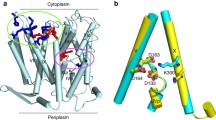
Abstract
The sodium-potassium (Na/K) pump is a P-type ATPase that generates Na+ and K+ concentration gradients across the cell membrane. For each hydrolyzed ATP molecule, the pump extrudes three Na+ and imports two K+ by alternating between outward- and inward-facing conformations that preferentially bind K+ or Na+, respectively. Remarkably, the selective K+ and Na+ binding sites share several residues, and how the pump is able to achieve the selectivity required for the functional cycle is unclear. Here, free energy–perturbation molecular dynamics (FEP/MD) simulations based on the crystal structures of the Na/K pump in a K+-loaded state (E2·Pi) reveal that protonation of the high-field acidic side chains involved in the binding sites is crucial to achieving the proper K+ selectivity. This prediction is tested with electrophysiological experiments showing that the selectivity of the E2P state for K+ over Na+ is affected by extracellular pH.


Similar content being viewed by others
References
Jorgensen, P.L., Hakansson, K.O. & Karlish, S.J.D. Structure and mechanism of Na+,K+-ATPase: functional sites and their interactions. Annu. Rev. Physiol. 65, 817–849 (2003).
Post, R.L., Sen, A.K. & Rosenthal, A.S. A phosphorylated intermediate in adenosine triphosphate-dependent sodium and potassium transport across kidney membranes. J. Biol. Chem. 240, 1437–1445 (1965).
Jardetzky, O. Simple allosteric model for membrane pumps. Nature 211, 969–970 (1966).
Albers, R.W. Biochemical aspects of active transport. Annu. Rev. Biochem. 36, 727–756 (1967).
Ogawa, H. & Toyoshima, C. Homology modeling of the cation binding sites of Na+/K+-ATPase. Proc. Natl. Acad. Sci. USA 99, 15977–15982 (2002).
Morth, J.P. et al. Crystal structure of the sodium-potassium pump. Nature 450, 1043–1049 (2007).
Shinoda, T., Ogawa, H., Cornelius, F. & Toyoshima, C. Crystal structure of the sodium-potassium pump at 2.4 Å resolution. Nature 459, 446–450 (2009).
Perrin, C.L. & Nielson, J.B. “Strong” hydrogen bonds in chemistry and biology. Annu. Rev. Phys. Chem. 48, 511–544 (1997).
Obara, K. et al. Structural role of countertransport revealed in Ca pump crystal structure in the absence of Ca. Proc. Natl. Acad. Sci. USA 102, 14489–14496 (2005).
Sugita, Y., Miyashita, N., Ikeguchi, M., Kidera, A. & Toyoshima, C. Protonation of the acidic residues in the transmembrane cation-binding sites of the Ca pump. J. Am. Chem. Soc. 127, 6150–6151 (2005).
Hauser, K. & Barth, A. Side-chain protonation and mobility in the sarcoplasmic reticulum Ca-ATPase: Implications for proton countertransport and Ca release. Biophys. J. 93, 3259–3270 (2007).
Andersson, J., Hauser, K., Karjalainen, E.L. & Barth, A. Protonation and hydrogen bonding of Ca site residues in the E2P phosphoenzyme intermediate of sarcoplasmic reticulum Ca-ATPase studied by a combination of infrared spectroscopy and electrostatic calculations. Biophys. J. 94, 600–611 (2008).
Weidemuller, C. & Hauser, K. Ion transport and energy transduction of P-type ATPases: implications from electrostatic calculations. Biochim. Biophys Acta 1787, 721–729 (2009).
Lauger, P. Electrogenic Ion Pumps 1st edn. (Sinauer Associates, Sunderland, Massachusetts, USA, 1991).
Bublitz, M., Poulsen, H., Morth, J. & Nissen, P. In and out of the cation pumps: P-Type ATPase structure revisited. Curr. Opin. Struct. Biol. 20, 431–439 (2010).
Yu, H., Noskov, S.Y. & Roux, B. Two mechanisms of ion selectivity in protein binding sites. Proc. Natl. Acad. Sci. USA 107, 20329–20334 (2010).
Roux, B. Exploring the ion selectivity properties of a large number of simplified binding site models. Biophys. J. 98, 2877–2885 (2010).
Ratheal, I.M. et al. Selectivity of externally facing ion-binding sites in the Na+/K+ pump to alkali metals and organic cations. Proc. Natl. Acad. Sci. USA 107, 18718–18723 (2010).
Noskov, S.Y., Bernèche, S. & Roux, B. Control of ion selectivity in potassium channels by electrostatic and dynamic properties of carbonyl ligands. Nature 431, 830–834 (2004).
Noskov, S.Y. & Roux, B. Importance of hydration and dynamics on the selectivity of the KcsA and NaK channels. J. Gen. Physiol. 129, 135–143 (2007).
Noskov, S.Y. & Roux, B. Control of ion selectivity in LeuT: two Na+ binding sites with two different mechanisms. J. Mol. Biol. 377, 804–818 (2008).
Skou, J.C. Effects of ATP on the intermediary steps of the reaction of the Na+/K+-ATPase. IV. Effect of ATP on K0.5 for Na+ and on hydrolysis at different pH and temperature. Biochim. Biophys. Acta 567, 421–435 (1979).
