Key Points
-
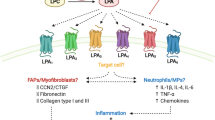
Skeletal muscles produce prostaglandins and leukotrienes that control multiple steps of myogenesis, and are critical for skeletal muscle development, growth and repair
-
Prostaglandins and leukotrienes are powerful mediators of inflammation; many pathological conditions in skeletal muscle are associated with altered prostaglandin and leukotriene production
-
NSAIDs or glucocorticoids that suppress prostaglandin production can have adverse effects on the recovery of the muscle strength of patients with polymyositis or dermatomyositis
-
The cyclooxygenase and arachidonate 5-lipoxygenase pathways, involved in the synthesis of prostaglandins and leukotrienes, are enhanced by inflammatory myopathies and might contribute to muscle atrophy, impairment, and pain
-
Fine-tuning of prostaglandin biosynthesis with selective inhibitors of terminal prostaglandin synthases is a viable alternative approach to suppress inflammation in muscle whilst avoiding detrimental effects on muscle repair
Abstract
Muscle atrophy and weakness are often observed in patients with chronic inflammatory diseases, and are the major clinical features of the autoimmune myopathies, polymyositis and dermatomyositis. A general understanding of the pathogenesis of muscle atrophy and the impaired muscle function associated with chronic inflammatory diseases has not been clarified. In this context, arachidonic acid metabolites, such as the prostaglandin and leukotriene subfamilies, are of interest because they contribute to immune and nonimmune processes. Accumulating evidence suggests that prostaglandins and leukotrienes are involved in causing muscular pain and inflammation, and also in myogenesis and the repair of muscles. In this Review, we summarize novel findings that implicate prostaglandins and leukotrienes in the muscle atrophy and weakness that occur in inflammatory diseases of the muscles, with a focus on inflammatory myopathies. We discuss the role of the arachidonic acid cascade in skeletal muscle growth and function, and individual metabolites as potential therapeutic targets for the treatment of inflammatory muscle diseases.



Similar content being viewed by others
References
Mastaglia, F. L., Garlepp, M. J., Phillips, B. A. & Zilko, P. J. Inflammatory myopathies: clinical, diagnostic and therapeutic aspects. Muscle Nerve 27, 407–425 (2003).
Lundberg, I., Ulfgren, A. K., Nyberg, P., Andersson, U. & Klareskog, L. Cytokine production in muscle tissue of patients with idiopathic inflammatory myopathies. Arthritis Rheum. 40, 865–874 (1997).
Nyberg, P., Wikman, A. L., Nennesmo, I. & Lundberg, I. Increased expression of interleukin 1α and MHC class I in muscle tissue of patients with chronic, inactive polymyositis and dermatomyositis. J. Rheumatol. 27, 940–948 (2000).
Figarella-Branger, D., Civatte, M., Bartoli, C. & Pellissier, J. F. Cytokines, chemokines, and cell adhesion molecules in inflammatory myopathies. Muscle Nerve 28, 659–682 (2003).
Zong, M. & Lundberg, I. E. Pathogenesis, classification and treatment of inflammatory myopathies. Nat. Rev. Rheumatol. 7, 297–306 (2011).
Nagaraju, K. & Lundberg, I. E. Polymyositis and dermatomyositis: pathophysiology. Rheum. Dis. Clin. North. Am. 37, 159–171 (2011).
Prisk, V. & Huard, J. Muscle injuries and repair: the role of prostaglandins and inflammation. Histol. Histopathol. 18, 1243–1256 (2003).
Sun, R., Ba, X., Cui, L., Xue, Y. & Zeng, X. Leukotriene B4 regulates proliferation and differentiation of cultured rat myoblasts via the BLT1 pathway. Mol. Cells 27, 403–408 (2009).
Loell, I. et al. Activated LTB4 pathway in muscle tissue of patients with polymyositis or dermatomyositis. Ann. Rheum. Dis. 72, 293–299 (2013).
Jakobsson, P. J., Thoren, S., Morgenstern, R. & Samuelsson, B. Identification of human prostaglandin E synthase: a microsomal, glutathione-dependent, inducible enzyme, constituting a potential novel drug target. Proc. Natl Acad. Sci. USA 96, 7220–7225 (1999).
