Abstract
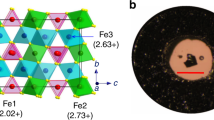
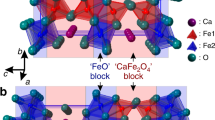
Phase transitions that occur in materials, driven, for instance, by changes in temperature or pressure, can dramatically change the materials’ properties. Discovering new types of transitions and understanding their mechanisms is important not only from a fundamental perspective, but also for practical applications. Here we investigate a recently discovered Fe4O5 that adopts an orthorhombic CaFe3O5-type crystal structure that features linear chains of Fe ions. On cooling below ∼150 K, Fe4O5 undergoes an unusual charge-ordering transition that involves competing dimeric and trimeric ordering within the chains of Fe ions. This transition is concurrent with a significant increase in electrical resistivity. Magnetic-susceptibility measurements and neutron diffraction establish the formation of a collinear antiferromagnetic order above room temperature and a spin canting at 85 K that gives rise to spontaneous magnetization. We discuss possible mechanisms of this transition and compare it with the trimeronic charge ordering observed in magnetite below the Verwey transition temperature.





Similar content being viewed by others
References
Verwey, E. J. W. Electronic conduction of magnetite (Fe3O4) and its transition point at low temperatures. Nature 144, 327–328 (1939).
Senn, M. S., Wright, J. P. & Attfield, J. P. Charge order and three-site distortions in the Verwey structure of magnetite. Nature 481, 173–176 (2012).
Peierls, R. E. Quantum Theory of Solids (Oxford Univ. Press, 1955).
Mott, N. F. Metal–insulator transition. Rev. Mod. Phys. 40, 677–683 (1968).
Mott, N. F. Metal–Insulator Transitions (Taylor and Francis Ltd, 1974).
Imada, M., Fujimori, A. & Tokura, Y. Metal insulator transitions. Rev. Mod. Phys. 70, 1039–1263 (1998).
Matsuoka, T. & Shimizu, K. Direct observation of a pressure-induced metal-to-semiconductor transition in lithium. Nature 458, 186–189 (2009).
Ma, Y. et al. Transparent dense sodium. Nature 458, 182–185 (2009).
Limelette, P. et al. Universality and critical behavior at the Mott transition. Science 302, 89–92 (2003).
Qazilbash, M. M. et al. Mott transition in VO2 revealed by infrared spectroscopy and nano-imaging. Science 318, 1750–1753 (2007).
Nakano, M. et al. Collective bulk carrier delocalization driven by electrostatic surface charge accumulation. Nature 487, 459–462 (2009).
Driscoll, T. et al. Memory metamaterials. Science 325, 1518–1521 (2009).
Lupi, S. et al. A microscopic view on the Mott transition in chromium-doped V2O3 . Nature Commun. 1, 105 (2010).
Budai, J. D. et al. Metallization of vanadium dioxide driven by large phonon entropy. Nature 515, 535–539 (2014).
Morrison, V. R. et al. A photoinduced metal-like phase of monoclinic VO2 revealed by ultrafast electron diffraction. Science 346, 445–448 (2014).
de Jong, S. et al. Speed limit of the insulator–metal transition in magnetite. Nature Mater. 12, 882–886 (2013).
Neaton, J. B. & Ashcroft, N. W. Pairing in dense lithium. Nature 400, 141–144 (1999).
Neaton, J. B. & Ashcroft, N. W. On the constitution of sodium at higher densities. Phys. Rev. Lett. 86, 2830–2833 (2001).
Lavina, B. et al. Discovery of the recoverable high-pressure iron oxide Fe4O5 . Proc. Natl Acad. Sci. USA 108, 17281–17285 (2011).
Ovsyannikov, S. V. et al. Perovskite-like Mn2O3: a path to new manganites. Angew. Chem. Int. Ed. 52, 1494–1498 (2013).
Ovsyannikov, S. V. et al. A hard oxide semiconductor with a direct and narrow bandgap and switchable p–n electrical conduction. Adv. Mater. 26, 8185–8191 (2014).
Gerardin, R., Millon, E., Brice, J. F., Evrard, O. & Le Caer, G. Transfert lectronique d'intervalence entre sites inequivalants: etude de CaFe3O5 par spestrometrie Müssbauer. J. Phys. Chem. Solids 46, 1163–1171 (1985).
