Abstract
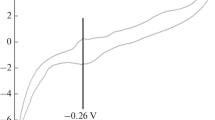
RESULTS of our potentiometric measurements of the photovoltaic effect in the ultra-violet region on platinum and mercury electrodes in aqueous solutions of sulphuric acid, potassium hydroxide and potassium sulphate led us to the conclusion that the observed potential changes of the illuminated electrode are caused by a competitive action of oxidizing and reducing species formed in water under the incident light.
Similar content being viewed by others
References
Swensson, T., Arkiv Kemi, Min. och Geologi, 7, No. 19, 1 (1919).
Lifschitz, J., and Hooghoudt, S. B., Z. phys. Chem., A, 141, 52 (1929).
Audubert, R., J. Chim. Phys., 27, 169 (1930).
Bowden, F. P., Trans. Farad. Soc., 27, 505 (1931).
Price, L. E., Dissertation, Cambridge Univ. (1938).
Hillson, P. J., and Rideal, E. K., Proc. Roy. Soc., A, 199, 295 (1949).
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
HEYROVSKÝ, M., NORRISH, R. Photovoltaic Phenomena in Aqueous Solutions. Nature 200, 880–881 (1963). https://doi.org/10.1038/200880a0
Issue Date:
DOI: https://doi.org/10.1038/200880a0
- Springer Nature Limited
This article is cited by
-
Nature of the Photoeffect in Aqueous Solutions
Nature (1965)