Abstract
Replying to: Frank, C. G., Sanyal, S., Rush, J. S., Waechter C. J. & Menon A. K. Nature 454, 10.1038/nature07165 (2008)
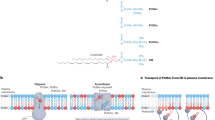
The controlled translocation of polar phospholipid head groups between the two leaflets of biological membranes is thought to be meditated by proteins called flippases. Several candidate polyisoprene phosphate–oligosaccharide flippases have been described1,2,3, including Rft1 for Dol-PP-GlcNAc2Man5 by us4 and now questioned by Frank et al.5.
Similar content being viewed by others
References
Alaimo, C. et al. Two distinct but interchangeable mechanisms for flipping of lipid-linked oligosaccharides. EMBO J. 25, 967–976 (2006)
Raetz, C. R. & Whitfield, C. Lipopolysaccharide endotoxins. Annu. Rev. Biochem. 71, 635–700 (2002)
Yan, A., Guan, Z. & Raetz, C. R. An undecaprenyl phosphate-aminoarabinose flippase required for polymyxin resistance in Escherichia coli . J. Biol. Chem. 282, 36077–36089 (2007)
Helenius, J. et al. Translocation of lipid-linked oligosaccharides across the ER membrane requires Rft1 protein. Nature 415, 447–450 (2002)
Frank, C. G., Sanyal, S., Rush, J. S., Waechter, C. J. & Menon, A. K. Does Rft1 flip an N-glycan lipid precursor? Nature 454 10.1038/nature07165 (2008)
Pomorski, T. & Menon, A. K. Lipid flippases and their biological functions. Cell. Mol. Life Sci. 63, 2908–2921 (2006)
Rick, P. D. et al. Evidence that the wzxE gene of Escherichia coli K-12 encodes a protein involved in the transbilayer movement of a trisaccharide-lipid intermediate in the assembly of enterobacterial common antigen. J. Biol. Chem. 278, 16534–16542 (2003)
Kol, M. A. et al. Translocation of phospholipids is facilitated by a subset of membrane-spanning proteins of the bacterial cytoplasmic membrane. J. Biol. Chem. 278, 24586–24593 (2003)
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Helenius, J., Ng, D., Marolda, C. et al. Helenius et al. reply. Nature 454, E4–E5 (2008). https://doi.org/10.1038/nature07164
Issue Date:
DOI: https://doi.org/10.1038/nature07164
- Springer Nature Limited