Abstract
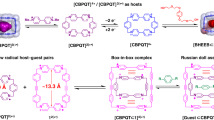
The ‘blue box’ (cyclobis(paraquat-p-phenylene) or CBPQT4+), developed by Stoddart and colleagues, forms effective charge transfer complexes with a variety of electron-rich species and has been used to support the formation of a wide range of interlocked structures. However, little effort seems to have been devoted to generalizing the blue box concept. We describe a new flexible tetracationic macrocycle—a ‘Texas-sized’ molecular box. This positively charged receptor is capable of binding the mono-terephthalate anion, forming pseudorotaxanes. These pseudorotaxanes self-assemble into short supramolecular pseudo-oligorotaxanes in solution and more extended pseudo-polyrotaxanes in the solid state. The supramolecular oligomers formed in solution are environmentally responsive; they undergo deaggregation as the overall concentration of the cationic and anionic constituents is reduced, the temperature is increased, or the protonation state of the threading mono-terephthalate anion is changed.




Similar content being viewed by others
References
Schill, G. Catenanes, Rotaxanes and Knots (Academic Press, 1971).
Dietrich-Buchecker, C. & Sauvage, J.-P. Interlocking of molecular threads: from the statistical approach to the templated synthesis of catenands. Chem. Rev. 87, 795–810 (1987), and references therein.
Fujita, M. Self-assembly of [2]catenanes containing metals in their backbones. Acc. Chem. Res. 32, 53–61 (1999), and references therein.
Amabilino, D. B. & Stoddart, J. F. Interlocked and intertwined structures and superstructures. Chem. Rev. 95, 2725–2828 (1995), and references therein.
Schalley, C. A., Weilandt, T., Brüggemann, J. & Vögtle, F. Hydrogen-bond-mediated template synthesis of rotaxanes, catenanes and knotanes. Top. Curr. Chem. 248, 141–200 (2004).
Lankshear, M. D. & Beer, P. D. Interweaving anion templation. Acc. Chem. Res. 40, 657–668 (2007), and references therein.
Chmielewski, M. J. et al. Sulfate anion templation of a neutral pseudorotaxane assembly using an indolocarbazole threading component. Chem. Commun. 3154–3156 (2008).
Beer, P. D., Sambrook, M. R. & Curiel, D. Anion-templated assembly of interpenetrated and interlocked structures. Chem. Commun. 2105–2117 (2006).
Chiu, S.-H. et al. Making molecular-necklaces from rotaxanes. Tetrahedron 58, 807–814 (2002).
Chang, T. et al. Toward interlocked molecules beyond catenanes and rotaxanes. Org. Lett. 2, 2943–2946 (2000).
Lipatov, Y. S., Lipatova, T. E. & Kosyanchuk, L. F. Synthesis and structure of macromolecular topological compounds. Adv. Polym. Sci. 88, 49–76 (1989).
Shen, Y. X., Xie, D. & Gibson, H. W. Polyrotaxanes based on polyurethane backbones and crown ether cyclics. 1. Synthesis. J. Am. Chem. Soc. 116, 537–548 (1994).
Feringa, B. L. Molecular Switches (Wiley-VCH, 2001), and references therein.
Pease, A. R. et al. Switching devices based on interlocked molecules. Acc. Chem. Res. 34, 433–444 (2001), and references therein.
Muraoka, T., Kinbara, K. & Aida, T. Mechanical twisting of a guest by a photoresponsive host. Nature 440, 512–515 (2006).
Kay, E. R., Leigh, D. A. & Zerbetto, F. Synthetic molecular motors and mechanical machines. Angew. Chem. Int. Ed. 46, 72–191 (2007).
Odell, B. et al. Cyclobis(paraquat-p-phenylene). A tetracationic multipurpose receptor. Angew. Chem. Int. Ed. Engl. 27, 1547–1550 (1988).
Amabilino, D. B. & Stoddart, J. F. Self-assembly and macromolecular design. Pure Appl. Chem. 65, 2351–2359 (1993).