Thoren, S. et al. Human microsomal prostaglandin E synthase-1: purification, functional characterization and projection structure determination. J. Biol. Chem. 278, 22199–22209 (2003).
Murakami, M. et al. Regulation of prostaglandin E2 biosynthesis by inducible membrane-associated prostaglandin E2 synthase that acts in concert with cyclooxygenase-2. J. Biol. Chem. 275, 32783–32792 (2000).
Murakami, M. et al. Cellular prostaglandin E2 production by membrane-bound prostaglandin E synthase-2 via both cyclooxygenases-1 and -2. J. Biol. Chem. 278, 37937–37947 (2003).
Tanioka, T., Nakatani, Y., Semmyo, N., Murakami, M. & Kudo, I. Molecular identification of cytosolic prostaglandin E2 synthase that is functionally coupled with cyclooxygenase-1 in immediate prostaglandin E2 biosynthesis. J. Biol. Chem. 275, 32775–32782 (2000).
Berlin, T., Cronestrand, R., Nowak, J., Sonnenfeld, T. & Wennmalm, A. Conversion of arachidonic acid to prostaglandins in homogenates of human skeletal muscle and kidney. Acta Physiol. Scand. 106, 441–445 (1979).
Nowak, J. et al. Biosynthesis of prostaglandins in microsomes of human skeletal muscle and kidney. Prostaglandins Leukot. Med. 11, 269–279 (1983).
Karamouzis, M. et al. In situ microdialysis of intramuscular prostaglandin and thromboxane in contracting skeletal muscle in humans. Acta Physiol. Scand. 171, 71–76 (2001).
Zalin, R. J. The role of hormones and prostanoids in the in vitro proliferation and differentiation of human myoblasts. Exp. Cell Res. 172, 265–281 (1987).
Otis, J. S., Burkholder, T. J. & Pavlath, G. K. Stretch-induced myoblast proliferation is dependent on the COX2 pathway. Exp. Cell Res. 310, 417–425 (2005).
McElligott, M. A., Chaung, L. Y., Baracos, V. & Gulve, E. A. Prostaglandin production in myotube cultures. Influence on protein turnover. Biochem. J. 253, 745–749 (1988).
Bondesen, B. A., Jones, K. A., Glasgow, W. C. & Pavlath, G. K. Inhibition of myoblast migration by prostacyclin is associated with enhanced cell fusion. FASEB J. 21, 3338–3345 (2007).
Velica, P., Khanim, F. L. & Bunce, C. M. Prostaglandin D2 inhibits C2C12 myogenesis. Mol.Cell. Endocrinol. 319, 71–78 (2010).
Testa, M. et al. Expression and activity of cyclooxygenase isoforms in skeletal muscles and myocardium of humans and rodents. J. Appl. Physiol. 103, 1412–1418 (2007).
Weinheimer, E. M. et al. Resistance exercise and cyclooxygenase (COX) expression in human skeletal muscle: implications for COX-inhibiting drugs and protein synthesis. Am. J. Physiol. Regul. Integr. Comp. Physiol. 292, R2241–R2248 (2007).
Studynkova, J. T. et al. The expression of cyclooxygenase-1, cyclooxygenase-2 and 5-lipoxygenase in inflammatory muscle tissue of patients with polymyositis and dermatomyositis. Clin. Exp. Rheumatol. 22, 395–402 (2004).
Buford, T. W. et al. Effects of eccentric treadmill exercise on inflammatory gene expression in human skeletal muscle. Appl. Physiol. Nutr. Metab. 34, 745–753 (2009).
Rabuel, C. et al. Human septic myopathy: induction of cyclooxygenase, heme oxygenase and activation of the ubiquitin proteolytic pathway. Anesthesiology 101, 583–590 (2004).
Sudbo, J. et al. COX-2 expression in striated muscle under physiological conditions. Oral Dis. 9, 313–316 (2003).
Korotkova, M. et al. Effects of immunosuppressive treatment on microsomal prostaglandin E synthase 1 and cyclooxygenases expression in muscle tissue of patients with polymyositis or dermatomyositis. Ann. Rheum. Dis. 67, 1596–1602 (2008).
