Abstract
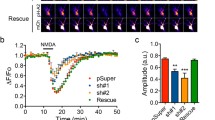
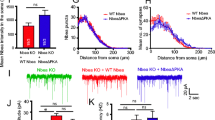
Understanding how the subcellular fate of newly synthesized AMPA receptors (AMPARs) is controlled is important for elucidating the mechanisms of neuronal function. We examined the effect of increased synthesis of AMPAR subunits on their subcellular distribution in rat hippocampal neurons. Virally expressed AMPAR subunits (GluR1 or GluR2) accumulated in cell bodies and replaced endogenous dendritic AMPAR with little effect on total dendritic amounts and caused no change in synaptic transmission. Coexpressing stargazin (STG) or mimicking GluR1 phosphorylation enhanced dendritic GluR1 levels by protecting GluR1 from lysosomal degradation. However, STG interaction or GluR1 phosphorylation did not increase surface or synaptic GluR1 levels. Unlike GluR1, STG did not protect GluR2 from lysosomal degradation or increase dendritic GluR2 levels. In general, AMPAR surface levels, and not intracellular amounts, correlated strongly with synaptic levels. Our results suggest that AMPAR surface expression, but not its intracellular production or accumulation, is critical for regulating synaptic transmission.







Similar content being viewed by others
Change history
09 July 2009
In the version of the Online Methods originally posted online, R1u in equation (1) should be R1, and R2u in equation (2) should be R2. In the text below equation (3), R1u should be R1. These errors have been corrected as of 9 July 2009.
References
Dingledine, R., Borges, K., Bowie, D. & Traynelis, S.F. The glutamate receptor ion channels. Pharmacol. Rev. 51, 7–61 (1999).
Terasaki, M., Slater, N.T., Fein, A., Schmidek, A. & Reese, T.S. Continuous network of endoplasmic reticulum in cerebellar Purkinje neurons. Proc. Natl. Acad. Sci. USA 91, 7510–7514 (1994).
Aridor, M., Guzik, A.K., Bielli, A. & Fish, K.N. Endoplasmic reticulum export site formation and function in dendrites. J. Neurosci. 24, 3770–3776 (2004).
Nicoll, R.A., Tomita, S. & Bredt, D.S. Auxiliary subunits assist AMPA-type glutamate receptors. Science 311, 1253–1256 (2006).
Ziff, E.B. TARPs and the AMPA receptor trafficking paradox. Neuron 53, 627–633 (2007).
Tomita, S. et al. Functional studies and distribution define a family of transmembrane AMPA receptor regulatory proteins. J. Cell Biol. 161, 805–816 (2003).
Vandenberghe, W., Nicoll, R.A. & Bredt, D.S. Interaction with the unfolded protein response reveals a role for stargazin in biosynthetic AMPA receptor transport. J. Neurosci. 25, 1095–1102 (2005).
Wenthold, R.J., Petralia, R.S., Blahos, J. II & Niedzielski, A.S. Evidence for multiple AMPA receptor complexes in hippocampal CA1/CA2 neurons. J. Neurosci. 16, 1982–1989 (1996).
O'Brien, R.J. et al. Activity-dependent modulation of synaptic AMPA receptor accumulation. Neuron 21, 1067–1078 (1998).
Ehlers, M.D. Reinsertion or degradation of AMPA receptors determined by activity-dependent endocytic sorting. Neuron 28, 511–525 (2000).
Patrick, G.N., Bingol, B., Weld, H.A. & Schuman, E.M. Ubiquitin-mediated proteasome activity is required for agonist-induced endocytosis of GluRs. Curr. Biol. 13, 2073–2081 (2003).
Lee, S.H., Simonetta, A. & Sheng, M. Subunit rules governing the sorting of internalized AMPA receptors in hippocampal neurons. Neuron 43, 221–236 (2004).
Nayak, A., Zastrow, D.J., Lickteig, R., Zahniser, N.R. & Browning, M.D. Maintenance of late-phase LTP is accompanied by PKA-dependent increase in AMPA receptor synthesis. Nature 394, 680–683 (1998).
Ju, W. et al. Activity-dependent regulation of dendritic synthesis and trafficking of AMPA receptors. Nat. Neurosci. 7, 244–253 (2004).
Smith, W.B., Starck, S.R., Roberts, R.W. & Schuman, E.M. Dopaminergic stimulation of local protein synthesis enhances surface expression of GluR1 and synaptic transmission in hippocampal neurons. Neuron 45, 765–779 (2005).
