Abstract
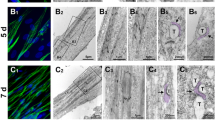
Muscular dysgenesis (mdg) is a spontaneous recessive lethal mutation in the mouse. The disease is characterized by a total lack of excitation-contraction coupling in embryonic skeletal muscle1–4. This developmental abnormality is associated with a drastic deficiency in the expression of voltage-sensitive Ca2+ channels in skeletal muscle3–5 without alteration of the properties of voltage-sensitive Na+ channels4 or of voltage-sensitive Ca2+ channels in cardiac3,5 and neuronal cells5. Membrane couplings between sarcoplasmic reticulum and the transverse tubules, known as triads, were also found to be drastically altered in embryonic muscle of the homozygous mutant (mdg / mdg). Triads in the mdg/mdg muscle were less numerous, disorganized and lacked spaced densities3,6. This paper shows that co-culture of mdg/mdg myotubes with normal spinal cord neurons re-establishes Ca2+ channel activity, contraction and normal triad organization. The decrease thus cannot be due to a mutation of the Ca2+ channel as previously suggested5. Normal nerve cells may supply an essential factor to mutant muscle cells.
Similar content being viewed by others
References
1. Bowden-Essien, F. Devl Biol. 27, 351–364 (1972). 2. Koenig, J., Bournaud, R., Powell, J. A. & Rieger, F. Devl Biol. 92, 188–196 (1982). 3. Pitmen-Raymond, M., Rieger, F., Fosset, M.& Lazdunski, M. Devl Biol. 112,458–466(1985). 4. Romey, G., Rieger, F., Renaud, J. F., Pin9on-Raymond, M. & Lazdunski, M. Biochem. biophys. Res. Commun. 136, 935–940 (1986). 5. Beam, K. G., Knudson, C. M. & Powell, J. A. Nature 320, 168–170 (1986). 6. Banker, B. M. J. Neuropath, exp. Neurol. 36, 100–127 (1977). 7. Bordes, M., Bernengo, J. C. & Renaud, J. F. Rev. sclent. Instrum. 54, 1053–1058 (1983). 8. Hugues, M. et al. EMBO J. 1, 1039–1042 (1982). 9. Romey, G. & Lazdunski, M. Biochem. biophys. Res. Commun. 118, 669–674 (1984). 10. Cohen, M. W. Brain Res. 41, 457–463 (1972). 11. Cognard, C., Lazdunski, M. & Romey, G. Proc. natn. Acad. Sci. U.S.A. 83, 517–521 (1986). 12. Carbone, E. & Lux, H. D. Nature 310, 501–502 (1984). 13. Nowycky, M. C., Fox, A. P. & Tsien, R. W. Nature 316, 440–442 (1985). 14. Nilius, B., Hess, P., Lansman, J. B. & Tsien, R. W. Nature 316, 443–446 (1985). 15. Bossu, J. L., Feltz, A. & Thomann, J. M. Pfliigers Arch. ges. Physiol. 403, 360–368 (1985). 16. Bean, B. P. J. gen. Physiol. 86, 1–30 (1985). 17. Hamill, P., Marty, A., Neher, E., Sakman, B. & Sigworth, F. J. Pfliigers Arch. ges. Physiol. 391,85–100 (1981). 18. Pincon-Raymond, M., Murawsky, M., Mege, R. M. & Rieger, F. Devl Biol. (in the press).
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Rieger, F., Bournaud, R., Shimahara, T. et al. Restoration of dysgenic muscle contraction and calcium channel function by co-culture with normal spinal cord neurons. Nature 330, 563–566 (1987). https://doi.org/10.1038/330563a0
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1038/330563a0
- Springer Nature Limited
This article is cited by
-
Neuromuscular synaptic patterning requires the function of skeletal muscle dihydropyridine receptors
Nature Neuroscience (2011)
-
Reciprocal dihydropyridine and ryanodine receptor interactions in skeletal muscle activation
Journal of Muscle Research and Cell Motility (2011)
-
DHPR α1S subunit controls skeletal muscle mass and morphogenesis
The EMBO Journal (2010)
-
The gene coding for the ?1 subunit of the skeletal dihydropyridine receptor (Cchl1a3=mdg) maps to mouse Chromosome 1 and human 1q32
Mammalian Genome (1993)
-
Intramembrane charge movement in developing skeletal muscle cells from fetal mice
Pfl�gers Archiv European Journal of Physiology (1992)