Abstract
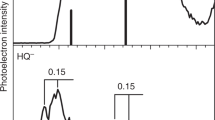
MOST of the better known radical anions and dianions have in common the fact that their precursors possess low-lying vacant molecular orbitals. It is deduced that compounds in which atoms are suitably disposed to afford the maximum overlap of vacant orbitals should constitute suitable electron acceptors so that a stable reduced species might be obtained.
Similar content being viewed by others
References
Rajbenbach, L. A., J. Amer. Chem. Soc., 88, 4275 (1966); J. Chem. Phys., 47, 242 (1967).
Hamill, W. H., J. Amer. Chem. Soc., 84, 500 (1962).
Ronayne, M. R., Guarino, J. P., and Hamill, W. H., J. Amer. Chem. Soc., 84, 4230 (1962).
Smith, D. R., and Pieroni, J. J., Canad. J. Chem., 43, 2141 (1965).
Hamill, W. H., in Radical Ions (edit. by Kevan, L.) (John Wiley, New York, 1968).
Mahan, B. H., and Young, C. E., J. Chem. Phys., 44, 2192 (1966).
Sagert, K. H., Canad. J. Chem., 46, 95 (1968).
Korff, S. A., Electronic and Nuclear Counters (D. Van Nostrand Co., New York, 1946).
Altshuler, A. A., and Clemons, C. A., Anal. Chem., 38, 133 (1966).
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
MITTAL, J., LIBBY, W. Intramolecular Electrostatic Electron Traps. Nature 220, 1027–1028 (1968). https://doi.org/10.1038/2201027a0
Received:
Revised:
Issue Date:
DOI: https://doi.org/10.1038/2201027a0
- Springer Nature Limited
This article is cited by
-
Light Emission from the Dissolution of Gamma Irradiated Alkali Halides in Water
Nature Physical Science (1971)