Abstract
Background
Optimal CPAP strategy to prevent CPAP failure defined as need for endotracheal intubation is unknown.
Objective
To evaluate the risk of CPAP failure in infants treated with high vs low CPAP strategy while receiving aerosolized calfactant in the AERO-02 clinical trial and AERO-03 expanded access program.
Methods
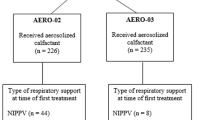
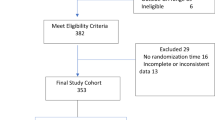
Infants born between 29 0/7 to 36 6/7 weeks were included. Comparisons were made between low and high CPAP groups (Low, 4–7 cm H2O; High, 8–10 cm H2O).
Results
CPAP failure and pneumothorax were not different between the groups. Odds of CPAP failure were not different after adjustment for baseline characteristics (OR = 0.61; 95% CI: 0.29, 1.24).
Conclusion
We found no difference in CPAP failure among infants who received aerosolized calfactant that were treated with high vs low CPAP strategy. Efficacy of high CPAP strategy with aerosolized surfactant treatment needs to be evaluated in future studies.

Similar content being viewed by others
Data availability
The data that support the findings of this study are available from the corresponding author, [DK], upon request.
References
Polin RA, Carlo WA. Committee on Fetus and Newborn; American Academy of Pediatrics. Surfactant replacement therapy for preterm and term neonates with respiratory distress. Pediatrics. 2014;133:156–63.
Sweet DG, Carnielli V, Greisen G, Hallman M, Klebermass-Schrehof K, Ozek E, et al. European Consensus guidelines on the management of respiratory distress syndrome—2022 update. Neonatology. 2023;120:3–23.
Ng EH, Shah V, Fetus and Newborn Committee. Canadian Paediatric Society. Guidelines for surfactant replacement therapy in neonates. Paediatr Child Health. 2021;26:35–49.
Bhandari V, Black R, Gandhi B, Hogue S, Kakkilaya V, Mikhael M, et al. RDS-NExT workshop: consensus statements for the use of surfactant in preterm neonates with RDS. J Perinatol. 2023;43:982–90.
Banerjee S, Fernandez R, Fox GF, Goss KCW, Mactier H, Reynolds P, et al. Surfactant replacement therapy for respiratory distress syndrome in preterm infants: United Kingdom national consensus. Pediatr Res. 2019;86:12–14.
Aly H, Mohamed MA, Wung JT. Surfactant and continuous positive airway pressure for the prevention of chronic lung disease: history, reality, and new challenges. Semin Fetal Neonat Med 2017;22:348–53.
Dargaville PA, Gerber A, Johansson S, De Paoli AG, Kamlin CO, Orsini F, et al. Australian, New Zealand Neonatal Network: Incidence and outcome of CPAP failure in preterm infants. Pediatrics. 2016;138:e20153985.
Dargaville PA, Aiyappan A, De Paoli AG, Dalton RGB, Kuschel CA, Orsini F, et al. Continuous positive airway pressure failure in preterm infants: incidence, predictors and consequences. Neonatology. 2013;104:8–14.
Glaser K, Bamat NA, Wright CJ. Can we balance early exogenous surfactant therapy and non-invasive respiratory support to optimize outcomes in extremely preterm infants? A nuanced review of the current literature. Arch Dis Child Fetal Neonatal Ed. 2023;108:554–60.
Cummings JJ, Gerday E, Minton S, Katheria A, Albert G, Flores-Torres J, et al. Aerosolized calfactant for newborns with respiratory distress: a randomized trial. Pediatrics. 2020;146:e20193967.
Kaluarachchi DC, Zapata HA, Becker HL, Lasarev MR, Fort P, Guthrie SO. Response to aerosolized calfactant in infants with respiratory distress syndrome; a post-hoc analysis of AERO-02 trial. Perinatol. 2023;43:998–1003.
