Abstract
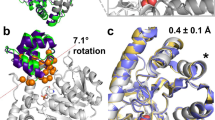
The substrate specificity of α-lytic protease has been changed dramatically, with a concomitant increase in activity, by replacing an active-site Met with Ala. The substrate specificity of both this mutant and another similar mutant are extraordinarily broad. X-ray crystallographic analysis shows that structural plasticity, a combination of alternate side-chain conformations and binding-site flexibility, allows both large and small substrates to be well accommodated.
Similar content being viewed by others
References
Fersht, A. R. Enzyme Structure and Mechanism (Freeman, San Francisco, 1985).
Wells, J. A. Cunningham, B. C., Graycar, T. P. & Estell, D. A. Proc. natn. Acad. Sci. U.S.A. 84, 5167 (1987).
Estell, D. A. et al. Science 233, 659 (1986).
Wilks, H. M. et al. Science 242, 1541–1544 (1988).
Craik, C. S. et al. Science 228, 291–297 (1985).
Cronin, C. N. & Kirsch, J. F. Biochemistry 27, 4572 (1988).
Fujinaga, M., Delbaere, L. T. J., Brayer, G. D. & James, M. N. G. J. molec. Biol. 183, 479 (1985).
Brayer, G. D., Delbaere, L. T. J. & James, M. N. G. J. molec. Biol. 131, 743 (1979).
Hunkapiller, M. W., Forgac, M. D. & Richards, J. H. Biochemistry 15, 5581 (1976).
Bachovchin, W. W., Kaiser, R., Richards, J. H. & Roberts, J. D. Proc. natn. Acad. Sci. U.S.A. 78, 7323 (1981).
Robillard, G. & Schulman, R. G. J. molec. Biol. 86, 541 (1974).
Bone, R., Shenvi, A. B., Kettner, C. A. & Agard, D. A. Biochemistry 27, 7609 (1987).
Schecter, I. & Berger, A. Biochem. Biophys. Res. Commun. 27, 157 (1967).
Bone, R., Frank, D., Kettner, C. A. & Agard, D. A. Biochemistry (in the press).
Bauer, C.-A., Brayer, G. D., Sielecki, A. R. & James, M. N. G. Eur. J. Biochem. 120, 289–294 (1981).
Kettner, C. A., Bone, R., Agard, D. A. & Bachovchin, W. W. Biochemistry 27, 7682–7688 (1988).
Dorovska, V. M., Variolomegev, S. D., Kazanskaya, N. F., Klyosov, A. A. & Martinek, K. FEBS Lett. 23, 122 (1972).
Harper, W. J., Cook, R. R., Roberts, C. J., McLaughlin, B. J. & Powers, J. C. Biochemistry 23, 2995 (1984).
Nakajima, K., Powers, J. C., Ashe, B. M. & Zimmerman, M. J. Biol. Chem. 254, 4027 (1979).
Connolly, M. L. J. Appl. Crystallogr. 16, 548 (1983).
Connolly, M. L. Science 221, 709 (1983).
Silen, J. L., Frank, D., Fujishige, A., Bone, R. & Agard, D. A. J. Bacteriol. 171, 1320 (1989).
Wells, J. A., Vasser, M. & Powers, D. B. Gene 34, 315 (1985).
Whitaker, D. R. Meth. Enzymol. 19, 599 (1970).
Hunkapiller, M. W., Smallcombe, S. H., Whitaker, D. R. & Richards, J. H. Biochemistry 12, 4732 (1973).
Williams, J. W. & Morrison, J. F. Meth. Enzymol. 63, 437–467 (1979).
Hendrickson, W. A. & Konnert, J. in Biomolecular Structure, Function, Conformation, and Evolution (ed. Srinivasam, R.), Vol. 1, 43–47 (Pergamon, Oxford, 1981).
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Bone, R., Silen, J. & Agard, D. Structural plasticity broadens the specificity of an engineered protease. Nature 339, 191–195 (1989). https://doi.org/10.1038/339191a0
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1038/339191a0
- Springer Nature Limited
This article is cited by
-
Engineering subtilisin proteases that specifically degrade active RAS
Communications Biology (2021)
-
The effect of calciums on molecular motions of proteinase K
Journal of Molecular Modeling (2011)
-
Analysis of binding properties and specificity through identification of the interface forming residues (IFR) for serine proteases in silico docked to different inhibitors
BMC Structural Biology (2010)
-
How to evolve a silk purse from a sow's ear
Nature Chemical Biology (2006)
-
Designed divergent evolution of enzyme function
Nature (2006)