Abstract
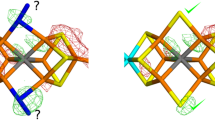
The secondary structure of the haemoglobin chains is similar to that of myoglobin, but some of the helical segments are more irregular and some parts of the non-helical segments have different conformations. The structure of the contacts between unlike subunits suggests that the tetramer, rather than the αβ dimer, is the functional unit of haemoglobin.
Similar content being viewed by others
References
Perutz, M. F., J. Mol. Biol., 13, 646 (1965).
Kendrew, J. C., Brookhaven Symposia in Biology, 15, 216 (1962).
Diamond, R., Acta Cryst., 21, 253 (1966).
Kendrew, J. C., Watson, H. C., Strandberg, B. E., Dickerson, R. E., Phillips, D. C., and Shore, V. C., Nature, 190, 663 (1961).
Low, B. W., and Grenville-Wells, H. J., Proc. US Nat. Acad. Sci., 39, 785 (1953).
Ramachandran, G. N., Ramakrishnan, C., and Sasisekharan, V., J. Mol. Biol., 7, 95 (1963).
Ramakrishnan, C., and Ramachandran, G. N., Biophys. J., 5, 909 (1965).
Watson, H. C., Progress in Stereochemistry IV (edit. by Harris and Aylett) (Butterworths, London, in the press).
Muirhead, H., Cox, J. M., Mazzarella, L., and Perutz, M. F., J. Mol. Biol., 28, 117 (1967).
Bolton, W., Cox, J. M., and Perutz, M. J., J. Mol. Biol., 33, 283 (1968).
Nakahara, A., and Wang, J. H., J. Amer. Chem. Soc., 80, 6526 (1958).
Vinograd, S., and Hutchinson, W. O., Nature, 187, 216 (1960).
Guidotti, G., and Craig, L. C., Proc. US Nat. Acad. Sci., 50, 46 (1963).
Guidotti, G., Konigsberg, W., and Craig, L. C., Proc. US Nat. Acad. Sci., 50, 774 (1963).
Rosemeyer, M. A., and Huehns, E. R., J. Mol. Biol., 25, 253 (1967).
Rossi Fanelli, A., Antonini, E., and Caputo, A., Adv. Protein Chem., 19, 73 (1964).
Guidotti, G., J. Biol. Chem., 242, 3704 (1967).
Nagel, R. L., Gibson, Q. H., and Charache, S., Biochemistry, 6, 2395 (1967).
Bonaventura, J., and Riggs, A., J. Biol. Chem., 243, 980 (1968).
Simon, S. R., and Konigsberg, W. H., Proc. US Nat. Acad. Sci., 56, 749 (1966).
Simon, S. R., Konigsberg, W. H., Bolton, W., and Perutz, M. F., J. Mol. Biol., 28, 451 (1967).
Gibson, Q. H., and Parkhurst, L. J., J. Biol. Chem. (in the press).
Chanutin, A., and Curnish, R. R., Arch. Biochim. Biophys., 121, 96 (1967).
Benesch, R., Benesch, R. E., and Yu, C. I., Proc. US Nat. Acad. Sci., 59, 526 (1968).
Dickerson, R. E., in The Proteins (second ed.) (edit. by Neurath, H.), 2, 603 (Academic Press, New York, 1964).
Cullis, A. F., Muirhead, H., Perutz, M. F., Rossmann, M. G., and North, A. C. T., Proc. Roy. Soc., A, 265, 161 (1962).
Lines, J. G., and McIntosh, R., Nature, 215, 297 (1967).
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
PERUTZ, M., MUIRHEAD, H., COX, J. et al. Three-dimensional Fourier Synthesis of Horse Oxyhaemoglobin at 2.8 Å Resolution: The Atomic Model. Nature 219, 131–139 (1968). https://doi.org/10.1038/219131a0
Received:
Published:
Issue Date:
DOI: https://doi.org/10.1038/219131a0
- Springer Nature Limited
This article is cited by
-
Analysis of the Contribution of Chromophores in Side Groups of Amino Acids to the Absorption Spectrum of Hemoglobin
Journal of Applied Spectroscopy (2014)
-
Excitation wavelength dependent pump–probe signatures of molecular crystals
Applied Physics A (2009)
-
The protein meta-structure: a novel concept for chemical and molecular biology
Cellular and Molecular Life Sciences (2009)
-
Oximetry with the NMR signals of hemoglobin Val E11 and Tyr C7
European Journal of Applied Physiology (2009)
-
Crystallographic evidence for dioxygen interactions with iron proteins
JBIC Journal of Biological Inorganic Chemistry (2007)