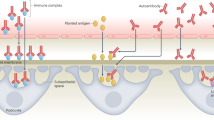
Abstract
The spontaneously occurring autoantibodies that are associated with human diseases bear the hallmarks of a typical immune response. The repertoire of autoantibodies is surprisingly limited, however, and is the same in both humans and mice. Neither molecular mimicry nor immune dysregulation accounts for this unexpectedly narrow focus of specificities. Experimental data on the properties of the target autoantigens — such as their structure, catabolism, exposure to the immune system after cell death and recently described immunostimulatory effects on immature dendritic cells — indicate that these properties, in conjunction with the tissue microenvironment, help to select the autoantibody repertoire.

Similar content being viewed by others
References
Shlomchik, M. et al. Anti-DNA antibodies from autoimmune mice arise by clonal expansion and somatic mutation. J. Exp. Med. 171, 265–292 (1990).
Miller, F. W., Waite, K. A., Biswas, T. & Plotz, P. H. The role of an autoantigen, histidyl-tRNA synthetase, in the induction and maintenance of autoimmunity. Proc. Natl Acad. Sci. USA 87, 9933–9937 (1990).
Lernmark, A. Autoimmune diseases: are markers ready for prediction? J. Clin. Invest. 108, 1091–1096 (2001).
Satoh, M. et al. Widespread susceptibility among inbred mouse strains to the induction of lupus autoantibodies by pristine. Clin. Exp. Immunol. 121, 399–405 (2000).
Gelpi, C., Martinez, M. A., Vidal, S., Targoff, I. N. & Rodriguez-Sanchez, J. L. Autoantibodies to a transfer RNA-associated protein in a murine model of chronic graft-versus-host disease. J. Immunol. 152, 1989–1999 (1994).
Mak, T. W., Penninger, J. M. & Ohashi, P. S. Knockout mice: a paradigm shift in modern immunology. Nature Rev. Immunol. 1, 11–19 (2001).
Nagaraju, K. et al. Conditional up-regulation of MHC class I in skeletal muscle leads to self-sustaining autoimmune myositis and myositis-specific autoantibodies. Proc. Natl Acad. Sci. USA 97, 9209–9214 (2000).
Winter, S. F. et al. Development of antibodies against p53 in lung cancer patients appears to be dependent on the type of p53 mutation. Cancer Res. 52, 4168–4174 (1992).
Levin, M. C. et al. Autoimmunity due to molecular mimicry as a cause of neurological disease. Nature Med. 8, 509–513 (2002).
Naparstek, Y. & Plotz, P. H. The role of autoantibodies in autoimmune disease. Annu. Rev. Immunol. 11, 79–104 (1993).
Hibbs, M. L. et al. Multiple defects in the immune system of Lyn-deficient mice, culminating in autoimmune disease. Cell 83, 301–311 (1995).
Bachmaier, K. et al. Negative regulation of lymphocyte activation and autoimmunity by the molecular adaptor Cbl-b. Nature 403, 211–216 (2000).
Mackay, F. et al. Mice transgenic for BAFF develop lymphocytic disorders along with autoimmune manifestations. J. Exp. Med. 190, 1697–1710 (1999).
Lipsky, P. E. Systemic lupus erythematosus: an autoimmune disease of B-cell hyperactivity. Nature Immunol. 2, 764–766 (2001).
Wucherpfennig, K. W. Mechanisms for the induction of autoimmunity by infectious agents. J. Clin. Invest. 108, 1097–1104 (2001).
Wucherpfennig, K. W. Structural basis of molecular mimicry. J. Autoimmun. 16, 293–302 (2001).
Maverakis, E., van den Elzen, P. & Sercarz, E. E. Self-reactive T cells and degeneracy of T-cell recognition: evolving concepts — from sequence homology to shape mimicry and TCR flexibility. J. Autoimmun. 16, 201–209 (2001).
Maldonado, M. A. et al. The role of environmental antigens in the spontaneous development of autoimmunity in MRL-lpr mice. J. Immunol. 162, 6322–6330 (1999).
Wanstrat, A. & Wakeland, E. The genetics of complex autoimmune diseases: non-MHC susceptibility genes. Nature Immunol. 2, 802–809 (2001).
Janeway, C. A. Jr. How the immune system works to protect the host from infection: a personal view. Proc. Natl Acad. Sci. USA 98, 7461–7468 (2001).
Matzinger, P. The danger model: a renewed sense of self. Science 296, 301–305 (2002).
Gallucci, S., Lolkema, M. & Matzinger, P. Natural adjuvants: endogenous activators of dendritic cells. Nature Med. 5, 1249–1255 (1999).
Aliberti, J. & Sher, A. Positive and negative regulation of pathogen-induced dendritic-cell function by G-protein-coupled receptors. Mol. Immunol. 38, 891–893 (2002).
Gill, T. J. III, Kunz, H. W. & Papermaster, D. S. Studies on synthetic polypeptide antigens. 18. The role of composition, charge and optical isomerism in the immunogenicity of synthetic polypeptides. J. Biol. Chem. 242, 3308–3318 (1967).
Brendel, V., Dohlman, J., Blaisdell, B. E. & Karlin, S. Very long charge runs in systemic lupus erythematosus-associated autoantigens. Proc. Natl Acad. Sci. USA 88, 1536–1540 (1991).
Dohlman, J. G., Lupas, A. & Carson, M. Long charge-rich α-helices in systemic autoantigens. Biochem. Biophys. Res. Commun. 195, 686–696 (1993).