Langford, S. J. & Stoddart, J. F. Self-assembly in chemical systems. Pure Appl. Chem. 68, 1255–1260 (1996).
Anelli, P. L., Spencer, N. & Stoddart, J. F. A molecular shuttle. J. Am. Chem. Soc. 113, 5131–5133 (1991).
Trabolsi, A. et al. Radically enhanced molecular recognition. Nature Chem. 2, 42–49 (2010).
Caldwell, S. T. et al. Tuneable pseudorotaxane formation between a biotin–avidin bioconjugate and CBPQT4+. Chem. Commun. 2650–2652 (2008).
Northrop, B. H., Khan, S. J. & Stoddart, J. F. Kinetically controlled self-assembly of pseudorotaxanes on crystallization. Org. Lett. 8, 2159–2162 (2006).
Raymo, F. M. & Stoddart, J. F. Interlocked macromolecules. Chem. Rev. 99, 1643–1663 (1999), and references therein.
Anelli, P. L. et al. Molecular meccano. 1. [2]Rotaxanes and a [2]catenane made to order. J. Am. Chem. Soc. 114, 193–218 (1992).
Klajn, R. et al. Dynamic hook-and-eye nanoparticle sponges. Nature Chem. 1, 733–738 (2009).
Mason, P. E., Parsons, I. W. & Tolley, M. S. The first demonstration of molecular queuing in pseudo[n]polyrotaxanes: a novel variant of supramolecular motion. Angew. Chem. Int. Ed. Engl. 35, 2238–2241 (1996).
Bissell, R. A., Córdova, E., Kaifer, A. E. & Stoddart, J. F. A chemically and electrochemically switchable molecular shuttle. Nature 369, 133–137 (1994).
Green, J. E. et al. A 160-kilobit molecular electronic memory patterned at 1011 bits per square centimetre. Nature 445, 414–417 (2007).
Spruell, J. M. et al. A push-button molecular switch. J. Am. Chem. Soc. 131, 11571–11580 (2009).
Amabilino, D. B., Ashton, P. R., Tolley, M. S., Stoddart, J. F. & Williams, D. J. Isomeric self-assembling [2]catenanes. Angew. Chem. Int. Ed. Engl. 32, 1297–1303 (1993).
Ashton, P. R. et al. Molecular meccano, 51 diastereoselective self-assembly of [2]catenanes. Eur. J. Org. Chem. 995–1004 (1999).
Asakawa, M. et al. Cyclobis(paraquat-4,4′-biphenylene)—an organic molecular square. Chem. Eur. J. 2, 877–893 (1996).
Amabilino, D. B. et al. Molecular meccano. 2. Self-assembly of [n]catenanes. J. Am. Chem. Soc. 117, 1271–1293 (1995).
Ashton, P. R. et al. The template-directed synthesis of cyclobis(paraquat-4,4′-biphenylene). Chem. Commun. 487–490 (1996).
Ashton, P. R. et al. Molecular meccano, 57 template-directed syntheses, spectroscopic properties and electrochemical behavior of [n]catenanes. Eur. J. Org. Chem. 1121–1130 (2000).
Balzani, B. et al. Spectroscopic and electrochemical properties of catenanes containing the 2,7-diazapyrenium units. Supramol. Chem. 13, 303–311 (2001).
Ashton, P. R., Lluïsa, P.-G., Stoddart, J. F., White, A. J. P. & Williams, D. J. Controlling translational isomerism in [2]catenanes. Angew. Chem. Int. Ed. Engl. 34, 571–574 (1995).
Cantrill, S. J., Pease, A. R. & Stoddart, J. F. A molecular meccano kit. J. Chem. Soc., Dalton Trans. 3715–3734 (2000).
Ashton, P. R. et al. Pseudorotaxanes formed between secondary dialkylammonium salts and crown ethers. Chem. Eur. J. 2, 709–728 (1996).
Ashton, P. R. et al. An interwoven supramolecular cage. Angew. Chem. Int. Ed. Engl. 36, 59–62 (1997).
