Abstract
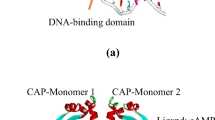
The 2.9 Å resolution crystal structure of Escherichia coli catabolite gene activator protein (CAP) completed with cyclic AMP reveals two distinct structural domains separated by a cleft. The smaller carboxy-terminal domain is presumed to bind DNA while the amino-terminal domain is seen to bind cyclic AMP. Model building studies suggest that CAP binds to left-handed B-type DNA, contacting its major groove via two a-helices. It is possible that the CAP conversion of right- to left-handed DNA in a closed supercoil, is what activates transcription by RNA polymerase.
Similar content being viewed by others
References
Zubay, G. & Doty, P. J. molec. Biol. 1, 1–20 (1959).
Sung, M. T. & Dixon, G. H. Proc. natn. Acad. Sci. U.S.A. 67, 1616–1623 (1970).
Adler, K. et al. Nature 237, 322–327 (1972).
Church, G. M., Sussman, J. L. & Kim, S. H. Proc. natn. Acad. Sci. U.S.A. 74, 1458–1462 (1977).
Zubay, G., Schwartz, D. & Beckwith, J. Proc. natn. Acad. Sci. U.S.A. 66, 104–110 (1970).
Epstein, W., Rothman-Denes, L. B. & Hesse, J. Proc. natn. Acad. Sci. U.S.A. 72, 2300–2304 (1975).
deCrombrugghe, B. et al. Nature new Biol. 231, 139–142 (1971).
Ogden, S., Haggerty, D., Stoner, C. M., Kolodrubetz, D. & Schleif, R. Proc. natn. Acad. Sci. U.S.A. 77, 3346–3350 (1980).
Musso, R. E., DiLauro, R., Adhya, S. & deChrombrugghe, B. Cell 12, 847–854 (1977).
Anderson, W. B., Schneider, A. B., Emmer, M., Perlman, R. L. & Pastan, I. J. biol. Chem. 246, 5929–5937 (1971).
Riggs, A. D., Reiness, G. & Zubay, G. Proc. natn. Acad. Sci. U.S.A. 68, 1222–1225 (1971).
Krakow, J. G. & Pastan, I. Proc. natn. Acad. Sci. U.S.A. 70, 2529–2533 (1973).
Eilen, E., Pampeno, G. & Krakow, J. S. Biochemistry 17, 2469–2473 (1978).
Wu, C.-W. & Wu, F. Y.-H. Biochemistry 13, 2573–2578 (1974).
Wu, F. Y.-H., Nath, K. & Wu, C.-W. J. molec. Biol. 100, 459–472 (1976).
Eilen, E. & Krakow, J. S. J. molec. Biol. 114, 47–60 (1977).
Kumar, S. A., Murthy, N. S. & Krakow, J. S. FEBS Lett. 109, 121–124 (1980).
McKay, D. B. & Fried, M. G. J. molec Biol. 139, 95–96 (1980).
Wyckoff, H. W. et al. J. molec. Biol. 27, 563–578 (1967).
Rossmann, M. G. Acta crystallogr. A 32, 774–777 (1976).
Strahs, G. & Kraut, J. J. molec. Biol. 35, 503–512 (1968).
Harrison, S. C., Olson, A. J., Schutt, C. E., Winkler, F. K. & Bricogne, G. Nature 276, 368–373 (1978).
Abad-Zapatero, C. et al. Nature 286, 33–39 (1980).
Bennett, W. S. Jr & Steitz, T. A. Proc. natn. Acad. Sci. U.S.A. 75, 4848–4852 (1978).
Amzel, M. & Poljak, R. J. A. Rev. Biochem. 48, 961–998 (1979).
Eklund, H., Samama, J.-P., Wallen, L. & Branden, C.-I. J. molec. Biol. (in the press).
Takahashi, M., Blazy, B. & Baudras, A. Biochemistry 19, 5124–5130 (1980).
Platt, T., Files, J. G. & Weber, K. J. biol. Chem. 248, 110–121 (1973).
Geisler, N. & Weber, K. Biochemistry 16, 938–943 (1977).
Jovin, T. M., Geisler, N. & Weber, K. Nature 269, 668–672 (1977).
Pabo, C. O., Saver, R. T., Sturtevant, J. N. & Ptashne, M. Proc. natn. Acad. Sci. U.S.A. 76, 1608–1612 (1979).
Ogata, R. T. & Gilbert, W. Proc. natn. Acad. Sci. U.S.A. 75, 5851–5854 (1978).
Simpson, R. B. Nucleic Acids Res. 8, 759–766 (1980).
Majors, J. thesis, Harvard Univ. (1979).
Gupta, G., Bansal, M. & Sasisekharan, V. Proc. natn. Acad. Sci. U.S.A. 77, 6486–6490 (1980).
Dickson, R. C., Abelson, J., Johnson, P., Reznikoff, W. S. & Barnes, W. M. J. molec Biol. 111, 65–79 (1977).
Taniguchi, T., O'Neill, M. & deCrombrugghe, B. Proc. natn. Acad. Sci. U.S.A. 76, 5090–5094 (1979).
Wang, A. H.-J. et al. Nature 282, 680–686 (1979).
Drew, H., Takano, T., Tanaka, S., Itakura, K. & Dickerson, R. E. Nature 286, 567–573 (1980).
Sanzey, B. J. Bact. 138, 40–47 (1979).
Cozzarelli, N. R. Science 207, 953–960 (1980).
Dickson, R. G., Abelson, J., Barnes, W. M. & Reznikoff, N. S. Science 187, 27–35 (1975).
Siebenlist, U. Nature 279, 651–652 (1979).
Anderson, W. F., Ohlendorf, D. H., Takeda, Y. & Matthews, B. W. Nature 290, 754–757 (1981).
Arnott, S. & Hukins, D. W. L. Biochem. biophys. Res. Commun. 47, 1504–1509 (1972).
Wilson, I. A., Skehel, J. J. & Wiley, D. C. Nature 289, 366–373 (1981).
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
McKay, D., Steitz, T. Structure of catabolite gene activator protein at 2.9 Å resolution suggests binding to left-handed B-DNA. Nature 290, 744–749 (1981). https://doi.org/10.1038/290744a0
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1038/290744a0
- Springer Nature Limited
This article is cited by
-
cAMP Activation of the cAMP Receptor Protein, a Model Bacterial Transcription Factor
Journal of Microbiology (2023)
-
Evolutionary History of TOPIIA Topoisomerases in Animals
Journal of Molecular Evolution (2022)
-
DNA–protein interaction studies: a historical and comparative analysis
Plant Methods (2021)
-
UV oxidation of cyclic AMP receptor protein, a global bacterial gene regulator, decreases DNA binding and cleaves DNA at specific sites
Scientific Reports (2020)
-
Structural study to analyze the DNA-binding properties of DsrC protein from the dsr operon of sulfur-oxidizing bacterium Allochromatium vinosum
Journal of Molecular Modeling (2019)