Abstract
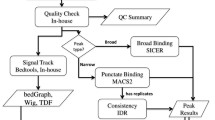
We evaluated how variations in sequencing depth and other parameters influence interpretation of chromatin immunoprecipitation–sequencing (ChIP-seq) experiments. Using Drosophila melanogaster S2 cells, we generated ChIP-seq data sets for a site-specific transcription factor (Suppressor of Hairy-wing) and a histone modification (H3K36me3). We detected a chromatin-state bias: open chromatin regions yielded higher coverage, which led to false positives if not corrected. This bias had a greater effect on detection specificity than any base-composition bias. Paired-end sequencing revealed that single-end data underestimated ChIP-library complexity at high coverage. Removal of reads originating at the same base reduced false-positives but had little effect on detection sensitivity. Even at mappable-genome coverage depth of ∼1 read per base pair, ∼1% of the narrow peaks detected on a tiling array were missed by ChIP-seq. Evaluation of widely used ChIP-seq analysis tools suggests that adjustments or algorithm improvements are required to handle data sets with deep coverage.





Similar content being viewed by others
Accession codes
References
Johnson, D.S., Mortazavi, A., Myers, R.M. & Wold, B. Genome-wide mapping of in vivo protein-DNA interactions. Science 316, 1497–1502 (2007).
Robertson, G. et al. Genome-wide profiles of STAT1 DNA association using chromatin immunoprecipitation and massively parallel sequencing. Nat. Methods 4, 651–657 (2007).
Barski, A. et al. High-resolution profiling of histone methylations in the human genome. Cell 129, 823–837 (2007).
Mikkelsen, T.S. et al. Genome-wide maps of chromatin state in pluripotent and lineage-committed cells. Nature 448, 553–560 (2007).
Johnson, D.S. et al. Systematic evaluation of variability in ChIP-chip experiments using predefined DNA targets. Genome Res. 18, 393–403 (2008).
Ho, J.W. et al. ChIP-chip versus ChIP-seq: lessons for experimental design and data analysis. BMC Genomics 12, 134 (2011).
Zhang, Y. et al. Model-based analysis of ChIP-Seq (MACS). Genome Biol. 9, R137 (2008).
Laajala, T.D. et al. A practical comparison of methods for detecting transcription factor binding sites in ChIP-seq experiments. BMC Genomics 10, 618 (2009).
Wilbanks, E.G. & Facciotti, M.T. Evaluation of algorithm performance in ChIP-seq peak detection. PLoS ONE 5, e11471 (2010).
Rozowsky, J. et al. PeakSeq enables systematic scoring of ChIP-seq experiments relative to controls. Nat. Biotechnol. 27, 66–75 (2009).
Negre, N. et al. A comprehensive map of insulator elements for the Drosophila genome. PLoS Genet. 6, e1000814 (2010).
Myers, R.M. et al. A user's guide to the encyclopedia of DNA elements (ENCODE). PLoS Biol. 9, e1001046 (2011).
Pepke, S., Wold, B. & Mortazavi, A. Computation for ChIP-seq and RNA-seq studies. Nat. Methods 6, S22–S32 (2009).
Kolasinska-Zwierz, P. et al. Differential chromatin marking of introns and expressed exons by H3K36me3. Nat. Genet. 41, 376–381 (2009).
Dohm, J.C., Lottaz, C., Borodina, T. & Himmelbauer, H. Substantial biases in ultra-short read data sets from high-throughput DNA sequencing. Nucleic Acids Res. 36, e105 (2008).
Kozarewa, I. et al. Amplification-free Illumina sequencing-library preparation facilitates improved mapping and assembly of (G+C)-biased genomes. Nat. Methods 6, 291–295 (2009).
Kharchenko, P.V. et al. Comprehensive analysis of the chromatin landscape in Drosophila melanogaster. Nature 471, 480–485 (2011).
Negre, N. et al. A cis-regulatory map of the Drosophila genome. Nature 471, 527–531 (2011).
Roy, S. et al. Identification of functional elements and regulatory circuits by Drosophila modENCODE. Science 330, 1787–1797 (2010).
Larschan, E. et al. X chromosome dosage compensation via enhanced transcriptional elongation in Drosophila. Nature 471, 115–118 (2011).
Teytelman, L. et al. Impact of chromatin structures on DNA processing for genomic analyses. PLoS ONE 4, e6700 (2009).
Feng, X., Grossman, R. & Stein, L. PeakRanger: a cloud-enabled peak caller for ChIP-seq data. BMC Bioinformatics 12, 139 (2011).
Rashid, N.U., Giresi, P.G., Ibrahim, J.G., Sun, W. & Lieb, J.D. ZINBA integrates local covariates with DNA-seq data to identify broad and narrow regions of enrichment, even within amplified genomic regions. Genome Biol. 12, R67 (2011).
Ji, H. et al. An integrated software system for analyzing ChIP-chip and ChIP-seq data. Nat. Biotechnol. 26, 1293–1300 (2008).