Ashton, P. R. et al. Combining different hydrogen-bonding motifs to self-assemble interwoven superstructures. Chem. Eur. J. 4, 577–589 (1998).
Cantrill, S. J. et al. Molecular meccano, part 60, the influence of macrocyclic polyether constitution upon ammonium ion/crown ether recognition processes. Chem. Eur. J. 6, 2274–2287 (2000).
Cantrill, S. J. et al. Tribenzo[27]crown-9: a new ring for dibenzylammonium rods. Org. Lett. 2, 61–64 (2000).
Tiburcio, J., Davidson, G. J. E. & Loeb, S. J. Pseudo-polyrotaxanes based on a protonated version of the 1,2-bis(4,4′-bipyridinium)ethane-24-crown-8 ether motif. Chem. Commun. 1282–1283 (2002).
Kim, K. Mechanically interlocked molecules incorporating cucurbituril and their supramolecular assemblies. Chem. Soc. Rev. 31, 96–107 (2002), and references therein.
Yoon, J., Kim, S. K., Singh, N. J. & Kim, K. S. Imidazolium receptors for the recognition of anions. Chem. Soc. Rev. 35, 355–360 (2006), and references therein.
Caltagirone, C. & Gale, P. A. Anion receptor chemistry: highlights from 2007. Chem. Soc. Rev. 38, 520–563 (2009), and references therein.
Alcalde, E., Mesquida, N., Vilaseca, M., Alvarez-Rúa, C. & Garcia-Granda, S. Imidazolium-based dicationic cyclophanes. Solid-state aggregates with unconventional (C–H)+···Cl− hydrogen bonding revealed by X-ray diffraction. Supramol. Chem. 19, 501–509 (2007).
Morris, C. & Coll, H. Determination of Molecular Weight 15–44 (John Wiley & Sons, 1989).
Acknowledgements
The authors are grateful to the National Science Foundation (grant no. CHE 0749571 to J.L.S. and grant no. 0741973 for the X-ray diffractometer) and the Robert A. Welch Foundation (grant F-1018 to J.L.S.) for financial support. Thanks also go to S. Sorey, C. Bielawski, A. Tennyson and K. Keller for their assistance with the NMR spectral, VPO and mass spectrometric studies.
Author information
Authors and Affiliations
Contributions
J.L.S. and H.-Y.G. conceived this project and designed the experiments. H.-Y.G. contributed to the experimental work. V.M.L. contributed to the crystallographic study. H.-Y.G., B.M.R and E.K. contributed to the data analysis. H.-Y.G., B.M.R., E.K. and J.L.S. co-wrote the paper.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Supplementary information
Supplementary information
Supplementary information (PDF 2976 kb)
Supplementary information
Crystallographic data for compound 1·4PF6·2CH3CN (CIF 21 kb)
Supplementary information
Crystallographic data for compound 1·4PF6·CH3CN (CIF 32 kb)
Supplementary information
Crystallographic data for compound 1·mono-terephthalate anion·3PF6·4H2O (CIF 22 kb)
Rights and permissions
About this article
Cite this article
Gong, HY., Rambo, B., Karnas, E. et al. A ‘Texas-sized’ molecular box that forms an anion-induced supramolecular necklace. Nature Chem 2, 406–409 (2010). https://doi.org/10.1038/nchem.597
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/nchem.597
- Springer Nature Limited
This article is cited by
-
Ring-through-ring molecular shuttling in a saturated [3]rotaxane
Nature Chemistry (2018)
-
Coronarenes: recent advances and perspectives on macrocyclic and supramolecular chemistry
Science China Chemistry (2018)
-
A simple fluorescent probe for detecting mercury(II) ion in aqueous solution and on agar gels
Journal of the Iranian Chemical Society (2017)
-
A molecular shuttle that operates inside a metal–organic framework
Nature Chemistry (2015)
-
A protein engineered to bind uranyl selectively and with femtomolar affinity
Nature Chemistry (2014)