Abstract
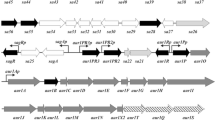
ERYTHROMYCIN A, a clinically important polyketide antibiotic, is produced by the Gram-positive bacterium Saccharopolyspora erythraea.. In an arrangement that seems to be generally true of antibiotic biosynthetic genes in Streptomyces and related bacteria like S. erythraea1, the ery genes encoding the biosynthetic pathway to erythromvein are clustered around the gene (ermE) that confers self-resistance on S. erythraea2–6. The aglycone core of erythro-mycin A is derived from one propionyl-CoA and six methylmalonyl-CoA units, which are incorporated head-to-tail7–10 into the growing polyketide chain, in a process similar to that of fatty-acid biosynthesis1, to generate a macrolide intermediate, 6-deoxyeryth-ronolide B10. 6-Deoxyerythronolide B is converted into erythro-mycin A through the action2–5,10 of specific hydroxylases, glycosyItransferases and a methyltransferase. We report here the analysis of about 10 kilobases of DNA from S. erythraea, cloned by chromosome 'walking' outwards from the erythromycin-resistance determinant ermE, and previously shown to be essential for erythromycin biosynthesis5,11. Partial sequencing of this region12 indicates that it encodes the synthase. Our results confirm this, and reveal a novel organization of the erythromycin-producing polyketide synthase, which provides further insight into the mechanism of chain assembly.
Similar content being viewed by others
References
Sherman, D. H. & Hopwood, D. A. A. Rev. Genet. 24, (in the press).
Stanzak, R., Matsushima, P., Baltz, R. H. & Rao, R. N. B. Bio/technology 4, 229–232 (1986).
Weber, J. M. & Losick, R. Gene 68, 173–180 (1988).
Weber, J. M., Wierman, C. K. & Hutchinson, C. R. J. Bact. 164, 425–433 (1985).
Weber, J. M., Leung, J. O., Maine, G. T., Potenz, R. H. B., Paulus, T. J. & DeWitt, J. P. J. Bact. 172, 2372–2383 (1990).
Dhillon, N., Hale, R. S., Cortes, J. & Leadlay, P. F. Molec. Microbiol. 3, 1405–1414 (1989).
Kaneda, T., Butte, J. C., Taubman, S. B. & Corcoran, J. W. J. biol. Chem. 237, 322–328 (1962).
Cane, D. E., Hasler, H. & Liang, T.-C. J. Am. chem. Soc. 103, 5960–5962 (1981).
Cane, D. E., Liang, T.-C., Taylor, P. B., Chang, C. & Yang, C.-C. J. Am. chem. Soc. 108, 4957–4964 (1986).
Seno, E. T. & Hutchinson, C. R. in The Bacteria Vol IX (eds Queener, S. W. & Day, L. E) 231–279 (Academic, New York, 1986).
Tuan, J. S. et al. Gene 90, 21–29 (1990).
Donadio, S. et al. In: Genetics and Microbiology of Industrial Microorganisms (eds Hershberger, C. L., Queener, S. W. & Hageman, G.) 53–59 (Am. Soc. of Microbiol., Washington, DC 1989).
Kauppinen, S., Siggaard-Andersen, M. & von Wettstein-Knowles, P. Carlsberg Res. Commun. 53, 357–370 (1988).
Bibb, M. J., Biro, S., Motamedi, H., Collins, J. F. & Hutchinson, C. R. EMBO J. 8, 2727–2736 (1989).
Sherman, D. H. et al. EMBO J. 8, 2727–2735 (1989).
Schweizer, M. et al. Molec. gen. genet. 203, 479–486 (1986).
Schweizer, M., Takabayashi, K., Laux, T., Beck, K. F. & Schreglmann, R. Nucleic Acids Res. 17, 567–586 (1988).
Chirala, S. S. et al. J. biol. Chem. 264, 3750–3757 (1989).
Mohamed, A. H., Chirala, S. S., Mody, N. H., Huang, W.-Y. & Wakil, S. J. J. biol. Chem. 263, 12315–12325 (1988).
Staden, R. Nucleic Acids Res. 12, 521–528 (1984).
Wakil, S. J. Biochemistry 28, 4523–4530 (1990).
Lawen, A. & Zocher, R. J. biol. Chem. 265, 11355–11360 (1990).
Skarpeid, H.-J., Zimmer, T.-L. & Von Doehren, H. Eur. J. Biochem. 189, 517–522 (1990).
Sanger, F., Nicklen, S. & Coulson, A. R. Proc. natn. Acad. Sci. U.S.A. 74, 5463–5467 (1977).
Frischauf, A.-M., Garoff, H. & Lehrach, H. Nucleic Acids Res. 8, 5541–5549 (1980).
Devereux, J., Haeberli, P. & Smithies, O. Nucleic Acids Res. 12, 387–395 (1984).
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Cortes, J., Haydock, S., Roberts, G. et al. An unusually large multifunctional polypeptide in the erythromycin-producing polyketide synthase of Saccharopolyspora erythraea. Nature 348, 176–178 (1990). https://doi.org/10.1038/348176a0
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1038/348176a0
- Springer Nature Limited
This article is cited by
-
Enzymology of assembly line synthesis by modular polyketide synthases
Nature Chemical Biology (2023)
-
Insights into azalomycin F assembly-line contribute to evolution-guided polyketide synthase engineering and identification of intermodular recognition
Nature Communications (2023)
-
Saccharomyces cerevisiae as host for the recombinant production of polyketides and nonribosomal peptides
Microbial Cell Factories (2021)
-
Defining the genes for the final steps in biosynthesis of the complex polyketide antibiotic mupirocin by Pseudomonas fluorescens NCIMB10586
Scientific Reports (2019)
-
Full-length title: NRPPUR database search and in vitro analysis identify an NRPS-PKS biosynthetic gene cluster with a potential antibiotic effect
BMC Bioinformatics (2018)