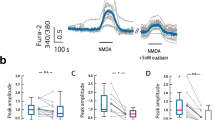
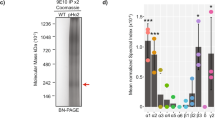
Abstract
The mechanisms by which neurotransmitter receptors are immobilized at postsynaptic sites in neurons are largely unknown. The activity of NMDA (N-methyl-D-aspartate) receptors is mechanosensitive1 and dependent on the integrity of actin2, suggesting a functionally important interaction between NMDA receptors and the postsynaptic cytoskeleton. α-Actinin-2, a member of the spectrin/dystrophin family of actin-binding proteins, is identified here as a brain postsynaptic density protein that colocalizes in dendritic spines with NMDA receptors and the putative NMDA receptor-clustering molecule PSD-95. α-Actinin-2 binds by its central rod domain to the cytoplasmic tail of both NR1 and NR2B subunits of the NMDA receptor, and can be immunoprecipitated with NMDA receptors and PSD-95 from rat brain. Intriguingly, NR1-α-actinin binding is directly antagonized by Ca2+/calmodulin. Thus α-actinin may play a role in both the localization of NMDA receptors and their modulation by Ca2+.
Similar content being viewed by others
References
Paoletti, P. & Ascher, P. Neuron 13, 645–655 (1994).
Rosenmund, C. & Westbrook, G. L. Neuron 10, 805–814 (1993).
Ishii, T. et al. J. Biol. Chem. 268, 2836–2843 (1992).
Meguro, H. et al. Nature 357, 70–73 (1992).
Monyer, H. et al. Science 256, 1217–1221 (1992).
Kutsuwada, T. et al. Nature 358, 36–41 (1993).
Monyer, H., Burnashev, N., Laurie, D. J., Sakmann, B. & Seeburg, P. H. Neuron 12, 529–540 (1994).
Sheng, M., Cummings, J., Roldan, L. A., Jan, Y. N. & Jan, L. Y. Nature 368, 144–147 (1994).
Kornau, H.-C., Schenker, L. T., Kennedy, M. B. & Seeburg, P. H. Science 269, 1737–1740 (1995).
Niethammer, M., Kim, E. & Sheng, M. J. Neurosci. 16, 2157–2163 (1996).
Ehlers, M. D., Tingley, W. G. & Huganir, R. L. Science 269, 1734–1737 (1995).
Sugihara, H., Moriyoshi, K., Ishii, T., Masu, M. & Nakanishi, S. Biochem. Biophys. Res. Commun. 185, 826–832 (1992).
Beggs, A. H. et al. J. Biol. Chem. 267, 9281–9288 (1992).
Bennett, V. Physiol. Rev. 70, 1029–1065 (1990).
Blanchard, A., Ohanian, V. & Critchley, D. J. Muscle Res. Cell Motil. 10, 280–289 (1989).
Hollmann, M. et al. Neuron 10, 943–954 (1993).
Kim, E., Cho, K.-O., Rothschild, A. & Sheng, M. Neuron 17, 103–113 (1996).
Müller, B. M. et al. J. Neurosci. 15, 2354–2366 (1995).
Miller, S. G. & Kennedy, M. B. J. Biol. Chem. 260, 9039–9046 (1985).
Ehlers, M. D., Zhang, S., Bernhardt, J. P. & Huganir, R. L. Cell 84, 745–755 (1996).
Kim, E., Niethammer, M., Rothschild, A., Jan, Y. N. & Sheng, M. Nature 378, 85–88 (1995).
Li, M., Jan, Y. N. & Jan, L. Y. Science 257, 1225–1230 (1992).
Müller, B. M. et al. Neuron 17, 255–265 (1996).
Banker, G. A. & Cowan, W. M. Brain Res. 126, 397–425 (1977).
Moriyoshi, K. et al. Nature 354, 31–37 (1991).
Cho, K.-O., Hunt, C. A. & Kennedy, M. B. Neuron 9, 929–942 (1992).
Moon, I. S., Apperson, M. L. & Kennedy, M. B. Proc. Natl Acad. Sci. USA 91, 3954–3958 (1994).
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Wyszynski, M., Lin, J., Rao, A. et al. Competitive binding of α-actinin and calmodulin to the NMDA receptor. Nature 385, 439–442 (1997). https://doi.org/10.1038/385439a0
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1038/385439a0
- Springer Nature Limited
This article is cited by
-
Synaptic-like transmission between neural axons and arteriolar smooth muscle cells drives cerebral neurovascular coupling
Nature Neuroscience (2024)
-
Excitation–transcription coupling, neuronal gene expression and synaptic plasticity
Nature Reviews Neuroscience (2023)
-
Schizophrenia-associated SAP97 mutations increase glutamatergic synapse strength in the dentate gyrus and impair contextual episodic memory in rats
Nature Communications (2022)
-
Adult re-expression of IRSp53 rescues NMDA receptor function and social behavior in IRSp53-mutant mice
Communications Biology (2022)
-
Inhibiting the β1integrin subunit increases the strain threshold for neuronal dysfunction under tensile loading in collagen gels mimicking innervated ligaments
Biomechanics and Modeling in Mechanobiology (2022)