Jansen, K. M. & Pavlath, G. K. Prostaglandin F2α promotes muscle cell survival and growth through upregulation of the inhibitor of apoptosis protein BRUCE. Cell Death Differ. 15, 1619–1628 (2008).
Horsley, V. & Pavlath, G. K. Prostaglandin F2α stimulates growth of skeletal muscle cells via an NFATC2-dependent pathway. J. Cell. Biol. 161, 111–118 (2003).
Markworth, J. F. & Cameron-Smith, D. Prostaglandin F2α stimulates PI3K/ERK/mTOR signaling and skeletal myotube hypertrophy. Am. J. Physiol. Cell Physiol. 300, C671–C682 (2011).
Shen, W., Prisk, V. R., Li, Y., Foster, W. & Huard, J. Inhibited skeletal muscle healing in cyclooxygenase-2 gene-deficient mice: the role of PGE2 and PGF2α. J. Appl. Physiol. 101, 1215–1221 (2006).
Mo, C., Romero-Suarez, S., Bonewald, L., Johnson, M. & Brotto, M. Prostaglandin E2: from clinical applications to its potential role in bone- muscle crosstalk and myogenic differentiation. Recent Pat. Biotechnol. 6, 223–229 (2012).
Hunter, J. G., van Delft, M. F., Rachubinski, R. A. & Capone, J. P. Peroxisome proliferator-activated receptor gamma ligands differentially modulate muscle cell differentiation and MyoD gene expression via peroxisome proliferator-activated receptor gamma -dependent and -independent pathways. J. Biol. Chem. 276, 38297–38306 (2001).
Markworth, J. F. & Cameron-Smith, D. Arachidonic acid supplementation enhances in vitro skeletal muscle cell growth via a COX-2-dependent pathway. Am. J. Physiol. Cell Physiol. 304, C56–C67 (2013).
Mendias, C. L., Tatsumi, R. & Allen, R. E. Role of cyclooxygenase-1 and -2 in satellite cell proliferation, differentiation, and fusion. Muscle Nerve 30, 497–500 (2004).
Bondesen, B. A., Mills, S. T., Kegley, K. M. & Pavlath, G. K. The COX-2 pathway is essential during early stages of skeletal muscle regeneration. Am. J. Physiol. Cell Physiol. 287, C475–C483 (2004).
Bondesen, B. A., Mills, S. T. & Pavlath, G. K. The COX-2 pathway regulates growth of atrophied muscle via multiple mechanisms. Am. J. Physiol. Cell Physiol. 290, 1651–1659 (2006).
Trappe, T. A., Fluckey, J. D., White, F., Lambert, C. P. & Evans, W. J. Skeletal muscle PGF2α and PGE2 in response to eccentric resistance exercise: influence of ibuprofen acetaminophen. J. Clin. Endocrinol. Metab. 86, 5067–5070 (2001).
Mikkelsen, U. R. et al. Local NSAID infusion does not affect protein synthesis and gene expression in human muscle after eccentric exercise. Scand. J. Med. Sci. Sports 21, 630–644 (2011).
Reeds, P. J. & Palmer, R. M. Changes in prostaglandin release associated with inhibition of muscle protein synthesis by dexamethasone. Biochem. J. 219, 953–957 (1984).
Southorn, B. G. & Palmer, R. M. Inhibitors of phospholipase A2 block the stimulation of protein synthesis by insulin in L6 myoblasts. Biochem. J. 270, 737–739 (1990).
Chromiak, J. A. & Vandenburgh, H. H. Mechanical stimulation of skeletal muscle cells mitigates glucocorticoid-induced decreases in prostaglandin production and prostaglandin synthase activity. J. Cell. Physiol. 159, 407–414 (1994).
Kreiner, F. & Galbo, H. Elevated muscle interstitial levels of pain-inducing substances in symptomatic muscles in patients with polymyalgia rheumatica. Pain 152, 1127–1132 (2011).
