatrics. 2022;149:e2021050652.
Koch J, Hensley G, Roy L, Brown S, Ramaciotti C, Rosenfeld CR. Prevalence of spontaneous closure of the ductus arteriosus in neonates at a birth weight of 1000 grams or less. Pediatrics. 2006;117:1113–21.
Jensen EA, Dysart KC, Gantz MG, Carper B, Higgins RD, Keszler M, et al. Association between use of prophylactic indomethacin and the risk for bronchopulmonary dysplasia in extremely preterm infants. J Pediatr. 2017;186:34–40.
Liebowitz M, Clyman RI. Prophylactic indomethacin compared with delayed conservative management of the patent ductus arteriosus in extremely preterm infants: effects on neonatal outcomes. J Pediatr. 2017;187:119–26.
Clyman RI, Wickremasinghe A, Merritt TA, Solomon T, McNamara P, Jain A, et al. Hypotension following patent ductus arteriosus ligation: the role of adrenal hormones. J Pediatr. 2014;164:1449–55.
Liebowitz MC, Clyman RI. Predicting the need for home oxygen therapy in preterm infants born before 28 Weeks’ gestation. Am J Perinatol. 2016;33:34–9.
Fenton TR, Kim JH. A systematic review and meta-analysis to revise the Fenton growth chart for preterm infants. BMC Pediatr. 2013;13:59.
Papile LA, Burstein J, Burstein R, Koffler H. Incidence and evolution of subependymal and intraventricular hemorrhage: a study of infants with birth weights less than 1500 gm. J Pediatr. 1978;92:529–34.
Clyman RI, Hills NK. The effect of prolonged tracheal intubation on the association between patent ductus arteriosus and bronchopulmonary dysplasia (grades 2 and 3). J Perinatol. 2020;40:1358–65.
Clyman RI, Kaempf J, Liebowitz M, Erdeve O, Bulbul A, Hakansson S, et al. Prolonged tracheal intubation and the association between patent ductus arteriosus and bronchopulmonary dysplasia: a secondary analysis of the PDA-TOLERATE trial. J Pediatr. 2021;229:283–8.e2.
Clyman RI, Liebowitz M, Kaempf J, Erdeve O, Bulbul A, Hakansson S, et al. PDA-TOLERATE trial: an exploratory randomized controlled trial of treatment of moderate-to-large patent ductus arteriosus at 1 week of age. J Pediatr. 2019;205:41–8.e6.
Acknowledgements
We would like to thank Drs. Mark Cocalis, Michael Brook, Anita Moon-Grady, Shabnam Peyvandi, Hythem Nawaytou, and Nicole Cresalia for their expert help in reading and interpreting the echocardiograms, and to the neonatal faculty, fellows, nurses, respiratory therapists, and dieticians for their excellent care and their commitment to the nursery’s quality improvement projects and its consensus driven protocols. This work was supported by a grant from the U.S. Public Health Service National Heart, Lung, and Blood Institute (HL109199).
Funding
This work was supported by a grant from the U.S. Public Health Service National Heart, Lung, and Blood Institute (HL109199).
Author information
Authors and Affiliations
Contributions
Ronald Clyman was the principal investigator of the overall study and was involved with the conceptualization and design of the study, study oversight, funding acquisition, data acquisition and formal analysis, writing of the first draft in addition to performing all 4 of the following tasks: (1) participated in the design of the study, acquired patient data, and submitted IRB proposal. (2) reviewed, revised, and edited the manuscript. (3) gave approval to the final version of the manuscript. (4) agreed to be accountable for all aspects of the work. Nancy K. Hills performed the statistical consultation and analyses in addition to performing all 4 of the following tasks: (1) participated in the design of the original protocol and performed statistical analyses. (2) reviewed, revised, and edited the manuscript. (3) gave approval to the final version of the manuscript. (4) agreed to be accountable for all aspects of the work.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Clyman, R.I., Hills, N.K. Prophylactic indomethacin and the risk of serious pulmonary hemorrhages in preterm infants less than 28 weeks’ gestation. J Perinatol (2024). https://doi.org/10.1038/s41372-024-01971-x
Received:
Revised:
Accepted:
Published:
DOI: https://doi.org/10.1038/s41372-024-01971-x
- Springer Nature America, Inc.