Abstract
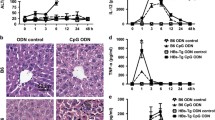
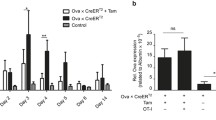
The Fas ligand (FasL) is expressed in activated T cells and induces apoptosis in Fas-bearing cells. A cytotoxic T lymphocyte (CTL) clone specific for hepatitis B surface antigen (HBsAg) causes an acute liver disease in HBsAg transgenic mice. Here we observed that the CTL clone killed hepatocytes expressing HBsAg in a Fas-dependent manner. Administration of the soluble form of Fas into HBsAg transgenic mice prevented the CTL-induced liver disease. In the second model, mice were primed with Propionibacterium acnes. A subsequent challenge with lipopolysaccharide (LPS) killed the mice by inducing liver injury. Neutralization of FasL rescued the mice from LPS-induced mortality, and Fas-null mice were resistant to LPS-induced mortality. These results suggest that FasL has an essential role in the development of hepatitis.
Similar content being viewed by others
References
Nagata, S. & Golstein, P., Fas death factor. Science 267, 1449–1456 (1995).
Rouvier, E., Luciani, M.-F. & Golstein, P. Fas involvement in Ca2+-independentT Cell-mediated cytotoxicity. J. Exp. Med. 177, 195–200 (1993).
Hanabuchi, S. et al. Fas and its ligand in a general mechanism of T-Cell mediated cy-totoxicity. Proc. Natl. Acad. Sci. USA 91, 4930–4934 (1994).
Suda, T. et al. Expression of the Fas ligand in T-Cell-lineage. J. Immunol. 154, 3806–3813 (1995).
Vignaux, F. et al. TCR/CD3 coupling to Fas-based cytotoxicity. J. Exp. Med. 181, 781–786 (1995).
Kägi, D. et al. Fas and perforin pathway as major mechanisms of T Cell-mediated cy-totoxicity. Science 265, 528–530 (1994).
Kojima, H. et al. Two distinct pathways of specific killing revealed by perforin mu-tant cytotoxic T lymphocytes. Immunity 1, 357–364 (1994).
Lowin, B., Hahne, M., Mattmann, C. & Tschopp, J. Cytolytic T-Cell cytotoxicity is mediated through perforin and Fas lytic pathways. Nature 370, 650–652 (1994).
Watanabe-Fukunaga, R. et al. The cDNA structure, expression, and chromosomal assignment of the mouse Fas antigen. J. Immunol. 148, 1274–1279 (1992).
Ogasawara, J. et al. Lethal effect of the anti-Fas antibody in mice. Nature 364, 806–809 (1993).
Hoofnagle, J., Carithers, R., Shapiro, C. & Ascher, N. Fulminant hepatic failure: Summary of a workshop. Hepatology 21, 240–252 (1995).
Berke, G., CTL's kiss of death. Cell 81, 9–12 (1994).
Rose, N.R. & Bona, C. Defining criteria for autoimmune disease (Witebsky's postu-lates revisited). Immunol. Today 14, 426–430 (1993).
Ando, K. et al. Mechanisms of class I restricted immunopathology. A transgenic mouse model of fulminant hepatitis. J. Exp. Med. 178, 1541–1554 (1993).
Moriyama, T. et al. Immunobiology and pathogenesis of hepatoCellular injury in hepatitis B virus transgenic mice. Science 248, 361–364 (1990).
Ferluga, J. & Allison, A. Role of mononuclear infiltrating Cells in pathogenesis of hepatitis. Lancet 2, 610–611 (1978).
Sato, T. et al. Inhibition of Corynebacterium porvum-primed and lipopolysaccharide-induced hepatic necrosis in rats by selective depletion of neutrophils using a mono-clonal antibody. J. Leuk. Biol. 53, 144–150 (1993).
Fujioka, N. et al. Preparation of specific antibodies against murine IL-1 ra and the establishment of IL-1 ra as an endogenous regulator of bacteria-induced fulminant hepatitis in mice. J. Leuk. Biol. 58, 90–98 (1995).
Chisari, F.V. & Ferrari, C. Hepatitis B virus immunopathogenesis. Annu. Rev. Immunol. 13, 29–60 (1995).
