Abstract
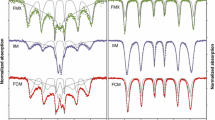
FRUCTOSE is capable of forming a highly stable chelate complex with iron, in contrast to some other hexose sugars and disaccharides which have little or no such ability1. Recently, attention has been focused on the iron–fructose complex because of its ability to move across biological membranes2 and to enhance gastrointestinal iron absorption in man3. The structure which has been proposed for this compound is one in which iron, incorporated into a five-membered ring, is bound through the hydroxyl and carbonyl oxygen atoms to carbon atoms one and two of the fructose molecule1,4.
Similar content being viewed by others
References
Charley, P. J., Sarkar, B., Stitt, C. F., and Saltman, P., Biochim. Biophys. Acta, 69, 313 (1963).
Stitt, C., Charley, P. J., Butt, E. M., and Saltman, P., Proc. Soc. Exp. Biol. and Med., 110, 70 (1962).
Davis, P. S., and Deller, D. J., Aust. Ann. Med. (in the press).
Aasa, R., Malmström, B., Saltman, P., and Vänngård, T., Biochim. Biophys. Acta, 80, 430 (1964).
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
DAVIS, P., DELLER, D. Prediction and Demonstration of Iron Chelating Ability of Sugars. Nature 212, 404–405 (1966). https://doi.org/10.1038/212404a0
Issue Date:
DOI: https://doi.org/10.1038/212404a0
- Springer Nature Limited
This article is cited by
-
The triose model: Glyceraldehyde as a source of energy and monomers for prebiotic condensation reactions
Origins of Life and Evolution of the Biosphere (1987)