Sutton, M.A. et al. Miniature neurotransmission stabilizes synaptic function via tonic suppression of local dendritic protein synthesis. Cell 125, 785–799 (2006).
Mameli, M., Balland, B., Lujan, R. & Luscher, C. Rapid synthesis and synaptic insertion of GluR2 for mGluR-LTD in the ventral tegmental area. Science 317, 530–533 (2007).
Chen, N. & Napoli, J.L. All-trans-retinoic acid stimulates translation and induces spine formation in hippocampal neurons through a membrane-associated RARalpha. FASEB J. 22, 236–245 (2008).
Thiagarajan, T.C., Lindskog, M. & Tsien, R.W. Adaptation to synaptic inactivity in hippocampal neurons. Neuron 47, 725–737 (2005).
Sutton, M.A. & Schuman, E.M. Dendritic protein synthesis, synaptic plasticity and memory. Cell 127, 49–58 (2006).
Hayashi, Y. et al. Driving AMPA receptors into synapses by LTP and CaMKII: requirement for GluR1 and PDZ domain interaction. Science 287, 2262–2267 (2000).
Shi, S., Hayashi, Y., Esteban, J.A. & Malinow, R. Subunit-specific rules governing AMPA receptor trafficking to synapses in hippocampal pyramidal neurons. Cell 105, 331–343 (2001).
Shi, S.H. et al. Rapid spine delivery and redistribution of AMPA receptors after synaptic NMDA receptor activation. Science 284, 1811–1816 (1999).
Boehm, J. et al. Synaptic incorporation of AMPA receptors during LTP is controlled by a PKC phosphorylation site on GluR1. Neuron 51, 213–225 (2006).
Roche, K.W., O'Brien, R.J., Mammen, A.L., Bernhardt, J. & Huganir, R.L. Characterization of multiple phosphorylation sites on the AMPA receptor GluR1 subunit. Neuron 16, 1179–1188 (1996).
Barria, A., Muller, D., Derkach, V., Griffith, L.C. & Soderling, T.R. Regulatory phosphorylation of AMPA-type glutamate receptors by CaM-KII during long-term potentiation. Science 276, 2042–2045 (1997).
Mammen, A.L., Kameyama, K., Roche, K.W. & Huganir, R.L. Phosphorylation of the alpha-amino-3-hydroxy-5-methylisoxazole4-propionic acid receptor GluR1 subunit by calcium/calmodulin-dependent kinase II. J. Biol. Chem. 272, 32528–32533 (1997).
Kopec, C.D., Li, B., Wei, W., Boehm, J. & Malinow, R. Glutamate receptor exocytosis and spine enlargement during chemically induced long-term potentiation. J. Neurosci. 26, 2000–2009 (2006).
Rouach, N. et al. TARP gamma-8 controls hippocampal AMPA receptor number, distribution and synaptic plasticity. Nat. Neurosci. 8, 1525–1533 (2005).
Tomita, S., Stein, V., Stocker, T.J., Nicoll, R.A. & Bredt, D.S. Bidirectional synaptic plasticity regulated by phosphorylation of stargazin-like TARPs. Neuron 45, 269–277 (2005).
Chen, L., El-Husseini, A., Tomita, S., Bredt, D.S. & Nicoll, R.A. Stargazin differentially controls the trafficking of alpha-amino-3-hydroxyl-5-methyl-4-isoxazolepropionate and kainate receptors. Mol. Pharmacol. 64, 703–706 (2003).
Priel, A. et al. Stargazin reduces desensitization and slows deactivation of the AMPA-type glutamate receptors. J. Neurosci. 25, 2682–2686 (2005).
Tomita, S. et al. Stargazin modulates AMPA receptor gating and trafficking by distinct domains. Nature 435, 1052–1058 (2005).
Schnell, E. et al. Direct interactions between PSD-95 and stargazin control synaptic AMPA receptor number. Proc. Natl. Acad. Sci. USA 99, 13902–13907 (2002).
Turetsky, D., Garringer, E. & Patneau, D.K. Stargazin modulates native AMPA receptor functional properties by two distinct mechanisms. J. Neurosci. 25, 7438–7448 (2005).
Sommer, B. et al. Flip and flop: a cell-specific functional switch in glutamate-operated channels of the CNS. Science 249, 1580–1585 (1990).
Greger, I.H., Khatri, L. & Ziff, E.B. RNA editing at arg607 controls AMPA receptor exit from the endoplasmic reticulum. Neuron 34, 759–772 (2002).
Noel, J. et al. Surface expression of AMPA receptors in hippocampal neurons is regulated by an NSF-dependent mechanism. Neuron 23, 365–376 (1999).
