Abstract
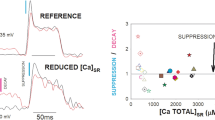
The transduction of action potential to muscle contraction (E–C coupling) is an example of fast communication between plasma membrane events and the release of calcium from an internal store, which in muscle is the sarcoplasmic reticulum (SR)1. One theory is that the release channels of the SR are controlled by voltage-sensing molecules or complexes, located in the transverse tubular (T)-membrane, which produce, as membrane voltage varies, 'intramembrane charge movements'2, but nothing is known about the structure of such sensors. Receptors of the Ca-channel-blocking dihydropyridines present in many tissues3, are most abundant in T-tubular muscle fractions4,5 from which they can be isolated as proteins. Fewer than 5% of muscle dihydropyridines are functional Ca channels6; there is no known role for the remainder in skeletal muscle physiology. We report here that low concentrations of a dihydropyridine inhibit charge movements and SR calcium release in parallel. The effect has a dependence on membrane voltage analogous to that of specific binding of dihydropyridines6. We propose specifically that the molecule that generates charge movement is the dihydropyridine receptor.
Similar content being viewed by others
References
Ebashi, S. A. Rev. Physiol. 38, 293–314 (1976).
Schneider, M. F. & Chandler, W. K. Nature 242, 244–246 (1973).
Janis, R. A. & Triggle, D. J. Drug Dev. Res. 4, 257–274 (1984).
Curtis, B. M. & Catternall, W. A. Biochemistry 23, 2113–2118 (1984).
Borsotto, M., Norman, R. I., Fosset, M. & Lazdunski, M. Eur. J. Biochem. 142, 449–455 (1984).
Schwartz, L. M., McCleskey, E. W. & Almers, W. Nature 314, 747–751 (1985).
Kovacs, L., Rios, E. & Schneider, M. F. Nature 279, 391–396 (1979).
McCleskey, E. W. J. Physiol., Lond. 361, 231–249 (1985).
Melzer, W., Rios, E. & Schneider, M. F. Biophys. J. 45, 637–641 (1984).
Melzer W., Rios, E & Schneider, M. F. Biophys. J. (in the press).
Beaty, G. N. & Stefani, E. J. Physiol., Lond. 260, 27P (1976).
Brum, G. & Rios E. J. Physiol, Lond (in the press).
Rios, E., Brum, G. & Stefani, E. Biophys. J. 49, 13a (1986).
Lamb, G. D. J. Physiol., Lond. 376, 85 (1986).
Morad, M., Goldman, Y. E. & Trentham, D. R. Nature 304, 635–638 (1983).
Fosset, M., Jaimovich, E., Delpont, E. & Lazdunski, M. J. biol. Chem. 258, 6086–6092 (1983).
Chandler, W. K., Rakowski, R. F. & Schneider, M. F. J. Physiol, Lond. 254, 245–283 (1972).
Eisenberg, R. S., McCarthy, R. T. & Milton, R. L. J. Physiol., Lond. 341, 495–595 (1983).
Hui, C. S., Milton, R. L. & Eisenberg, R. S. Proc. natn. Acad. Sci. U.S.A. 81, 2582–2585 (1984).
Toll, L. J. biol. Chem. 25, 13189–13192 (1982).
Gluecksohn-Waelsch, S. Science 142, 1269–1276 (1963).
Klaus, M. M., Stylianos, P. S., Rapalus, J. M., Briggs, R. T. & Powell, J. A. Devl. Biol. 99, 152–165 (1983).
Pincon-Raymond, M., Rieger, F., Fosset, M. & Lazdunski, M. Neurosci. Abstr. 10, 202 (1984).
Beam, K. G., Knudson, C. M. & Powell, J. A. Nature 320, 168–170 (1986).
Bean, B. P. Proc. natn. Acad. Sci. U.S.A. 81, 6388–6392 (1984).
Sanguinetti, M. C. & Kass, R. S. Circulation, Res. 55, 336–348 (1984).
Vergara, J., Tsien, R. Y. & Delay, M. Proc. natn. Acad. Sci. U.S.A. 82, 6352–6356 (1985).
Scarpa, A., Brinley, F. J. & Dubyak, G. Biochemistry 17, 1378–1386 (1978).
Rios, E. & Schneider, M. F. Biophys. J. 36, 607–621 (1981).
Kovacs, L., Rios, E. & Schneider, M. F. J. Physiol. Lond. 343, 161–196 (1983).
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Rios, E., Brum, G. Involvement of dihydropyridine receptors in excitation–contraction coupling in skeletal muscle. Nature 325, 717–720 (1987). https://doi.org/10.1038/325717a0
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1038/325717a0
- Springer Nature Limited
This article is cited by
-
Biophysical reviews top five: voltage-dependent charge movement in nerve and muscle
Biophysical Reviews (2023)
-
A recurrent RYR1 mutation associated with early-onset hypotonia and benign disease course
Acta Neuropathologica Communications (2021)
-
Excitation-contraction coupling in skeletal muscle: recent progress and unanswered questions
Biophysical Reviews (2020)
-
Postsynaptic CaV1.1-driven calcium signaling coordinates presynaptic differentiation at the developing neuromuscular junction
Scientific Reports (2019)
-
Ca2+-activated Cl− channel TMEM16A/ANO1 identified in zebrafish skeletal muscle is crucial for action potential acceleration
Nature Communications (2019)