Hojnicki M, Zapata HA, Kaluarachchi DC, Fort P, Minton S, Albert G, et al. Predictors of successful treatment of respiratory distress with aerosolized calfactant. J Perinatol. 2023;43:991–7.
Waitz M, Engel C, Schloesser R, Rochwalsky U, Meyer S, Larsen A, et al. Application of two different nasal CPAP levels for the treatment of respiratory distress syndrome in preterm infants-“The OPTTIMMAL-Trial”-Optimizing PEEP To The IMMAture Lungs: study protocol of a randomized controlled trial. Trials. 2020;21:822.
R Core Team. R: a language and environment for statistical computing. R Foundation for Statistical Computing, Vienna, Austria (2022).
Mulrooney N, Champion Z, Moss TJ, Nitsos I, Ikemagi M, Jobe AH. Surfactant and physiologic responses of preterm lambs to continuous positive airway pressure. Am J Respir Crit Care Med. 2005;171:488–93.
Probyn ME, Hooper SB, Dargaville PA, McCallion N, Crossley K, Harding R, et al. Positive end expiratory pressure during resuscitation of premature lambs rapidly improves blood gases without adversely affecting arterial pressure. Pediatr Res. 2004;56:198–204.
Gregory GA, Kitterman JA, Phibbs RH, Tooley WH, Hamilton WK. Treatment of the idiopathic respiratory distress syndrome with continuous positive airway pressure. N Engl J Med. 1971;284:1333–40.
Kidman AM, Manley BJ, Boland RA, Malhotra A, Donath SM, Beker F, et al. Higher versus lower nasal continuous positive airway pressure for extubation of extremely preterm infants in Australia (ÉCLAT): a multicentre, randomised, superiority trial. https://doi.org/10.1016/S2352-4642(23)00235-3.
Buzzella B, Claure N, D’Ugard C, Bancalari E. A randomized controlled trial of two nasal continuous positive airway pressure levels after extubation in preterm infants. J Pediatr. 2014;164:46–51.
Morley CJ, Davis PG, Doyle LW, Brion LP, Hascoet JM, Carlin JB. Nasal CPAP or intubation at birth for very preterm infants. N Engl J Med. 2008;358:700–8.
Acknowledgements
All AERO-02 and AERO-03 study investigators, Melissa Brown, Corey Commaroto, ONY Biotech Inc.
Author information
Authors and Affiliations
Contributions
Dinushan Kaluarachchi: Dr. Kaluarachchi designed the study, drafted the project proposal, obtained data, drafted and approved final manuscript as submitted. Henry A. Zapata: Dr. Zapata involved in designing of the study, drafted the project proposal, reviewed and approved final manuscript as submitted. Erick Gerday: Dr. Gerday involved in designing of the study, reviewed and approved final manuscript as submitted. Timothy Bahr: Dr. Bhar involved in designing of the study, reviewed and approved final manuscript as submitted. Michael R. Lasarev: Mr. Lasarev involved in designing of the study, did the data analysis, reviewed and approved final manuscript as submitted. Scott O. Guthrie: Dr. Guthrie involved in designing of the study, reviewed and approved final manuscript as submitted. Stephen Minton: Dr. Minton involved in designing of the study, reviewed and approved final manuscript as submitted. All authors approved the final manuscript as submitted and agree to be accountable for all aspects of the work.
Corresponding author
Ethics declarations
Competing interests
DK and SG serve as consultants for ONY Biotech. The remaining authors declare that the research was conducted in the absence of any commercial or financial relationships that could be construed as a potential conflict of interest.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Kaluarachchi, D.C., Gerday, E., Bahr, T. et al. High vs low CPAP strategy with aerosolized calfactant in preterm infants with respiratory distress syndrome. J Perinatol (2024). https://doi.org/10.1038/s41372-024-01959-7
Received:
Revised:
Accepted:
Published:
DOI: https://doi.org/10.1038/s41372-024-01959-7
- Springer Nature America, Inc.