Chen, L. et al. Stargazin regulates synaptic targeting of AMPA receptors by two distinct mechanisms. Nature 408, 936–943 (2000).
Chung, H.J., Xia, J., Scannevin, R.H., Zhang, X. & Huganir, R.L. Phosphorylation of the AMPA receptor subunit GluR2 differentially regulates its interaction with PDZ domain-containing proteins. J. Neurosci. 20, 7258–7267 (2000).
Seidenman, K.J., Steinberg, J.P., Huganir, R. & Malinow, R. Glutamate receptor subunit 2 serine 880 phosphorylation modulates synaptic transmission and mediates plasticity in CA1 pyramidal cells. J. Neurosci. 23, 9220–9228 (2003).
Lu, W. & Ziff, E.B. PICK1 interacts with ABP/GRIP to regulate AMPA receptor trafficking. Neuron 47, 407–421 (2005).
Osten, P. & Stern-Bach, Y. Learning from stargazin: the mouse, the phenotype and the unexpected. Curr. Opin. Neurobiol. 16, 275–280 (2006).
Cummins, T.R. et al. Nav1.3 sodium channels: rapid repriming and slow closed-state inactivation display quantitative differences after expression in a mammalian cell line and in spinal sensory neurons. J. Neurosci. 21, 5952–5961 (2001).
Baroudi, G., Carbonneau, E., Pouliot, V. & Chahine, M. SCN5A mutation (T1620M) causing Brugada syndrome exhibits different phenotypes when expressed in Xenopus oocytes and mammalian cells. FEBS Lett. 467, 12–16 (2000).
Mantegazza, M. et al. Molecular determinants for modulation of persistent sodium current by G protein betagamma subunits. J. Neurosci. 25, 3341–3349 (2005).
Wang, R. et al. Evolutionary conserved role for TARPs in the gating of glutamate receptors and tuning of synaptic function. Neuron 59, 997–1008 (2008).
Milstein, A.D., Zhou, W., Karimzadegan, S., Bredt, D.S. & Nicoll, R.A. TARP subtypes differentially and dose-dependently control synaptic AMPA receptor gating. Neuron 55, 905–918 (2007).
Menuz, K., Kerchner, G.A., O'Brien, J.L. & Nicoll, R.A. Critical role for TARPs in early development despite broad functional redundancy. Neuropharmacology 56, 22–29 (2008).
Hu, H. et al. Emotion enhances learning via norepinephrine regulation of AMPA-receptor trafficking. Cell 131, 160–173 (2007).
Acknowledgements
We thank N. Dawkins for expert technical assistance, J. Boehm for making viral constructs, R. Huganir for kindly providing STG cDNA and GluR1-deficient mice, and all of the members of the Malinow laboratory for useful discussions. This work was supported by grants from the Netherlands Organization for Scientific Research (H.W.K.), the Undergraduate Research Program of Cold Spring Harbor Laboratory (M.E.K.) and the US National Institutes of Health (R.M.).
Author information
Authors and Affiliations
Contributions
H.W.K. performed all of the experimental work. H.W.K. and R.M. designed the experiments and wrote the manuscript. C.D.K. wrote custom software for imaging analysis. M.E.K. designed the protocol for the immunohistochemistry experiments.
Corresponding author
Supplementary information
Supplementary Text and Figures
Supplementary Figures 1–5 (PDF 289 kb)
Rights and permissions
About this article
Cite this article
Kessels, H., Kopec, C., Klein, M. et al. Roles of stargazin and phosphorylation in the control of AMPA receptor subcellular distribution. Nat Neurosci 12, 888–896 (2009). https://doi.org/10.1038/nn.2340
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/nn.2340
- Springer Nature America, Inc.
This article is cited by
-
Oxidative stress and inflammation cause auditory system damage via glial cell activation and dysregulated expression of gap junction proteins in an experimental model of styrene-induced oto/neurotoxicity
Journal of Neuroinflammation (2024)
-
AMPA receptors and their minions: auxiliary proteins in AMPA receptor trafficking
Cellular and Molecular Life Sciences (2019)
-
Palmitoylation-mediated synaptic regulation of AMPA receptor trafficking and function
Archives of Pharmacal Research (2019)
-
R/G editing in GluA2Rflop modulates the functional difference between GluA1 flip and flop variants in GluA1/2R heteromeric channels
Scientific Reports (2017)
-
Synaptic AMPA receptor composition in development, plasticity and disease
Nature Reviews Neuroscience (2016)