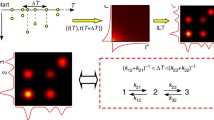
Abstract
Proteins are dynamic molecular machines having structural flexibility that allows conformational changes1,2. Current methods for the determination of protein flexibility rely mainly on the measurement of thermal fluctuations and disorder in protein conformations3,4,5 and tend to be experimentally challenging. Moreover, they reflect atomic fluctuations on picosecond timescales, whereas the large conformational changes in proteins typically happen on micro- to millisecond timescales6,7. Here, we directly determine the flexibility of bacteriorhodopsin—a protein that uses the energy in light to move protons across cell membranes—at the microsecond timescale by monitoring force-induced deformations across the protein structure with a technique based on atomic force microscopy. In contrast to existing methods, the deformations we measure involve a collective response of protein residues and operate under physiologically relevant conditions with native proteins.



Similar content being viewed by others
References
Falke, J. J. & Koshland, D. E. Global flexibility in a sensory receptor—a site-directed cross-linking approach. Science 237, 1596–1600 (1987).
Frauenfelder, H., Sligar, S. G. & Wolynes, P. G. The energy landscapes and motions of proteins. Science 254, 1598–1603 (1991).
Frauenfelder, H., Petsko, G. A. & Tsernoglou, D. Temperature-dependent X-ray-diffraction as a probe of protein structural dynamics. Nature 280, 558–563 (1979).
Mittermaier, A. & Kay, L. E. New tools provide new insights in NMR studies of protein dynamics. Science 312, 224–228 (2006).
Zaccai, G. How soft is a protein? A protein dynamics force constant measured by neutron scattering. Science 288, 1604–1607 (2000).
Subramaniam, S., Gerstein, M., Oesterhelt, D. & Henderson, R. Electron-diffraction analysis of structural changes in the photocycle of bacteriorhodopsin. EMBO J. 12, 1–8 (1993).
Rousso, I. et al. Microsecond atomic force sensing of protein conformational dynamics: implications for the primary light-induced events in bacteriorhodopsin. Proc. Natl Acad. Sci. USA 94, 7937–7941 (1997).
Dodson, G. & Verma, C. S. Protein flexibility: its role in structure and mechanism revealed by molecular simulations. Cell. Mol. Life Sci. 63, 207–219 (2006).
Clausen-Schaumann, H., Seitz, M., Krautbauer, R. & Gaub, H. E. Force spectroscopy with single bio-molecules. Curr. Opin. Chem. Biol. 4, 524–530 (2000).
Sulchek, T. A. et al. Dynamic force spectroscopy of parallel individual Mucin1-antibody bonds. Proc. Natl Acad. Sci. USA 102, 16638–16643 (2005).
Dietz, H., Berkemeier, F., Bertz, M. & Rief, M. Anisotropic deformation response of single protein molecules. Proc. Natl Acad. Sci. USA 103, 12724–12728 (2006).
Knowles, T. P. et al. Role of intermolecular forces in defining material properties of protein nanofibrils. Science 318, 1900–1903 (2007).
Sahin, O., Magonov, S., Su, C., Quate, C. F. & Solgaard, O. An atomic force microscope tip designed to measure time-varying nanomechanical forces. Nature Nanotech. 2, 507–514 (2007).
Martinez, N. F. et al. Bimodal atomic force microscopy imaging of isolated antibodies in air and liquids. Nanotechnology 19, 384011 (2008).
Preiner, J., Tang, J. L., Pastushenko, V. & Hinterdorfer, P. Higher harmonic atomic force microscopy: Imaging of biological membranes in liquid. Phys. Rev. Lett. 99, 046102 (2007).
Legleiter, J., Park, M., Cusick, B. & Kowalewski, T. Scanning probe acceleration microscopy (SPAM) in fluids: Mapping mechanical properties of surfaces at the nanoscale. Proc. Natl Acad. Sci. USA 103, 4813–4818 (2006).
Haupts, U., Tittor, J. & Oesterhelt, D. Closing in on bacteriorhodopsin: progress in understanding the molecule. Annu. Rev. Biophys. Biomol. Struct. 28, 367–399 (1999).
Luecke, H., Schobert, B., Richter, H. T., Cartailler, J. P. & Lanyi, J. K. Structure of bacteriorhodopsin at 1.55 angstrom resolution. J. Mol. Biol. 291, 899–911 (1999).
Muller, D. J. & Engel, A. The height of biomolecules measured with the atomic force microscope depends on electrostatic interactions. Biophys. J. 73, 1633–1644 (1997).
Zhong, S. et al. Different interactions between the two sides of purple membrane with atomic force microscope tip. Langmuir 23, 4486–4493 (2007).
Sahin, O. Harnessing bifurcations in tapping-mode atomic force microscopy to calibrate time-varying tip–sample force measurements. Rev. Sci. Instrum. 78, 103707 (2007).
Parlak, Z. & Degertekin, F. L. Contact stiffness of finite size subsurface defects for atomic force microscopy: three-dimensional finite element modeling and experimental verification. J. Appl. Phys. 103, 114910 (2008).
Stark, M., Moller, C., Muller, D. J. & Guckenberger, R. From images to interactions: high-resolution phase imaging in tapping-mode atomic force microscopy. Biophys. J. 80, 3009–3018 (2001).
Alexiev, U., Marti, T., Heyn, M. P., Khorana, H. G. & Scherrer, P. Surface-charge of bacteriorhodopsin detected with covalently bound pH indicators at selected extracellular and cytoplasmic sites. Biochemistry 33, 298–306 (1994).
Engel, A. & Muller, D. J. Observing single biomolecules at work with the atomic force microscope. Nature Struct. Biol. 7, 715–718 (2000).
Muller, D. J., Fotiadis, D. & Engel, A. Mapping flexible protein domains at subnanometer resolution with the atomic force microscope. FEBS Lett. 430, 105–111 (1998).
Muller, D. J. & Engel, A. Atomic force microscopy and spectroscopy of native membrane proteins. Nature Protoc. 2, 2191–2197 (2007).
Patrick, D. L., Cee, V. J. & Beebe, T. P. Molecule corrals for studies of monolayer organic films. Science 265, 231–234 (1994).
Israelachvili, J. Intermolecular and Surface Forces (Academic Press, 2003).
Johnson, K. L. Contact Mechanics (Cambridge Univ. Press, 2003).
Acknowledgements
This work was supported by the Rowland Junior Fellows program.
Author information
Authors and Affiliations
Corresponding author
Supplementary information
Supplementary information
Supplementary information (PDF 1110 kb)
Rights and permissions
About this article
Cite this article
Dong, M., Husale, S. & Sahin, O. Determination of protein structural flexibility by microsecond force spectroscopy. Nature Nanotech 4, 514–517 (2009). https://doi.org/10.1038/nnano.2009.156
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/nnano.2009.156
- Springer Nature Limited
This article is cited by
-
Force spectroscopy of single cells using atomic force microscopy
Nature Reviews Methods Primers (2021)
-
Conventional Molecular and Novel Structural Mechanistic Insights into Orderly Organelle Interactions
Chemical Research in Chinese Universities (2021)
-
Assembly of a patchy protein into variable 2D lattices via tunable multiscale interactions
Nature Communications (2020)
-
Cellular nanoscale stiffness patterns governed by intracellular forces
Nature Materials (2019)
-
Atomic force microscopy-based mechanobiology
Nature Reviews Physics (2018)