Skou, J.C. & Esmann, M. Effects of ATP and protons on the Na:K selectivity of the Na+/K+-ATPase studied by ligand effects on intrinsic and extrinsic fluorescence. Biochim. Biophys. Acta 601, 386–402 (1980).
Breitwieser, G.E., Altamirano, A.A. & Russell, J.M. Effects of pH changes on sodium pump fluxes in squid giant axon. Am. J. Physiol. 253, C547–C554 (1987).
Salonikidis, P.S., Kirichenko, S.N., Tatjanenko, L.V., Schwarz, W. & Vasilets, L.A. Extracellular pH modulates kinetics of the Na+,K+-ATPase. Biochim. Biophys. Acta 1509, 496–504 (2000).
Milanick, M.A. & Arnett, K.L. Extracellular protons regulate the extracellular cation selectivity of the sodium pump. J. Gen. Physiol. 120, 497–508 (2002).
Kuntzweiler, T.A., Wallick, E.T., Johnson, C.L. & Lingrel, J.B. Glutamic acid 327 in the sheep α1 isoform of Na+,K+-ATPase stabilizes a K+-induced conformational change. J. Biol. Chem. 270, 2993–3000 (1995).
Koenderink, J.B. et al. Electrophysiological analysis of the mutated Na+,K+-ATPase cation binding pocket. J. Biol. Chem. 278, 51213–51222 (2003).
Koenderink, J.B., Swarts, H.G., Hermsen, H.P., Willems, P.H. & De Pont, J.J. Mutation of aspartate 804 of Na+,K+-ATPase modifies the cation binding pocket and thereby generates a high Na+-ATPase activity. Biochemistry 39, 9959–9966 (2000).
Toyoshima, C., Nomura, H. & Tsuda, T. Lumenal gating mechanism revealed in calcium pump crystal structures with phosphate analogues. Nature 432, 361–368 (2004).
Toyoshima, C. Structural aspects of ion pumping by Ca2+-ATPase of sarcoplasmic reticulum. Arch. Biochem. Biophys. 476, 3–11 (2008).
Feng, J. & Lingrel, J.B. Functional consequences of substitutions of the carboxyl residue glutamate 779 of the Na+,K+-ATPase. Cell Mol. Biol. Res. 41, 29–37 (1995).
Poulsen, H. et al. Neurological disease mutations compromise a C-terminal ion pathway in the Na+/K+-ATPase. Nature 467, 99–102 (2010).
Song, Y., Mao, J.J. & Gunner, M.R. MCCE2: Improving protein pKa calculations with extensive side chain rotamer sampling. J. Comput. Chem. 30, 2231–2247 (2009).
Li, H., Robertson, A.D. & Jensen, J.H. Very fast empirical prediction and rationalization of protein pKa values. Proteins 61, 704–721 (2005).
Brooks, B.R. et al. CHARMM: the biomolecular simulation program. J. Comput. Chem. 30, 1545–1614 (2009).
MacKerell, A.D. Jr. et al. All-atom empirical potential for molecular modeling and dynamics studies of proteins. J. Phys. Chem. B 102, 3586–3616 (1998).
Im, W., Berneche, S. & Roux, B. Generalized solvent boundary potential for computer simulations. J. Chem. Phys. 114, 2924–2937 (2001).
Nina, M., Beglov, D. & Roux, B. Atomic radii for continuum electrostatics calculations based on molecular dynamics free energy simulations. J. Phys. Chem. B 101, 5239–5248 (1997).
Davies, M.N., Toseland, C.P., Moss, D.S. & Flower, D.R. Benchmarking pKa prediction. BMC Biochem. 7, 18 (2006).
Yaragatupalli, S., Olivera, J.F., Gatto, C. & Artigas, P. Altered Na+ transport after an intracellular alpha-subunit deletion reveals strict external sequential release of Na+ from the Na+/K+ pump. Proc. Natl. Acad. Sci. USA 106, 15507–15512 (2009).
Acknowledgements
This work was supported by US National Institutes of Health grant GM062342 (H.Y. and B.R.) and by American Heart Association grant BGIA2140172 (P.A.).
Author information
Authors and Affiliations
Contributions
H.Y. and B.R. designed the computations, and H.Y. carried out the computations; P.A. and B.R. set the overall aim of the experiments; the electrophysiological experiments were designed by P.A. and carried out by I.R. and P.A.; H.Y., P.A. and B.R. wrote the manuscript.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Supplementary information
Supplementary Text and Figures
Supplementary Figs 1-9, Supplementary Tables 1-3 and Supplementary Methods (PDF 1954 kb)
Rights and permissions
About this article
Cite this article
Yu, H., Ratheal, I., Artigas, P. et al. Protonation of key acidic residues is critical for the K+-selectivity of the Na/K pump. Nat Struct Mol Biol 18, 1159–1163 (2011). https://doi.org/10.1038/nsmb.2113
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/nsmb.2113
- Springer Nature America, Inc.
This article is cited by
-
The CAPOS mutation in ATP1A3 alters Na/K-ATPase function and results in auditory neuropathy which has implications for management
Human Genetics (2018)
-
Glutamate Water Gates in the Ion Binding Pocket of Na+ Bound Na+, K+-ATPase
Scientific Reports (2017)
-
Mechanism of potassium ion uptake by the Na+/K+-ATPase
Nature Communications (2015)