Abstract
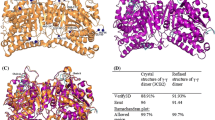
TUBULIN, the major component of microtubules, is a heterodimer of two chains, α and β1, both of relative molecular mass 50,000 (Mr50K) and with 40-50% identity. The isotypic variety2 and conformational flexibility of tubulin have so far made it impossible to obtain crystals for X-ray work3. Structural knowledge of tubulin has been limited to about 20 Å from X-ray diffraction of oriented microtubules4, and from electron microscopy of microtubules and zinc-induced crystalline sheets in negative stain5,6. The sheets consist of protofilaments similar to those in microtubules but associated in an antiparallel arrangement7, and their two-dimensional character is ideal for high-resolution electron microscopy8,9. Here we present a three-dimensional reconstruction of tubulin to 6.5 Å resolution, obtained by electron crystallography of zinc-induced two-dimensional crystals of the protein. The α- and β-subunits appear topologically similar, in agreement with their sequence homology10. Several features can be defined in terms of secondary structure. An apparent α-helical portion, adjacent to both interdimer and inter-protofilament contacts, is tentatively attributed to a segment near the carboxy terminus of the protein. We can assign the α- and β-subunits on the basis of projection studies of the binding of taxol*, which show one taxol site per tubulin heterodimer, in agreement with the known stoichiometry of taxol in microtubules11. These studies indicate that taxol affects the interaction between protofilaments; to our knowledge, this is the first time that a ligand-binding site has been visualized in the tubulin molecule.
Similar content being viewed by others
References
Ludueña, R. F., Shooter, E. M. & Wilson, L. J. biol. Chem. 252, 7006–7014 (1977).
Ludueña, R. F. Molec. Biol. Cell 44, 445–457 (1993).
Lobert, S. & Correia, J. J. Archs. Biochem. Biophys. 290, 93–102 (1991).
Beese, L., Stubbs, G. & Cohen, C. J. molec. Biol. 194, 257–264 (1987).
Amos, L. A. in Microtubules (eds Roberts, K. & Hyams, J. S.) 1–64 (Academic Press, New York, 1974).
Tamm, L. K., Crepeau, R. H. & Edelstein, S. J. J. molec. Biol. 130, 473–492 (1979).
Amos, L. A. & Baker, T. S. Nature 279, 607–612 (1979).
Downing, K. H. & Jontes, J. J. struct. Biol. 109, 152–159 (1992).
Wolf, S. G., Mosser, G. & Downing, K. H. J. struct. Biol. 111, 190–199 (1994).
Burns, R. G. & Surridge, C. D. in Microtubules (eds Hyams, J. S. & Lloyd, C. W.) 3–32 (Wiley, New York, 1993).
Díaz, J. F. & Andreu, J. M. Biochemistry 32, 2747–2755 (1993).
Kumar, N. J. biol. Chem. 250, 10435–10441 (1981).
Schiff, P. B. & Horwitz, S. B. Proc. natn. Acad. Sci. U.S.A. 77, 1561–1565 (1980).
Rowinsky, E. K., Wright, M., Monsarrat, B., Lesser, G. J. & Donerhower, R. C. Cancer Surv. 17, 283–304 (1993).
Schiff, P. B., Fant, J. & Horwitz, S. B. Nature 277, 665–667 (1979).
Nogales, E., Wolf, S. G., Zhang, S. & Downing, K. H. J. struct. Biol. (in the press).
Banerjee, A., Roach, M. C., Trcka, P. & Ludueña, R. F. J. biol. Chem. 267, 5625–5630 (1992).
Rao, S., Horwitz, S. B. & Ringel, I. J. natn. Cancer Inst. 84, 785–788 (1992).
Combeau, C. et al. Biochemistry 33, 6676–6683 (1994).
Parness, J. & Horowitz, S. B. J. Cell Biol. 91, 479–487 (1981).
Manfredi, J. J., Parness, J. & Horwitz, S. B. J. Cell Biol. 94, 688–696 (1982).
Andreu, J. M. et al. J. molec. Biol. 226, 169–184 (1992).
Díaz, J. F., Menéndez, M. & Andreu, J. M. Biochemistry 32, 10067–10077 (1993).
Howard, W. D. & Timasheff, S. N. J. biol. Chem. 263, 1342–1346 (1988).
Dye, R. B., Fink, S. P. & Williams, R. C. Jr J. biol. Chem. 268, 6847–6850 (1993).
Henderson, R. & Unwin, P. N. Nature 257, 28–32 (1975).
Henderson, R. et al. J. molec. Biol. 213, 899–929 (1990).
Kühlbrandt, W. & Wang, D. N. Nature 350, 130–134 (1991).
Kühlbrandt, W., Wang, D. N. & Fuiyoshi, Y. Nature 367, 614–621 (1994).
Mandelkow, E.-M., Thomas, J. & Cohen, C. Proc. natn. Acad. Sci. U.S.A. 74, 3370–3374 (1977).
Heuser, J. E. J. Cell Biol. 103, 2209–2227 (1986).
Kirchner, K. & Mandelkow, E.-M. EMBO J. 4, 2397–2402 (1985).
Sackett, D. L. & Varma, J. K. Biochemistry 32, 13560–13565 (1993).
Margolis, R., Rauch, C. T. & Wilson, L. Biochemistry 19, 5550–5557 (1980).
Palanivelu, P. & Ludueña, R. F. J. biol. Chem. 257, 6311–6315 (1982).
Rao, S., Krauss, N. E., Heerding, J. M., Orr, G. A. & Horwitz, S. B. J. biol. Chem. 269, 3132–3134 (1994).
Shivanna, B. D., Mejillano, M. R., Williams, T. D. & Himes, R. H. J. biol. Chem. 268, 127–132 (1993).
Schiff, P. B. & Horwitz, S. B. Biochemistry 20, 3247–3252 (1981).
Carlier, M.-F. & Pantaloni, D. Biochemistry 22, 4814–4822 (1983).
Trachtenberg, S. & DeRosier, D. J. J. molec. Biol. 195, 581–601 (1987).
Fellous, A., Francon, J., Lennon, A. M. & Nuñez, J. Eur. J. Biochem. 78, 167–174 (1977).
Wang, D. N. & Kühlbrandt, W. J. molec. Biol. 217, 691–699 (1991).
Downing, K. H. Science 251, 53–59 (1991).
Henderson, R., Baldwin, J. M., Downing, K. H., Lepault, J. & Zemlin, F. Ultramicroscopy 19, 147–178 (1986).
Downing, K. H. Ultramicroscopy 46, 199–206 (1992).
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Nogales, E., Grayer Wolf, S., Khan, I. et al. Structure of tubulin at 6.5 Å and location of the taxol-binding site. Nature 375, 424–427 (1995). https://doi.org/10.1038/375424a0
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1038/375424a0
- Springer Nature Limited
This article is cited by
-
Xanthatin and 8-epi-xanthatin as new potential colchicine binding site inhibitors: a computational study
Journal of Molecular Modeling (2023)
-
Role of inflammation and oxidative stress in chemotherapy-induced neurotoxicity
Immunologic Research (2022)
-
Phosphoproteomic analysis of neoadjuvant breast cancer suggests that increased sensitivity to paclitaxel is driven by CDK4 and filamin A
Nature Communications (2022)
-
The model of local axon homeostasis - explaining the role and regulation of microtubule bundles in axon maintenance and pathology
Neural Development (2019)
-
An overview of tubulin modulators deposited in protein data bank
Medicinal Chemistry Research (2019)