Abstract
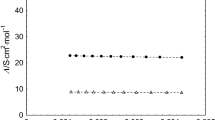
IT is interesting to compare the maximum values of the separation factor S obtained experimentally with the maximum theoretical value. For purely chemical processes the maximum theoretical value is that for the production of free gaseous atoms from the reactant molecules (in this case the water of the electrolyte1). For a 1 : 1 ratio of H and D in the electrolyte the separation factor is then the equilibrium constant for 
Similar content being viewed by others
References
Topley, B., and Eyring, H., J. Chem. Phys., 2, 217 (1934).
Urey, H. C., J. Chem. Soc., 562 (1947).
Benedict, W. S., Gailar, N., and Plyler, E. K., J. Chem. Phys., 34, 1139 (1956).
Jones, W. M., LADC 5905 Office of Tech. Services, Dept. Commerce, Washington, DC (1964).
Brun, J., and Varberg, T., Kgl. Norske Videnskabs. Selskabs. Forh., 26, 19 (1953).
Roy, L. P., Can. J. Chem., 40, 1452 (1960).
Rowland, P. R., AEEW-M 745 (HMSO, 1967).
Bockris, J. O'M., and Srinivasan, S., J. Electrochem. Soc., 111, 844 (1964).
De Boer, J. H., Vacuum, 16, 309 (1966).
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
ROWLAND, P. Electrolytic Separation Factor of Protium and Deuterium. Nature 218, 945–946 (1968). https://doi.org/10.1038/218945a0
Received:
Issue Date:
DOI: https://doi.org/10.1038/218945a0
- Springer Nature Limited
This article is cited by
-
Effects of additives on the practical electrolytic separation of H and D isotopes. I. Pt and Fe
Journal of Applied Electrochemistry (1975)