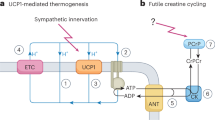
Abstract
Energy balance to prevent the development of obesity is dependent on energy expenditure. Although physical activity is the dominant mechanism for dissipating excess energy, a system of thermogenesis that evolved to protect the body from hypothermia is based upon the uncoupling of oxidative phosphorylation in brown adipocytes by the mitochondrial uncoupling protein (UCP1). It has been shown that upregulation of UCP1 by genetic manipulations or pharmacological agents can reduce obesity and improve insulin sensitivity. Recent evidence has shown the existence of two sources for brown adipocytes, one appearing as discrete brown fat depots during fetal development and the other appears during post-natal development as diffuse populations in traditional white fat depots. The latter can be induced by adrenergic stimulation depending on the genetic background of the animals and the nutritional environment. Understanding the biological and environmental factors controlling the expression of these two brown adipocyte populations promises to provide new strategies by which enhanced thermogenesis can be used to reduce obesity.


Similar content being viewed by others
References
Schwartz MW, Woods SC, Seeley RJ, Barsh GS, Baskin DG, Leibel RL . Is the energy homeostasis system inherently biased toward weight gain? Diabetes 2003; 52: 232–238.
Rothwell NJ, Stock MJ . A role for brown adipose tissue in diet-induced thermogenesis. Nature 1979; 281: 31–35.
Cannon B, Nedergaard J . Brown adipose tissue: function and physiological significance. Physiol Rev 2004; 84: 277–359.
Trayhurn P, Thurlby PL, James WPT . Thermogenic defect in pre-obese ob/ob mice. Nature 1977; 266: 60–62.
Himms-Hagen J, Desautels M . A mitochondrial defect in brown adipose tissue of the obese (ob/ob) mouse: reduced binding of purine nucleotides and a failure to respond to cold by an increase in binding. Biochem Biophys Res Commun 1978; 83: 628–634.
Liu X, Rossmeisl M, McClaine J, Riachi M, Harper ME, Kozak LP . Paradoxical resistance to diet-induced obesity in UCP1-deficient mice. J Clin Invest 2003; 111: 399–407.
Coulter AA, Bearden CM, Liu X, Koza RA, Kozak LP . Dietary fat interacts with QTLs controlling induction of Pgc-1 alpha and Ucp1 during conversion of white to brown fat. Physiol Genomics 2003; 14: 139–147.
Xue B, Rim JS, Hogan JC, Coulter AA, Koza RA, Kozak LP . Genetic variability affects the development of brown adipocytes in white fat but not in interscapular brown fat. J Lipid Res 2007; 48: 41–51.
Ukropec J, Anunciado RP, Ravussin Y, Hulver MW, Kozak LP . UCP1-independent thermogenesis in white adipose tissue of cold-acclimated Ucp1−/− mice. J Biol Chem 2006; 281: 31894–31908.
Hofmann WE, Liu X, Bearden CM, Harper M-E, Kozak LP . Effects of genetic background on thermoregulation and fatty acid-induced uncoupling of mitochondria in UCP1-deficient mice. J Biol Chem 2001; 276: 12460–12465.
Ukropec J, Anunciado RV, Ravussin Y, Kozak LP . Leptin is required for uncoupling protein-1-independent thermogenesis during cold stress. Endocrinology 2006; 147: 2468–2480.
Hirsch J . Obesity. Some heat but not enough light. Nature 1997; 387: 27–28.
Kontani Y, Wang Y, Kimura K, Inokuma KI, Saito M, Suzuki-Miura T et al. UCP1 deficiency increases susceptibility to diet-induced obesity with age. Aging Cell 2005; 4: 147–155.
Brown LJ, Koza RA, Everett C, Reitman ML, Marshall L, Fahien LA et al. Normal thyroid thermogenesis but reduced viability and adiposity in mice lacking the mitochondrial glycerol phosphate dehydrogenase. J Biol Chem 2002; 277: 32892–32898.