Symons, J. D., Theodossy, S. J., Longhurst, J. C. & Stebbins, C. L. Intramuscular accumulation of prostaglandins during static contraction of the cat triceps surae. J. Appl. Physiol. 71, 1837–1842 (1991).
Wilson, J. R. & Kapoor, S. C. Contribution of prostaglandins to exercise-induced vasodilation in humans. Am. J. Physiol. 265, H171–H175 (1993).
Young, M. K., Bocek, R. M., Herrington, P. T. & Beatty, C. H. Aging: effects on the prostaglandin production by skeletal muscle of male rhesus monkeys (Macaca mulatta). Mech. Ageing Dev. 16, 345–353 (1981).
Kozlovsky, N., Shohami, E. & Bashan, N. Increased PLA2 activity is not related to increase GLUT1 expression in L6 myotubes under hypoxic conditions. Prostaglandins Leukot. Essent. Fatty Acids 56, 17–22 (1997).
McArdle, A., Edwards, R. H. & Jackson, M. J. Release of creatine kinase and prostaglandin E2 from regenerating skeletal muscle fibers. J. Appl. Physiol. 76, 1274–1278 (1994).
Briolay, A., Jaafar, R., Nemoz, G. & Bessueille, L. Myogenic differentiation and lipid-raft composition of L6 skeletal muscle cells are modulated by PUFAs. Biochim. Biophys. Acta 1828, 602–613 (2012).
Hurley, M. S., Flux, C., Salter, A. M. & Brameld, J. M. Effects of fatty acids on skeletal muscle cell differentiation in vitro. Br. J. Nutr. 95, 623–630 (2006).
Lee, J. H., Tachibana, H., Morinaga, Y., Fujimura, Y. & Yamada, K. Modulation of proliferation and differentiation of C2C12 skeletal muscle cells by fatty acids. Life Sci. 84, 415–420 (2009).
Kadotani, A., Tsuchiya, Y., Hatakeyama, H., Katagiri, H. & Kanzaki, M. Different impacts of saturated and unsaturated free fatty acids on COX-2 expression in C2C12 myotubes. Am. J. Physiol. Endocrinol. Metab. 297, E1291–E1303 (2009).
McArdle, A., Foxley, A., Edwards, R. H. & Jackson, M. J. Prostaglandin metabolism in dystrophin-deficient MDX mouse muscle. Biochem. Soc. Trans. 19, 177S (1991).
Jackson, M. J., Brooke, M. H., Kaiser, K. & Edwards, R. H. Creatine kinase and prostaglandin E2 release from isolated Duchenne muscle. Neurology 41, 101–104 (1991).
Lindahl, M., Backman, E., Henriksson, K. G., Gorospe, J. R. & Hoffman, E. P. Phospholipase A2 activity in dystrophinopathies. Neuromuscul. Disord. 5, 193–199 (1995).
Okinaga, T. et al. Induction of hematopoietic prostaglandin D synthase in hyalinated necrotic muscle fibers: its implication in grouped necrosis. Acta Neuropathol. 104, 377–384 (2002).
Mohri, I. et al. Inhibition of prostaglandin D synthase suppresses muscular necrosis. Am. J. Pathol. 174, 1735–1744 (2009).
Turinsky, J., O'Sullivan, D. M. & Bayly, B. P. Modulation of prostaglandin E2 synthesis in rat skeletal muscle. Am. J. Physiol. 262, E476–482 (1992).
Jaweed, M. M., Alam, I., Herbison, G. J. & Ditunno, J. F. Jr. Prostaglandins in denervated skeletal muscle of the rat: effect of direct electrical stimulation. Neuroscience 6, 2787–2792 (1981).
Turinsky, J. Phospholipids, prostaglandin E2, and proteolysis in denervated muscle. Am. J. Physiol. 251, R165–R173 (1986).
Murray, M. A. & Robbins, N. Cell proliferation in denervated muscle: identity and origin of dividing cells. Neuroscience 7, 1823–1833 (1982).
Tegeder, L., Zimmermann, J., Meller, S. T. & Geisslinger, G. Release of algesic substances in human experimental muscle pain. Inflamm. Res. 51, 393–402 (2002).