Brese, N. E. & O'Keeffe, M. Bond-valence parameters for solids. Acta Cryst. B 47, 192–197 (1991).
Shchennikov, V. V., Ovsyannikov, S. V., Karkin, A. E., Todo, S. & Uwatoko, Y. Galvanomagnetic properties of fast neutron bombarded Fe3O4 magnetite: a case against charge ordering mechanism of the Verwey transition. Solid State Commun. 149, 759–762 (2009).
Morris, E. R. & Williams, Q. Electrical resistivity of Fe3O4 to 48 GPa compression-induced changes in electron hopping at mantle pressures. J. Geophys. Res. 102, 139–148 (1997).
Morin, F. J. Electrical properties of α-Fe2O3 . Phys. Rev. 93, 1195–1199 (1954).
Ovsyannikov, S. V., Morozova, N. V., Karkin, A. E. & Shchennikov, V. V. High-pressure cycling of hematite α-Fe2O3: nanostructuring, in situ electronic transport, and possible charge disproportionation. Phys. Rev. B 86, 205131 (2012).
Dedkov, Y. S., Rüdiger, U. & Güntherodt, G. Evidence for the half-metallic ferromagnetic state of Fe3O4 by spin-resolved photoelectron spectroscopy. Phys. Rev. B 65, 064417 (2002).
Fonin, M., Dedkov, Y. S., Pentcheva, R., Rüdiger, U. & Güntherodt, G. Magnetite: a search for the half-metallic state. J. Phys. Condens. Matter 19, 315217 (2007).
Müller, G. M. et al. Spin polarization in half-metals probed by femtosecond spin excitation. Nature Mater. 8, 56–61 (2009).
Chainani, A., Yokoya, T., Morimoto, T., Takahashi, T. & Todo, S. High-resolution photoemission spectroscopy of the Verwey transition in Fe3O4 . Phys. Rev. B 51, 17976–17979 (1995).
Stokes, H. T., Campbell, B. J. & van Smaalen, S. Generation of (3 + d)-dimensional superspace groups for describing the symmetry of modulated crystalline structures. Acta Crystallogr. A 67, 45–55 (2011).
Wright, J. P., Bell, A. M. T. & Attfield, J. P. Variable temperature powder neutron diffraction study of the Verwey transition in magnetite Fe3O4 . Solid State Sci. 2, 747–753 (2000).
Long, Y. W. et al. Temperature-induced A–B intersite charge transfer in an A-site-ordered LaCu3Fe4O12 perovskite. Nature 458, 60–63 (2009).
Morin, F. J. Magnetic susceptibility of α-Fe2O3 and α-Fe2O3 with added titanium. Phys. Rev. 78, 819–820 (1950).
Chatterji, T. & Hansen, T. C. Magnetoelastic effects in Jahn–Teller distorted CrF2 and CuF2 studied by neutron powder diffraction. J. Phys. Condens. Matter 23, 276007 (2011).
Vecchini, C. et al. Structural distortions in the spin-gap regime of the quantum antiferromagnet SrCu2(BO3)2 . J. Solid State Chem. 182, 3275–3281 (2009).
Walz, F. The Verwey transition—a topical review. J. Phys. Condens. Matter 14, R285–R340 (2002).
Angst, M. et al. Incommensurate charge order phase in Fe2OBO3 due to geometrical frustration. Phys. Rev. Lett. 99, 256402 (2007).
Frost, D. J. et al. A new large-volume multianvil system. Phys. Earth Planet. Inter. 143–144, 507–514 (2004).
Woodland, A. B., Frost, D. J., Trots, D. M., Klimm, K. & Mezouar, M. In situ observation of the breakdown of magnetite (Fe3O4) to Fe4O5 and hematite at high pressures and temperatures. Am. Mineral. 97, 1808–1811 (2012).
Kothapalli, K. et al. Nuclear forward scattering and first-principles studies of the iron oxide phase Fe4O5 . Phys. Rev. B 90, 024430 (2014).
Woodland, A. B. et al. Fe4O5 and its solid solutions in several simple systems. Contrib. Miner. Petrol. 166, 1677–1686 (2013).
Ishii, T. et al. High-pressure phase transitions in FeCr2O4 and structure analysis of new post-spinel FeCr2O4 and Fe2Cr2O5 phases with meteoritical and petrological implications. Am. Mineral. 99, 1788–1797 (2014).