Lardy H, Su CY, Kneer N, Wielgus S . Dehydroepiandosterone induces enzymes that permit thermogenesis and decrease metabolic efficiency. In: Lardy H, Stratman F (eds). Hormones, thermogenesis, and obesity. Elsevier: New York, 1989, pp 415–426.
Kopecky J, Clarke G, Enerback S, Spiegelman B, Kozak LP . Expression of the mitochondrial uncoupling protein gene from the aP2 gene promoter prevents genetic obesity. J Clin Invest 1995; 96: 2914–2923.
Kopecky J, Hodny Z, Rossmeisl M, Syrovy I, Kozak LP . Reduction of dietary obesity in aP2-Ucp transgenic mice: physiology and adipose tissue distribution. Am J Physiol 1996; 270: E768–E775.
Clapham JC, Arch JR, Chapman H, Haynes A, Lister C, Moore GB et al. Mice overexpressing human uncoupling protein-3 in skeletal muscle are hyperphagic and lean. Nature 2000; 406: 415–418.
Soloveva V, Graves RA, Rasenick MM, Spiegelman BM, Ross SR . Transgenic mice overexpressing the beta 1-adrenergic receptor in adipose tissue are resistant to obesity. Mol Endocrinol 1997; 11: 27–38.
Cummings DE, Brandon EP, Planas JV, Motamed K, Idzerda RL, McKnight GS . Genetically lean mice result from targeted disruption of the RII beta subunit of protein kinase A. Nature 1996; 382: 622–626.
Cederberg A, Gronning LM, Ahren B, Tasken P, Carlsson P, Enerback S . FOXC2 is a winged helix gene that counteracts obesity, hypertriglceridemia, and diet-induced insulin resistance. Cell 2001; 106: 563–573.
Rosen ED, Walkey CJ, Puigserver P, Spiegelman BM . Transcriptional regulation of adipogenesis. Genes Dev 2000; 14: 1293–1307.
Tontonoz P, Hu E, Spiegelman BM . Stimulation of adipogenesis in fibroblasts by PPARg2, a lipid-activated transcription factor. Cell 1994; 79: 1147–1156.
Sears IB, MacGinnitie MA, Kovacs LG, Graves RA . Differentiation-dependent expression of the brown adipocyte uncoupling protein gene: regulation by peroxisome proliferator-activated receptor gamma. Mol Cell Biol 1996; 16: 3410–3419.
Puigserver P, Wu Z, Park CW, Graves R, Wright M, Spiegelman BM . A cold-inducible coactivator of nuclear receptors linked to adaptive thermogenesis. Cell 1998; 92: 829–839.
Kozak UC, Kopecky J, Teisinger J, Enerback S, Boyer B, Kozak LP . An upstream enhancer regulating brown-fat-specific expression of the mitochondrial uncoupling protein gene. Mol Cell Biol 1994; 14: 59–67.
Cao W, Daniel KW, Robidoux J, Puigserver P, Medvedev AV, Bai X et al. p38 mitogen-activated protein kinase is the central regulator of cyclic AMP-dependent transcription of the brown fat uncoupling protein 1 gene. Mol Cell Biol 2004; 24: 3057–3067.
Graves RA, Tontonoz P, Spiegelman BM . Analysis of a tissue-specific enhancer: ARF6 regulates adipogenic gene expression. Mol Cell Biol 1992; 12: 1202–1208.
Barak Y, Nelson MC, Ong ES, Jones YZ, Ruiz-Lozano P, Chien KR et al. PPAR gamma is required for placental, cardiac, and adipose tissue development. Mol Cell 1999; 4: 585–595.
Linhart HG, Ishimura-Oka K, DeMayo F, Kibe T, Repka D, Poindexter B et al. C/EBPalpha is required for differentiation of white, but not brown, adipose tissue. Proc Natl Acad Sci USA 2001; 98: 12532–12537.
Tanaka T, Yoshida N, Kishimoto T, Akira S . Defective adipocyte differentiation in mice lacking the C/EBPβ and C/EPBd gene. EMBO J 1997; 16: 7432–7443.
