Abstract
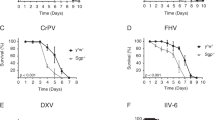
The response of drosophila to bacterial and fungal infections involves two signaling pathways, Toll and Imd, which both activate members of the transcription factor NF-κB family. Here we have studied the global transcriptional response of flies to infection with drosophila C virus. Viral infection induced a set of genes distinct from those regulated by the Toll or Imd pathways and triggered a signal transducer and activator of transcription (STAT) DNA-binding activity. Genetic experiments showed that the Jak kinase Hopscotch was involved in the control of the viral load in infected flies and was required but not sufficient for the induction of some virus-regulated genes. Our results indicate that in addition to Toll and Imd, a third, evolutionary conserved innate immunity pathway functions in drosophila and counters viral infection.






Similar content being viewed by others
References
Hoffmann, J. The immune response of Drosophila . Nature 426, 33–38 (2003).
Tzou, P., De Gregorio, E. & Lemaitre, B. How Drosophila combats microbial infection: a model to study innate immunity and host-pathogen interactions. Curr. Opin. Microbiol. 5, 102–110 (2002).
Hultmark, D. Drosophila immunity: paths and patterns. Curr. Opin. Immunol. 15, 12–19 (2003).
Silverman, N. & Maniatis, T. NF-κB signaling pathways in mammalian and insect innate immunity. Genes Dev. 15, 2321–2342 (2001).
Brennan, C.A. & Anderson, K.V. Drosophila: the genetics of innate immune recognition and response. Annu. Rev. Immunol. 22, 457–483 (2004).
Jousset, F.X., Bergoin, M. & Revet, B. Characterization of the Drosophila C virus. J. Gen. Virol. 34, 269–283 (1977).
Keene, K.M. et al. RNA interference acts as a natural antiviral response to O'nyong-nyong virus (Alphavirus; Togaviridae) infection of Anopheles gambiae . Proc. Natl. Acad. Sci. USA 101, 17240–17245 (2004).
Roxstrom-Lindquist, K., Terenius, O. & Faye, I. Parasite-specific immune response in adult Drosophila melanogaster: a genomic study. EMBO Rep. 5, 207–212 (2004).
Mackenzie, J.S., Gubler, D.J. & Petersen, L.R. Emerging flaviviruses: the spread and resurgence of Japanese encephalitis, West Nile and dengue viruses. Nat. Med. 10, S98–S109 (2004).
Cherry, S. & Perrimon, N. Entry is a rate-limiting step for viral infection in a Drosophila melanogaster model of pathogenesis. Nat. Immunol. 5, 81–87 (2004).
Sabatier, L. et al. Pherokine-2 and -3: Two Drosophila molecules related to pheromone/odor-binding proteins induced by viral and bacterial infections. Eur. J. Biochem. 270, 3398–3407 (2003).
Cherry, S. et al. Genome-wide RNAi screen reveals a specific sensitivity of IRES-containing RNA viruses to host translation inhibition. Genes Dev. 19, 445–452 (2005).
Johnson, K.N. & Christian, P.D. The novel genome organization of the insect picorna-like virus Drosophila C virus suggests this virus belongs to a previously undescribed virus family. J. Gen. Virol. 79, 191–203 (1998).
Yan, R., Small, S., Desplan, C., Dearolf, C.R. & Darnell, J.E., Jr. Identification of a Stat gene that functions in Drosophila development. Cell 84, 421–430 (1996).
Perrimon, N. & Mahowald, A.P. l(1) hopscotch, a larval-pupal zygotic lethal with a specific maternal effect on segmentation in Drosophila . Dev. Biol. 118, 28–41 (1986).
Irving, P. et al. A genome-wide analysis of immune responses in Drosophila . Proc. Natl. Acad. Sci. USA 98, 15119–15124 (2001).
De Gregorio, E., Spellman, P.T., Rubin, G.M. & Lemaitre, B. Genome-wide analysis of the Drosophila immune response by using oligonucleotide microarrays. Proc. Natl. Acad. Sci. USA 98, 12590–12595 (2001).
Boutros, M., Agaisse, H. & Perrimon, N. Sequential activation of signaling pathways during innate immune responses in Drosophila . Dev. Cell 3, 711–722 (2002).
Ekengren, S. et al. A humoral stress response in Drosophila . Curr. Biol. 11, 714–718 (2001).
Tihova, M. et al. Nodavirus coat protein imposes dodecahedral RNA structure independent of nucleotide sequence and length. J. Virol. 78, 2897–2905 (2004).