Jothi, R., Cuddapah, S., Barski, A., Cui, K. & Zhao, K. Genome-wide identification of in vivo protein-DNA binding sites from ChIP-Seq data. Nucleic Acids Res. 36, 5221–5231 (2008).
Kharchenko, P.V., Tolstorukov, M.Y. & Park, P.J. Design and analysis of ChIP-seq experiments for DNA-binding proteins. Nat. Biotechnol. 26, 1351–1359 (2008).
Nix, D.A., Courdy, S.J. & Boucher, K.M. Empirical methods for controlling false positives and estimating confidence in ChIP-Seq peaks. BMC Bioinformatics 9, 523 (2008).
Valouev, A. et al. Genome-wide analysis of transcription factor binding sites based on ChIP-Seq data. Nat. Methods 5, 829–834 (2008).
Johnson, W.E. et al. Model-based analysis of tiling-arrays for ChIP-chip. Proc. Natl. Acad. Sci. USA 103, 12457–12462 (2006).
Li, Q., Brown, J.B., Huang, H. & Bickel, P.J. Measuring reproducibility of high-throughput experiments. Annals of Applied Statistics 5, 1752–1779 (2011).
Benson, G. Tandem repeats finder: a program to analyze DNA sequences. Nucleic Acids Res. 27, 573–580 (1999).
Mortazavi, A., Williams, B.A., McCue, K., Schaeffer, L. & Wold, B. Mapping and quantifying mammalian transcriptomes by RNA-Seq. Nat. Methods 5, 621–628 (2008).
Boyle, A.P., Guinney, J., Crawford, G.E. & Furey, T.S. F-Seq: a feature density estimator for high-throughput sequence tags. Bioinformatics 24, 2537–2538 (2008).
Zhang, Y. et al. Expression in aneuploid Drosophila S2 cells. PLoS Biol. 8, e1000320 (2010).
Celniker, S.E. et al. Unlocking the secrets of the genome. Nature 459, 927–930 (2009).
Quinlan, A.R. & Hall, I.M. BEDTools: a flexible suite of utilities for comparing genomic features. Bioinformatics 26, 841–842 (2010).
Acknowledgements
We thank the authors of all of the algorithms that we evaluated in this study: H. Ji, R. Jothi, P. Kharchenko, W. Li, D. Nix, J. Rozowsky and A. Valouev. We thank N. Bild, D. Roqueiro and M. Sabala for help in performing PeakSeq on the Bionimbus Cloud, D. Schmidt and D. Odom for sharing their sequencing data of the ENCODE spike-in sample, A. Kundaje for sharing his unpublished results on IDR analysis of H3K36me3 in humans, N. Rashid for sharing the mappability data of Drosophila genome, M. Greenberg for support in the early stage of this project, and E. Birney, M. Snyder, J. Ahringer, M. Gerstein, M. Kellis, P. Park and other members of modENCODE consortium for helpful discussions. This work was partially funded by US National Institutes of Health (HG4069 to X.S.L., 3U01 HG004270-03S1 to X.S.L. and J.D.L., and U01HG004264 to K.P.W.).
Author information
Authors and Affiliations
Contributions
Y.C. performed bioinformatic analysis. N.N. performed cell culture, ChIP experiments and library preparation with help from J.Z. J.O.M. performed library preparation and sequencing experiments. Q.L. and P.J.B. contributed code for the IDR method. Q.L. participated in writing the description of IDR method and interpretation of the IDR analysis result. M.S. performed ChIP–quantitative (q)PCR validation of the selected array-specific Su(Hw) peaks and analyzed the ChIP-qPCR data. T.L., Y.Z., T.-K.K., H.H.H., Y.R., R.M.M. and B.J.W. contributed to the early development of the project. B.J.W., K.P.W., J.D.L. and X.S.L. conceived the project. T.-K.K., H.H.H., Y.R. and R.M.M. performed pilot experiments. Y.C., J.D.L. and X.S.L. wrote the manuscript with the help from other authors.
Corresponding authors
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Supplementary information
Supplementary Text and Figures
Supplementary Figures 1–16 , Supplementary Table 1, Supplementary Notes, Supplementary Methods (PDF 4131 kb)
Rights and permissions
About this article
Cite this article
Chen, Y., Negre, N., Li, Q. et al. Systematic evaluation of factors influencing ChIP-seq fidelity. Nat Methods 9, 609–614 (2012). https://doi.org/10.1038/nmeth.1985
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/nmeth.1985
- Springer Nature America, Inc.
This article is cited by
-
Hypoxic preconditioning induces epigenetic changes and modifies swine mesenchymal stem cell angiogenesis and senescence in experimental atherosclerotic renal artery stenosis
Stem Cell Research & Therapy (2021)
-
A computational method for estimating the PCR duplication rate in DNA and RNA-seq experiments
BMC Bioinformatics (2017)
-
Analysis and imaging of biocidal agrochemicals using ToF-SIMS
Scientific Reports (2017)
-
Systematic evaluation of the impact of ChIP-seq read designs on genome coverage, peak identification, and allele-specific binding detection
BMC Bioinformatics (2016)