Cassard-Doulcier AM, Larose M, Matamala JC, Champigny O, Bouillaud F, Ricquier D . In vitro interactions between nuclear proteins and uncoupling protein gene promoter reveal several putative transactivating factors including Ets1, retinoid X receptor, thyroid hormone receptor, and a CACCC box-binding protein. J Biol Chem 1994; 269: 24335–24342.
Silva JE . The multiple contributions of thyroid hormone to heat production. J Clin Invest 2001; 108: 35–37.
Hansen JB, Jorgensen C, Petersen RK, Hallenborg P, De Matteis R, Boye HA et al. Retinoblastoma protein functions as a molecular switch determining white versus brown adipocyte differentiation. Proc Natl Acad Sci USA 2004; 101: 4112–4117.
Seale P, Kajimura S, Yang W, Chin S, Rohas LM, Uldry M et al. Transcriptional control of brown fat determination by PRDM16. Cell Metab 2007; 6: 38–54.
Guerra C, Koza RA, Yamashita H, Walsh K, Kozak LP . Emergence of brown adipocytes in white fat in mice is under genetic control. Effects on body weight and adiposity. J Clin Invest 1998; 102: 412–420.
Koza RA, Hohmann SM, Guerra C, Rossmeisl M, Kozak LP . Synergistic gene interactions control the induction of the mitochondrial uncoupling protein (Ucp1) gene in white fat tissue. J Biol Chem 2000; 275: 34486–34492.
Xue B, Coulter A, Rim JS, Koza RA, Kozak LP . Transcriptional synergy and the regulation of Ucp1 during brown adipocyte induction in white fat depots. Mol Cell Biol 2005; 25: 8311–8322.
Xue B, Rim JS, Hogan JC, Coulter AA, Koza RA, Kozak LP . Genetic variability affects the development of brown adipocytes in white fat but not in interscapular brown fat. J Lipid Res 2007; 48/1: 41–51.
Timmons JA, Wennmalm K, Larsson O, Walden TB, Lassmann T, Petrovic N et al. Myogenic gene expression signature establishes that brown and white adipocytes originate from distinct cell lineages. Proc Natl Acad Sci USA 2007; 104: 4401–4406.
Acknowledgements
Starting with Anders Jacobsson, a graduate student from the Wenner-Gren Institute in Stockholm, who came to my laboratory in 1983 with an antibody against UCP1 to work on cloning of Ucp1 cDNA, I wish to thank the large number of post-doctoral associates and collaborators who have worked with me defining the structure and function of the Ucp1 gene and its role in thermogenesis and body weight regulation. I also acknowledge the financial support through R01 HD08431 from the NIH throughout this period.
Author information
Authors and Affiliations
Corresponding author
Additional information
Conflict of interest
The authors have declared no financial interests.
Rights and permissions
About this article
Cite this article
Kozak, L., Anunciado-Koza, R. UCP1: its involvement and utility in obesity. Int J Obes 32 (Suppl 7), S32–S38 (2008). https://doi.org/10.1038/ijo.2008.236
Published:
Issue Date:
DOI: https://doi.org/10.1038/ijo.2008.236
- Springer Nature Limited
Keywords
This article is cited by
-
Maternal thyroid hormone receptor β activation in mice sparks brown fat thermogenesis in the offspring
Nature Communications (2023)
-
Maternal helminth infection protects offspring from high-fat-diet-induced obesity through altered microbiota and SCFAs
Cellular & Molecular Immunology (2023)
-
Increased adipose catecholamine levels and protection from obesity with loss of Allograft Inflammatory Factor-1
Nature Communications (2023)
-
Effects of caffeoylquinic acid analogs derived from aerial parts of Artemisia iwayomogi on adipogenesis
Food Science and Biotechnology (2023)
-
Genetic advancements in obesity management and CRISPR–Cas9-based gene editing system
Molecular and Cellular Biochemistry (2023)