Casciola-Rosen, L. A., Anhalt, G. & Rosen, A. Autoantigens targeted in systemic lupus erythematosus are clustered in two populations of surface structures on apoptotic keratinocytes. J. Exp. Med. 179, 1317–1330 (1994).
Rosen, A., Casciola-Rosen, L. & Ahearn, J. Novel packages of viral and self-antigens are generated during apoptosis. J. Exp. Med. 181, 1557–1561 (1995).
Napirei, M. et al. Features of systemic lupus erythematosus in DNase1-deficient mice. Nature Genet. 25, 177–181 (2000).
Taylor, P. R. et al. A hierarchical role for classical-pathway complement proteins in the clearance of apoptotic cells in vivo. J. Exp. Med. 192, 359–366 (2000).
Scott, R. S. et al. Phagocytosis and clearance of apoptotic cells is mediated by MER. Nature 411, 207–211 (2001).
Casciola-Rosen, L., Andrade, F., Ulanet, D., Wong, W. B. & Rosen, A. Cleavage by granzyme B is strongly predictive of autoantigen status: implications for initiation of autoimmunity. J. Exp. Med. 190, 815–826 (1999).
Rutjes, S. A. et al. The La (SS-B) autoantigen, a key protein in RNA biogenesis, is dephosphorylated and cleaved early during apoptosis. Cell Death. Differ. 6, 976–986 (1999).
Degen, W. G., Aarssen, Y., Pruijn, G. J., Utz, P. J. & van Venrooij, W. J. The fate of U1 snRNP during anti-Fas-induced apoptosis: specific cleavage of the U1 snRNA molecule. Cell Death. Differ. 7, 70–79 (2000).
Utz, P. J. et al. The 72-kDa component of signal-recognition particle is cleaved during apoptosis. J. Biol. Chem. 273, 35362–35370 (1998).
Utz, P. J., Gensler, T. J. & Anderson, P. Death, autoantigen modifications and tolerance. Arthritis Res. 2, 101–114 (2000).
Huynh, M. L., Fadok, V. A. & Henson, P. M. Phosphatidylserine-dependent ingestion of apoptotic cells promotes TGF-β1 secretion and the resolution of inflammation. J. Clin. Invest. 109, 41–50 (2002).
Sauter, B. et al. Consequences of cell death: exposure to necrotic tumor cells, but not primary tissue cells or apoptotic cells, induces the maturation of immunostimulatory dendritic cells. J. Exp. Med. 191, 423–434 (2000).
Leadbetter, E. A. et al. Chromatin–IgG complexes activate B cells by dual engagement of IgM and Toll-like receptors. Nature 416, 603–607 (2002).
Martinis, S. A., Plateau, P., Cavarelli, J. & Florentz, C. Aminoacyl-tRNA synthetases: a new image for a classical family. Biochimie 81, 683–700 (1999).
Wakasugi, K. & Schimmel, P. Two distinct cytokines released from a human aminoacyl-tRNA synthetase. Science 284, 147–151 (1999).
Mathews, M. B. & Bernstein, R. M. Myositis autoantibody inhibits histidyl-tRNA synthetase: a model for autoimmunity. Nature 304, 177–179 (1983).
Howard, O. M. et al. Histidyl-tRNA synthetase and asparaginyl-tRNA synthetase, autoantigens in myositis, activate chemokine receptors on T lymphocytes and immature dendritic cells. J. Exp. Med. 196, 781–791 (2002).
Raben, N. et al. A motif in human histidyl-tRNA synthetase which is shared among several aminoacyl-tRNA synthetases is a coiled-coil that is essential for enzymatic activity and contains the major autoantigenic epitope. J. Biol. Chem. 269, 24277–24283 (1994).
Turville, S. G. et al. Diversity of receptors binding HIV on dendritic-cell subsets. Nature Immunol. 3, 975–983 (2002).
Litinskiy, M. B. et al. DCs induce CD40-independent immunoglobulin class switching through BLyS and APRIL. Nature Immunol. 3, 822–829 (2002).
MacLennan, I. C. M. et al. Dendritic cells, BAFF and APRIL: innate players in adaptive responses. Immunity 17, 235–238 (2002).
Rolink, A. G. et al. BAFFled B cells survive and thrive: roles of BAFF in B-cell development. Curr. Opin. Immunol. 14, 266–275 (2002).
Acknowledgements
I have had fruitful discussions with several colleagues on this subject, particularly L. Casciola-Rosen, P. Cohen, B. Eisenberg, R. Germain, Z. Howard, P. Lipsky, K. Nagaraju, J. Oppenheim, J. O'Shea, Judith Plotz, John Plotz, N. Raben, A. Rosen and R. Siegel.
Author information
Authors and Affiliations
Related links
Rights and permissions
About this article
Cite this article
Plotz, P. The autoantibody repertoire: searching for order. Nat Rev Immunol 3, 73–78 (2003). https://doi.org/10.1038/nri976
Issue Date:
DOI: https://doi.org/10.1038/nri976
- Springer Nature Limited
This article is cited by
-
GPCR-specific autoantibody signatures are associated with physiological and pathological immune homeostasis
Nature Communications (2018)
-
Analyzing pathogenic (double-stranded (ds) DNA-specific) plasma cells via immunofluorescence microscopy
Arthritis Research & Therapy (2015)
-
Perception of self: distinguishing autoimmunity from autoinflammation
Nature Reviews Rheumatology (2015)
-
Surface expression and genotypes of Toll-like receptors 2 and 4 in patients with juvenile idiopathic arthritis and systemic lupus erythematosus
Pediatric Rheumatology (2013)
-
Cartilage oligomeric matrix protein specific antibodies are pathogenic
Arthritis Research & Therapy (2012)