Hou, S.X., Zheng, Z., Chen, X. & Perrimon, N. The Jak/STAT pathway in model organisms: emerging roles in cell movement. Dev. Cell 3, 765–778 (2002).
Hombria, J.C. & Brown, S. The fertile field of Drosophila Jak/STAT signalling. Curr. Biol. 12, R569–R575 (2002).
Agaisse, H., Petersen, U.M., Boutros, M., Mathey-Prevot, B. & Perrimon, N. Signaling role of hemocytes in Drosophila JAK/STAT-dependent response to septic injury. Dev. Cell 5, 441–450 (2003).
Harrison, D., Binari, R., Stines Nahreini, T., Gilman, M. & Perrimon, N. Activation of a Drosophila Janus kinase (JAK) causes hematopoietic neoplasia and developmental defects. EMBO J. 14, 2857–2865 (1995).
Luo, H., Hanratty, W. & Dearolf, C. An amino acid sustitution in the Drosophila hopTum-l Jak kinase causes leukemia-like hematopoietic defects. EMBO J. 14, 1412–1420 (1995).
Brown, S., Hu, N. & Hombria, J.C. Identification of the first invertebrate interleukin JAK/STAT receptor, the Drosophila gene domeless. Curr. Biol. 11, 1700–1705 (2001).
Chen, H.W. et al. mom identifies a receptor for the Drosophila JAK/STAT signal transduction pathway and encodes a protein distantly related to the mammalian cytokine receptor family. Genes Dev. 16, 388–398 (2002).
Betz, A., Lampen, N., Martinek, S., Young, M.W. & Darnell, J.E., Jr. A Drosophila PIAS homologue negatively regulates stat92E. Proc. Natl. Acad. Sci. USA 98, 9563–9568 (2001).
Brandt, S.M. et al. Secreted bacterial effectors and host-produced Eiger/TNF drive death in a salmonella-infected fruit fly. PLoS Biol. 2, 2067–2075 (2004).
Ganz, T. Defensins: antimicrobial peptides of innate immunity. Nat. Rev. Immunol. 3, 710–720 (2003).
Chinchar, V.G. et al. Inactivation of viruses infecting ectothermic animals by amphibian and piscine antimicrobial peptides. Virology 323, 268–275 (2004).
Irusta, P.M., Chen, Y.B. & Hardwick, J.M. Viral modulators of cell death provide new links to old pathways. Curr. Opin. Cell Biol. 15, 700–705 (2003).
Lecellier, C.H. & Voinnet, O. RNA silencing: no mercy for viruses? Immunol. Rev. 198, 285–303 (2004).
Li, H., Li, W.X. & Ding, S.W. Induction and suppression of RNA silencing by an animal virus. Science 296, 1319–1321 (2002).
Gobert, V. et al. Dual activation of the Drosophila toll pathway by two pattern recognition receptors. Science 302, 2126–2130 (2003).
Pili-Floury, S. et al. In vivo RNA interference analysis reveals an unexpected role for GNBP1 in the defense against Gram-positive bacterial infection in Drosophila adults. J. Biol. Chem. 279, 12848–12853 (2004).
Harvey, N.L. et al. Characterization of the Drosophila caspase, DAMM. J. Biol. Chem. 276, 25342–25350 (2001).
Diveu, C. et al. GPL, a novel cytokine receptor related to GP130 and leukemia inhibitory factor receptor. J. Biol. Chem. 278, 49850–49859 (2003).
Harrison, D.A., McCoon, P.E., Binari, R., Gilman, M. & Perrimon, N. Drosophila unpaired encodes a secreted protein that activates the JAK signaling pathway. Genes Dev. 12, 3252–3263 (1998).
Barillas-Mury, C., Han, Y.S., Seeley, D. & Kafatos, F.C. Anopheles gambiae Ag-STAT, a new insect member of the STAT family, is activated in response to bacterial infection. EMBO J. 18, 959–967 (1999).
Lagueux, M., Perrodou, E., Levashina, E.A., Capovilla, M. & Hoffmann, J.A. Constitutive expression of a complement-like protein in toll and JAK gain-of-function mutants of Drosophila . Proc. Natl. Acad. Sci. USA 97, 11427–11432 (2000).
Lin, C.C. et al. Characterization of two mosquito STATs, AaSTAT and CtSTAT. Differential regulation of tyrosine phosphorylation and DNA binding activity by lipopolysaccharide treatment and by Japanese encephalitis virus infection. J. Biol. Chem. 279, 3308–3317 (2004).