Hedenberg-Agnusson, B., Ernberg, M., Alstergren, P. & Kopp, S. Pain mediation by prostaglandin E2 and leukotriene B4 in the human masseter muscle. Acta Odontol. Scand. 59, 348–355 (2001).
Rodemann, H. P. & Goldberg, A. L. Arachidonic acid, prostaglandin E2 and F2 alpha influence rates of protein turnover in skeletal and cardiac muscle. J. Biol. Chem. 257, 1632–1638 (1982).
Rodemann, H. P., Waxman, L. & Goldberg, A. L. The stimulation of protein degradation in muscle by Ca2+ is mediated by prostaglandin E2 and does not require the calcium-activated protease. J. Biol. Chem. 257, 8716–8723 (1982).
Barnett, J. G. & Ellis, S. Prostaglandin E2 and the regulation of protein degradation in skeletal muscle. Muscle Nerve 10, 556–559 (1987).
Hasselgren, P. O., Zamir, O., James, J. H. & Fischer, J. E. Prostaglandin E2 does not regulate total or myofibrillar protein breakdown in incubated skeletal muscle from normal or septic rats. Biochem. J. 270, 45–50 (1990).
Rieu, I. et al. Reduction of low grade inflammation restores blunting of postprandial muscle anabolism and limits sarcopenia in old rats. J. Physiol. 587, 5483–5492 (2009).
Granado, M., Martin, A. I., Villanua, M. A. & Lopez-Calderon, A. Experimental arthritis inhibits the insulin-like growth factor-I axis and induces muscle wasting through cyclooxygenase-2 activation. Am. J. Physiol. Endocrinol. Metab. 292, E1656–E1665 (2007).
Strelkov, A. B., Fields, A. L. & Baracos, V. E. Effects of systemic inhibition of prostaglandin production on protein metabolism in tumor-bearing rats. Am. J. Physiol. 257, C261–C269 (1989).
McCarthy, D. O., Whitney, P., Hitt, A. & Al-Majid, S. Indomethacin and ibuprofen preserve gastrocnemius muscle mass in mice bearing the colon-26 adenocarcinoma. Res. Nurs. Health 27, 174–184 (2004).
Trappe, T. A., Standley, R. A., Jemiolo, B., Carroll, C. C. & Trappe, S. W. Prostaglandin and myokine involvement in the cyclooxygenase-inhibiting drug enhancement of skeletal muscle adaptations to resistance exercise in older adults. Am. J. Physiol. Regul. Integr. Comp. Physiol. 304, R198–R205 (2013).
Standley, R. A., Liu, S. Z., Jemiolo, B., Trappe, S. W. & Trappe, T. A. Prostaglandin E2 induces transcription of skeletal muscle mass regulators interleukin-6 and muscle RING finger-1 in humans. Prostaglandins Leukot. Essent. Fatty Acids 88, 361–364 (2013).
Baracos, V., Rodemann, H. P., Dinarello, C. A. & Goldberg, A. L. Stimulation of muscle protein degradation and prostaglandin E2 release by leukocytic pyrogen (interleukin-1). A mechanism for the increased degradation of muscle proteins during fever. N. Engl. J. Med. 308, 553–558 (1983).
Schafers, M., Sorkin, L. S. & Sommer, C. Intramuscular injection of tumor necrosis factor-alpha induces muscle hyperalgesia in rats. Pain 104, 579–588 (2003).
Schneider, A. et al. Membrane-associated PGE synthase-1 (mPGES-1) is coexpressed with both COX-1 and COX-2 in the kidney. Kidney Int. 65, 1205–1213 (2004).
Boulet, L. et al. Deletion of microsomal prostaglandin E2 (PGE2) synthase-1 reduces inducible and basal PGE2 production and alters the gastric prostanoid profile. J. Biol. Chem. 279, 23229–23237 (2004).
Lundberg, I., Kratz, A. K., Alexanderson, H. & Patarroyo, M. Decreased expression of interleukin-1alpha, interleukin-1beta, and cell adhesion molecules in muscle tissue following corticosteroid treatment in patients with polymyositis and dermatomyositis. Arthritis Rheum. 43, 336–348 (2000).
