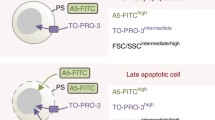
Abstract
Since its introduction, the propidium iodide (PI) flow cytometric assay has been widely used for the evaluation of apoptosis in different experimental models. It is based on the principle that apoptotic cells, among other typical features, are characterized by DNA fragmentation and, consequently, loss of nuclear DNA content. Use of a fluorochrome, such as PI, that is capable of binding and labeling DNA makes it possible to obtain a rapid (the protocol can be completed in about 2 h) and precise evaluation of cellular DNA content by flow cytometric analysis, and subsequent identification of hypodiploid cells. The original protocol enhanced the capacity for a rapid, quantitative measure of cell apoptosis. For this reason, since its publication, the PI assay has been widely used, as demonstrated by the large number of citations of the original paper and/or the continuous use of the method in many laboratories.


Similar content being viewed by others
References
Wyllie, A.H. Apoptosis: an overview. Br. Med. Bull. 53, 451–465 (1997).
Arends, M.J., Morris, R.G. & Wyllie, A.H. Apoptosis. The role of the endonuclease. Am. J. Pathol. 136, 593–608 (1990).
Sellins, K.S. & Cohen, J.J. Gene induction by gamma-irradiation leads to DNA fragmentation in lymphocytes. J. Immunol. 139, 3199–3206 (1987).
Gong, J., Traganos, F. & Darzynkiewicz, Z. A selective procedure for DNA extraction from apoptotic cells applicable for gel electrophoresis and flow cytometry. Anal. Biochem. 218, 314–319 (1994).
Nunez, R. DNA measurement and cell cycle analysis by flow cytometry. Curr. Issues Mol. Biol. 3, 67–70 (2001).
Nicoletti, I., Migliorati, G., Pagliacci, M.C., Grignani, F. & Riccardi, C. A rapid and simple method for measuring thymocyte apoptosis by propidium iodide staining and flow cytometry. J. Immunol. Methods 139, 271–279 (1991).
Ormerod, M.G. Investigating the relationship between the cell cycle and apoptosis using flow cytometry. J. Immunol. Methods 265, 73–80 (2002).
Darzynkiewicz, Z., Bedner, E. & Smolewski, P. Flow cytometry in analysis of cell cycle and apoptosis. Semin. Hematol. 38, 179–193 (2001).
Telford, W.G., King, L.E. & Fraker, P.J. Rapid quantitation of apoptosis in pure and heterogeneous cell populations using flow cytometry. J. Immunol. Methods 172, 1–16 (1994).
Wallen, C.A., Higashikubo, R. & Dethlefsen, L.A. Comparison of two flow cytometric assays for cellular RNA—acridine orange and propidium iodide. Cytometry 3, 155–160 (1982).
Pollack, A. & Ciancio, G. Cell cycle phase-specific analysis of cell viability using Hoechst 33342 and propidium iodide after ethanol preservation. Methods Cell. Biol. 33, 19–24 (1990).
Ormerod, M.G., Collins, M.K., Rodriguez-Tarduchy, G. & Robertson, D. Apoptosis in interleukin-3-dependent haemopoietic cells. Quantification by two flow cytometric methods. J. Immunol. Methods 153, 57–65 (1992).
Schmid, I., Uittenbogaart, C.H., Keld, B. & Giorgi, J.V. A rapid method for measuring apoptosis and dual-color immunofluorescence by single laser flow cytometry. J. Immunol. Methods 170, 145–157 (1994).
Wyllie, A.H., Morris, R.G., Smith, A.L. & Dunlop, D. Chromatin cleavage in apoptosis: association with condensed chromatin morphology and dependence on macromolecular synthesis. J. Pathol. 142, 67–77 (1984).
Gavrieli, Y., Sherman, Y. & Ben-Sasson, S.A. Identification of programmed cell death in situ via specific labeling of nuclear DNA fragmentation. J. Cell Biol. 119, 493–501 (1992).
Telford, W.G., Komoriya, A. & Packard, B.Z. Detection of localized caspase activity in early apoptotic cells by laser scanning cytometry. Cytometry 47, 81–88 (2002).
Acknowledgements
This work was supported by Associazione Italiana Ricerca sul Cancro (AIRC, to CR), Milan, Italy.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Rights and permissions
About this article
Cite this article
Riccardi, C., Nicoletti, I. Analysis of apoptosis by propidium iodide staining and flow cytometry. Nat Protoc 1, 1458–1461 (2006). https://doi.org/10.1038/nprot.2006.238
Published:
Issue Date:
DOI: https://doi.org/10.1038/nprot.2006.238
- Springer Nature Limited
This article is cited by
-
Anti-CD38 targeted nanotrojan horses stimulated by acoustic waves as therapeutic nanotools selectively against Burkitt’s lymphoma cells
Discover Nano (2024)
-
Antitumor effects of co-treatment of resveratrol with antitumor drugs in ER- and HER2-positive breast cancer cells are due to induction of apoptosis and modulation of estrogen receptor expression
Breast Cancer (2024)
-
Melatonin modulates L-arginine metabolism in tumor-associated macrophages by targeting arginase 1 in lymphoma
Naunyn-Schmiedeberg's Archives of Pharmacology (2024)
-
Sodium orthovanadate exhibits anti-angiogenic, antiapoptotic and blood glucose-lowering effect on colon cancer associated with diabetes
Cancer Chemotherapy and Pharmacology (2024)
-
Ethanolic extract from fruiting bodies of Cordyceps militaris HL8 exhibits cytotoxic activities against cancer cells, skin pathogenic yeasts, and postharvest pathogen Penicillium digitatum
Archives of Microbiology (2024)