Silvennoinen, O., Ihle, J.N., Schlessinger, J. & Levy, D.E. Interferon-induced nuclear signalling by Jak protein tyrosine kinases. Nature 366, 583–585 (1993).
Shuai, K. et al. Polypeptide signalling to the nucleus through tyrosine phosphorylation of Jak and Stat proteins. Nature 366, 580–583 (1993).
Watling, D. et al. Complementation by the protein tyrosine kinase JAK2 of a mutant cell line defective in the interferon-γ signal transduction pathway. Nature 366, 166–170 (1993).
Dupuis, S. et al. Impaired response to interferon-α/β and lethal viral disease in human STAT1 deficiency. Nat. Genet. 33, 388–391 (2003).
Karst, S.M., Wobus, C.E., Lay, M., Davidson, J. & Virgin, H.W. 4th. STAT1-dependent innate immunity to a Norwalk-like virus. Science 299, 1575–1578 (2003).
Lemaitre, B., Nicolas, E., Michaut, L., Reichhart, J. & Hoffmann, J. The dorsoventral regulatory gene cassette spätzle/Toll/cactus controls the potent antifungal response in Drosophila adults. Cell 86, 973–983 (1996).
Jung, A.C., Criqui, M.C., Rutschmann, S., Hoffmann, J.A. & Ferrandon, D. Microfluorometer assay to measure the expression of β-galactosidase and green fluorescent protein reporter genes in single Drosophila flies. Biotechniques 30, 594–8, 600–1 (2001).
Gottar, M. et al. The Drosophila immune response against Gram-negative bacteria is mediated by a peptidoglycan recognition protein. Nature 416, 640–644 (2002).
Acknowledgements
We thank S. Ozkan for help with transgenesis; M.-C. Lacombe for assistance with real-time quantitative RT-PCR analysis; J. Mutterer and D. Zachary for help with the confocal microscopy; E. Santiago for technical support; D. Ferrandon and J. Royet for discussions and critical reading of the manuscript; N. Perrimon, J. Castelli-Gair Hombria and J. Darnell for providing fly stocks; and A. Schneemann for the FHV stock and discussions. Supported by Centre National de la Recherche Scientifique and Ministère de la Recherche et de la Technologie (Actions Concertées Incitatives Physiologie Intégrative and Microbiologie, and Programme puces Affymetrix); Institut Universitaire de France; Ligue contre le Cancer (E.J.); Centre National de la Recherche Scientifique (D.G.A.); and Ministère de la Recherche du Grand-Duché de Luxembourg (C.D.).
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Supplementary information
Supplementary Fig. 1
Susceptibility of wild-type flies to DCV infection. (PDF 98 kb)
Supplementary Fig. 2
DCV infection upregulates expression of genes across a broad range of functional categories. (PDF 56 kb)
Supplementary Fig. 3
Characterization of the vir-1 transcript and promoter. (PDF 112 kb)
Supplementary Fig. 4
Constitutive activity of the vir-1 promoter in S2 cells is dependent on the presence of a consensus STAT92E binding site at position -342. (PDF 91 kb)
Supplementary Fig. 5
Susceptibility of wild-type and hopM38/msvl mutant flies to infection with different doses of DCV. (PDF 93 kb)
Supplementary Table 1
Genes up-regulated at least two-fold or induced by DCV infection. (PDF 91 kb)
Supplementary Table 2
Sequence and position of the STAT92E consensus binding sites in the promoters of the virus-regulated genes vir-1, CG12780, and CG9080. (PDF 76 kb)
Rights and permissions
About this article
Cite this article
Dostert, C., Jouanguy, E., Irving, P. et al. The Jak-STAT signaling pathway is required but not sufficient for the antiviral response of drosophila. Nat Immunol 6, 946–953 (2005). https://doi.org/10.1038/ni1237
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/ni1237
- Springer Nature America, Inc.
This article is cited by
-
Drosophila caspases as guardians of host-microbe interactions
Cell Death & Differentiation (2023)
-
Innate immune pathways act synergistically to constrain RNA virus evolution in Drosophila melanogaster
Nature Ecology & Evolution (2022)
-
Molecular characterization and expression patterns of heat shock proteins in Spodoptera littoralis, heat shock or immune response?
Cell Stress and Chaperones (2021)
-
Transcriptome response comparison between vector and non-vector aphids after feeding on virus-infected wheat plants
BMC Genomics (2020)
-
Interactions between Borrelia burgdorferi and ticks
Nature Reviews Microbiology (2020)