Kataoka, T. et al. Acidification is essential for maintaining the structure and function of lytic granules of CTL. J. Immunol. 153, 3938–3947 (1994).
Gavrieli, Y., Sherman, Y., St Ben-Sasson, S.A. Identification of programmed Cell death in situ via specific labeling of nuclear DNA fragmentation. J. Cell Biol. 119, 493–501 (1992).
Adachi, M. et al. Targeted mutation in the Fas gene causes hyperplasia in the pe-ripheral lymphoid organs and liver. Nature Cenet. 11, 294–300 (1995).
Hiramatsu, N. et al. Immunohistochemical detection of Fas antigen in liver tissue of patients with chronic hepatitis C. Hepatology 19, 1354–1359 (1994).
Vilanova, M. et al. Role of monocytes in the up-regulation of the early activation marker CD69 on B and T murine lymphocytes induced by microbial mitogens. Scand. J. Immunol. 43, 155–163 (1996).
Kägi, D. et al. Cytotoxicity mediated by T Cells and natural killer Cells is greatly impaired in perforin-deficient mice. Nature 369, 31–37 (1994).
Guidotti, L. et al. IntraCellular inactivation of the hepatitis B virus by cytotoxic T lymphocytes. Immunity 4, 25–36 (1996).
Ellis, R.E., Yuan, J. & Horvitz, H.R. Mechanisms and functions of Cell death. Annu. Rev. Cell Biol. 7, 663–698 (1991).
Ding, A., Nathan, C. & Stuehr, D. Release of reactive nitrogen intermediates and re active oxygen intermediates from mouse peritoneal macrophages. J. Immunol. 141, 2407–2412 (1988).
Stuehr, D. & Marietta, M. Induction of nitrite/nitrate synthesis in murine macrophages by BCC infection, lymphokines or interferon-γ. J. Immunol. 139, 518–525 (1987).
Tanaka, M. et al. Fas ligand in human serum. Nature Med. 2, 317–322 (1996).
Enari, M., Hug, H., St Nagata, S. Involvement of an ICE-like protease in Fas-mediated apoptosis. Nature 375, 78–81 (1995).
Los, M. et al. Requirement of an ICE/CED-3 protease for Fas/APO-1-mediated apop-tosis. Nature 375, 81–83 (1995).
Suda, T. & Nagata, S. Purification and characterization of the Fas ligand that in-duces apoptosis. J. Exp. Med. 179, 873–878 (1994).
Torres, R.M. & Clark, E.A. Differential increase of an alternatively polyadenylated mRNA species of murine CD40 upon B lymphocyte activation. J. Immunol. 148, 620–626 (1992).
Adachi, M. et al. Enhanced and accelerated lymphoproliferation in Fas-null mice. Proc. Natl. Acad. Sci. USA 93, 2137–2142 (1996).
Ni, R. et al. Fas-mediated apoptosis in primary cultured mouse hepatocytes. Exp. Cell Res. 215, 332–337 (1994).
Selgen, P.O. Preparation of isolated rat liver Cells. Methods Cell Biol. 13, 29–83 (1976).
Takahashi, T. et al. Generalized lymphoproliferative disease in mice, caused by a point mutation in the Fas ligand. Cell 76, 969–976 (1994).
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Kondo, T., Suda, T., Fukuyama, H. et al. Essential roles of the Fas ligand in the development of hepatitis. Nat Med 3, 409–413 (1997). https://doi.org/10.1038/nm0497-409
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1038/nm0497-409
- Springer Nature America, Inc.
This article is cited by
-
Harnessing TRAIL-induced cell death for cancer therapy: a long walk with thrilling discoveries
Cell Death & Differentiation (2023)
-
Adenosine deaminase as a marker for the severity of infectious mononucleosis secondary to EBV in children
BMC Infectious Diseases (2022)
-
Potent pro-apoptotic combination therapy is highly effective in a broad range of cancers
Cell Death & Differentiation (2022)
-
Hypertonicity-enforced BCL-2 addiction unleashes the cytotoxic potential of death receptors
Oncogene (2018)
-
Liver receptor homolog-1 (NR5a2) regulates CD95/Fas ligand transcription and associated T-cell effector functions
Cell Death & Disease (2017)