Helliwell, P. S. & Jackson, S. Relationship between weakness and muscle wasting in rheumatoid arthritis. Ann. Rheum. Dis 53, 726–728 (1994).
Stenstrom, C. H. & Minor, M. A. Evidence for the benefit of aerobic and strengthening exercise in rheumatoid arthritis. Arthritis Rheum. 49, 428–434 (2003).
Donaldson, A. V., Maddocks, M., Martolini, D., Polkey, M. I. & Man, W. D. Muscle function in COPD: a complex interplay. Int. J. Chron. Obstruct. Pulmon. Dis. 7, 523–535 (2012).
Turesson, C. et al. Increased endothelial expression of HLA-DQ and interleukin 1α in extra-articular rheumatoid arthritis. Results from immunohistochemical studies of skeletal muscle. Rheumatology 40, 1346–1354 (2001).
Bhattacharya, A. et al. Genetic ablation of 12/15-lipoxygenase but not 5-lipoxygenase protects against denervation-induced muscle atrophy. Free Radic. Biol. Med. 67, 30–40 (2013).
Young, J. F., Hansen-Moller, J. & Oksbjerg, N. Effect of flavonoids on stress responses in myotube cultures. J. Agric. Food Chem. 52, 7158–7163 (2004).
Tager, A. M. et al. Leukotriene B4 receptor BLT1 mediates early effector T cell recruitment. Nat. Immunol. 4, 982–990 (2003).
Liu, A., Claesson, H. E., Mahshid, Y., Klein, G. & Klein, E. Leukotriene B4 activates T cells that inhibit B-cell proliferation in EBV-infected cord blood-derived mononuclear cell cultures. Blood 111, 2693–2703 (2008).
Chen, H. et al. Effects of leukotriene B4 and prostaglandin E2 on the differentiation of murine Foxp3+ T regulatory cells and Th17 cells. Prostaglandins Leukot. Essent. Fatty Acids 80, 195–200 (2009).
Ricciotti, E. & FitzGerald, G. A. Prostaglandins and inflammation. Arteriosc. Throm. Vascul. Biol. 31, 986–1000 (2011).
Nakamura, M. & Shimizu, T. Leukotriene receptors. Chem. Rev. 111, 6231–6298 (2011).
Acknowledgements
Research by the authors is supported by The Swedish Research Council, The Swedish Rheumatism Association, Karolinska Institutet Foundation, the King Gustaf V 80 year foundation and “The regional agreement on medical training and clinical research (ALF) between Stockholm County Council and Karolinska Institutet”.
Author information
Authors and Affiliations
Contributions
I. E. Lundberg contributed substantially to the discussion of content and writing, reviewing and editing the manuscript. M. Korotkova researched data for the article, and contributed substantially to discussion of content, and writing, reviewing and editing the manuscript.
Corresponding author
Ethics declarations
Competing interests
I. E. Lundberg is a stockholder in Pfizer. M. Korotkova declares no competing interests.
Rights and permissions
About this article
Cite this article
Korotkova, M., Lundberg, I. The skeletal muscle arachidonic acid cascade in health and inflammatory disease. Nat Rev Rheumatol 10, 295–303 (2014). https://doi.org/10.1038/nrrheum.2014.2
Published:
Issue Date:
DOI: https://doi.org/10.1038/nrrheum.2014.2
- Springer Nature Limited
This article is cited by
-
Quercetin inhibits the metabolism of arachidonic acid by inhibiting the activity of CYP3A4, thereby inhibiting the progression of breast cancer
Molecular Medicine (2023)
-
Metabolic profiling of patients with different idiopathic inflammatory myopathy subtypes reveals potential biomarkers in plasma
Clinical and Experimental Medicine (2023)
-
Lipocalin-Type Prostaglandin D2 Synthase Protein- A Central Player in Metabolism
Pharmaceutical Research (2022)
-
Depletion of essential fatty acids in muscle is associated with shorter survival of cancer patients undergoing surgery-preliminary report
Scientific Reports (2021)
-
Targeted lipidomics analysis identified altered serum lipid profiles in patients with polymyositis and dermatomyositis
Arthritis Research & Therapy (2018)