Abstract
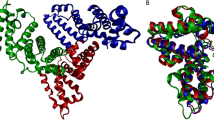
The three-dimensional structure of human serum albumin has been determined crystallographically to a resolution of 2.8 Å. It comprises three homologous domains that assemble to form a heart-shaped molecule. Each domain is a product of two subdomains that possess common structural motifs. The principal regions of ligand binding to human serum albumin are located in hydrophobic cavities in subdomains IIA and IIIA, which exhibit similar chemistry. The structure explains numerous physical phenomena and should provide insight into future pharmacokinetic and genetically engineered therapeutic applications of serum albumin.
Similar content being viewed by others
References
Partridge, W. M. Am. J. Physiol. 252, 157–164 (1987).
Schnitzer, J. E., Carley, W. W. & Palade, G. E. Proc. natn. Acad. Sci. U.S.A. 85, 6773–6777 (1988).
Brown, J. R. & Shockley, P. in Lipid-Protein Interactions Vol. 1 (eds Jost, P. & Griffith, O. H.) 25–68 (Wiley, New York, 1982).
Sargent, T. D., Yang, M. & Bonner, J. Proc. natn. Acad. Sci. U.S.A. 78, 243–246 (1981).
Dugiaczyk, A., Law, S. W. & Dennison, O. E. Proc. natn. Acad. Sci. U.S.A. 79, 71–75 (1982).
Brown, W. M., Dziegielewska, K. M., Foreman, R. C. & Saunders, N. R. Nucleic Acids Res. 17, 10495 (1989).
Moskaitis, J. E., Sargent, T. D., Smith, L. H. Jr, Pastori, R. L. & Scnoenberg, D. R. Molec. Endocr. 3, 464–473 (1989).
Byrnes, L. & Gannon, F. DNA Cell Biol. 9, 647–655 (1990).
Minghetti, P. P., Law, S. W. & Dugaiczyk, A. Molec. biol. Evol. 2, 347–358 (1985).
Weinstock, J. & Baldwin, G. S. Nucleic Acids Res. 16, 9045 (1988).
Gray, J. E. & Doolittle, R. F. Protein Sci. 1, 289–302 (1992).
Moringa, T., Sakai, M., Wegmann, T. G. & Tamaoki, T. Proc. natn. Acad. Sci. U.S.A. 80, 4604–4608 (1983).
Jagdodzinski, L. L., Sargent, T. D., Yang, M., Glackin, C. & Bonner, J. Proc. natn. Acad. Sci. U.S.A. 78, 3521–3525 (1981).
Gorin, M. B. & Hoffman, B. J. Proc. natn. Acad. Sci. U.S.A. 77, 1351–1355 (1980).
Schoentgen, F. Metz-Boutigue, M.-H., Jolles, J., Constants, J. & Lolles, P. Biochim. biophys. Acta 871, 189–198.
Cooke, N. E. & David, E. V. J. clin. Invest. 76, 2420–2424 (1985).
Yang, F. et al. Genomics 7, 509–516 (1990).
Brown, J. R. Fed. Proc. 35, 2141–2144 (1976).
Carter, D. C. et al. Science 244, 1195–1198 (1989).
Carter, D. C. & He, X. M. Science 249, 302–303 (1990).
Putnam, F. W. The Plasma Proteins 2nd edn Vol. 4 (Academic, London, 1984).
Peters, T. Jr Adv. Protein Chem. 37, 161–245 (1985).
Fehske, K. J., Muller, W. E. & Wollert, U. Biochem. Pharmac. 30, 687–692 (1981).
Kragh-Hannsen, U. Pharmac. Rev. 33, 17–53 (1981).
Quirk, A. V. et al. Biotechnol. appl. Biochem. 11, 273–287 (1989).
Luft, A. J. & Lorscheider, F. L. Biochemistry 22, 5978–5980 (1983).
Aoki, K., Sato, K., Nagoaka, S., Kamada, M. & Hiramatsu, K. Biochim. biophys. Acta 328, 323–333 (1973).
Sudlow, G., Birkett, D. J. & Wade, D. N. Molec. Pharmac. 11, 824–832 (1975).
