Abstract
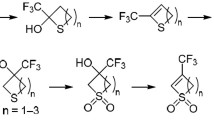
IN recent work1 we showed that bis(1,2-o-isopropylidene-3-o-thiocarbonyl-α-D-glucofuranose) disulphide rearranged on standing in basic organic solvents to give equimolar amounts of carbon disulphide, elemental sulphur, 1,2-o-isopropylidene-α-D-glucofuranose, and 1,2-o-isopropyli-dene-α-D-glucofuranose 5,6-thionocarbonate. That work has now been extended to an investigation of bis(o-thio-carbonyl) disulphide derivatives (referred to as xanthides) of 1,2-, 1,3-, 1,4-, 1,5-, and 1,6-dihydroxy alkanes.
Similar content being viewed by others
References
Doane, W. M., Shasha, B. S., Russell, C. R., and Rist, C. E., J. Org. Chem., 30, 162 (1965).
Feigl, F., Spot Tests, 2, 1964, fourth ed. (Elsevier Publishing Company, New York, 1954).
Sarel, S., Pohoryles, L. A., and Ben-Shoshan, R., J. Org. Chem., 24, 1873 (1959).
Twiss, D., J. Amer. Chem. Soc., 49, 491 (1927).
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
SHASHA, B., DOANE, W., RUSSELL, C. et al. Rearrangement of Bis(o-thiocarbonyl) Disulphides. Nature 211, 965–966 (1966). https://doi.org/10.1038/211965b0
Issue Date:
DOI: https://doi.org/10.1038/211965b0
- Springer Nature Limited