Guignard, J. & Crichton, W. A. Synthesis and recovery of bulk Fe4O5 from magnetite, Fe3O4. A member of a self-similar series of structures for the lower mantle and transition zone. Mineral. Mag. 78, 361–371 (2014).
Verbeeck, J. & Van Aert, S. Model based quantification of EELS spectra. Ultramicroscopy 101, 207–224 (2004).
Palatinus, L. & Chapuis, G. SUPERFLIP—a computer program for the solution of crystal structures by charge flipping in arbitrary dimensions. J. Appl. Crystallogr. 40, 786–790 (2007).
Petricek, V., Dusek, M. & Palatinus, L. Crystallographic computing system JANA2006: general features. Z. Kristallogr. 229, 345–352 (2014).
Aksenov, V. L. et al. DN-12 time of flight high pressure neutron spectrometer for investigation of microsamples. Physica B 265, 258–262 (1999).
Rodriguez-Carvajal, J. Recent advances in magnetic structure determination by neutron powder diffraction. Physica B 192, 55–69 (1993).
Acknowledgements
S.V.O. acknowledges the financial support of the Deutsche Forschungsgemeinschaft (DFG) under project OV-110/1-3. A.E.K. and V.V.S. acknowledge the support of the Russian Foundation for Basic Research (Project 14–02–00622a). H.G. acknowledges the support from the Alexander von Humboldt (AvH) Foundation and the National Natural Science Foundation of China (No. 51201148). A.M.A., R.E. and J.V. acknowledge financial support from the European Commission (EC) under the Seventh Framework Programme (FP7) under a contract for an Integrated Infrastructure Initiative, Reference No. 312483-ESTEEM2. R.E. acknowledges support from the EC under FP7 Grant No. 246102 IFOX. A.M.A. acknowledges funding from the Russian Science Foundation (Grant No. 14-13-00680). A.A.T. acknowledges funding and from the Federal Ministry for Education and Research through the Sofja Kovalevkaya Award of the AvH Foundation. Funding from the Fund for Scientific Research Flanders under FWO Project G.0044.13N is acknowledged. M.B. and S.v.S. acknowledge support from the DFG under Project Sm55/15-2. We acknowledge the European Synchrotron Radiation Facility for the provision of synchrotron radiation facilities.
Author information
Authors and Affiliations
Contributions
S.V.O. synthesized and characterized the Fe4O5 samples. M.B., E.B., S.v.S., V.D. and D.C. performed the single-crystal X-ray diffraction study at low temperatures. M.B. and S.v.S. resolved the structure of the new low-temperature phase. S.E.K. performed the neutron-diffraction measurements. D.P.K. analysed the neutron-diffraction data and derived magnetic-structure models. A.A.T. measured the magnetic properties. A.E.K. and V.V.S. measured the electronic-transport properties. H.G. synthesized the samples and discussed the results. A.M.A., R.E. and J.V. collected and analysed the EELS spectra. D.P.K., A.M.A., A.A.T and C.M. discussed the magnetic properties and contributed to writing the manuscript. S.V.O. wrote a first draft of the manuscript, and all the co-authors read, revised and commented on it. S.V.O. and L.S.D. initiated and designed the research.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Supplementary information
Supplementary information
Supplementary information (PDF 3052 kb)
Supplementary information
Crystallographic data for the incommensurate structure (CIF 172 kb)
Supplementary information
Crystallographic data for the commensurate approximation (CIF 39 kb)
Rights and permissions
About this article
Cite this article
Ovsyannikov, S., Bykov, M., Bykova, E. et al. Charge-ordering transition in iron oxide Fe4O5 involving competing dimer and trimer formation. Nature Chem 8, 501–508 (2016). https://doi.org/10.1038/nchem.2478
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/nchem.2478
- Springer Nature Limited
This article is cited by
-
The electrical conductivity of Fe4O5, Fe5O6, and Fe7O9 up to 60 GPa
Physics and Chemistry of Minerals (2022)
-
Magnetic sources in the Earth’s mantle
Nature Reviews Earth & Environment (2020)
-
Single phase charge ordered stoichiometric CaFe3O5 with commensurate and incommensurate trimeron ordering
Nature Communications (2019)
-
Magnetism in cold subducting slabs at mantle transition zone depths
Nature (2019)
-
Pressure tuning of charge ordering in iron oxide
Nature Communications (2018)