Sudlow, G., Birkett, D. J. & Wade, D. N. Molec. Pharmac. 12, 1052–1061 (1977).
Sollene, N. P. & Means, G. E. Molec. Pharmac. 14, 754–757 (1979).
Ozeki, Y., Kurono, Y., Yotsuyanagi, T. & Ikeda, K. Pharmac. Bull. 28, 535–540 (1980).
Kurono, Y., Ozeki, Y., Yamada, H., Takeuchi, T. & Ikeda, K. Chem. Pharmac. Bull. 35, 734–739 (1987).
Hagag, N., Birnbaum, E. R. & Darnall, D. W. Biochemistry 22, 2420–2427 (1983).
Feldoff, R. C. & Ledden, D. J. Bioch. biophys. Res. Commun. 114, 20–27 (1983).
Takahashi, N. et al. Proc. natn. Acad. Sci. U.S.A. 84, 8001–8005 (1987).
Arai, K., Ishioka, N., Huss, K., Madison, J. & Putnam, F. W. Proc. natn Acad. Sci. U.S.A. 86, 434–438 (1989).
Galliano, M. et al. Proc. natn. Acad. Sci. U.S.A. 87, 8721–8725 (1990).
King, T. P. Arch. biochem. Biophys. 156, 509–520 (1973).
Bos, O. J. M., Remijn, J. P. M., Fischer, J. E., Witling, J. & Janssen, L. H. M. Biochem. Pharmac. 37, 3905–3909 (1988).
Pedersen, S. M. Biochem. Pharmac. 35, 2661–2666 (1987).
Dixon, J. W. & Sarkar, B. J. biol. Chem. 249, 5872–5877 (1974).
Howard, A. J. et al. J. appl. Crystallogr. 20, 383–387 (1987).
Furey, W. & Swaminathan, S. Am. crystallogr. Assoc. Mtg. Abstr. Ser 2 18, 73 (1990).
Wang, B. C. Meth. Enzym. 115, 90–112 (1985).
Jones, T. A. J. appl. Crystallogr. 20, 383 (1987).
Brünger, A. T., Kuriyan, J. & Karplus, M. Science 235, 458–460 (1987).
Rossmann, M. G. & Blow, D. M. Acta Crystallogr. 15, 24–31 (1962).
Crowther, R. H. in The Molecular Replacement Method (ed. Rossmann, M. G.) 173–178 (Gordon & Breach, New York, 1972).
Lattman, E. E. & Love, W. E. Acta Crystallogr. B26, 1854–1857 (1970).
Crowther, R. A. & Blow, D. M. Acta Crystallogr. 23, 544–548 (1967).
Fitzgerald, P. M. D. J. appl. Crystallogr. 21, 273–278 (1988).
Carson, M. J. molec. Graphics 5, 103–106 (1987).
Cambillau, C. & Horjales, E. J. molec. Graphics 5, 174 (1987).
Walker, J. E. FEBS Lett. 66, 173–175 (1976).
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
He, X., Carter, D. Atomic structure and chemistry of human serum albumin. Nature 358, 209–215 (1992). https://doi.org/10.1038/358209a0
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/358209a0
- Springer Nature Limited
This article is cited by
-
Novel nanotherapeutics for cancer immunotherapy by albumin nanoparticles functionalized with PD-1 and PD-L1 aptamers
Cancer Nanotechnology (2024)
-
A novel inflammatory biomarker, high-sensitivity C-reactive protein-to-albumin ratio, is associated with 5-year outcomes in patients with type 2 diabetes who undergo percutaneous coronary intervention
Diabetology & Metabolic Syndrome (2023)
-
Derivatization with fatty acids in peptide and protein drug discovery
Nature Reviews Drug Discovery (2023)
-
Organ-On-A-Chip Technology: An In-depth Review of Recent Advancements and Future of Whole Body-on-chip
BioChip Journal (2023)
-
High power impulse magnetron sputtering (HiPIMS) prepared ultrathin gold film for plasmonic biosensor application
Gold Bulletin (2023)