Abstract
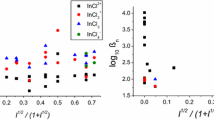
THERE is very little quantitative information about the relative co-ordinating tendencies of the heavier donor atoms of Groups V and VI, and this relates mainly to acceptor atoms (class a), which form their most stable complexes with the lightest donor atom of each group, namely, N, O and F−. This information has been obtained from vapour phase equilibria1. There is none relating to those metals (class b) which form their most stable complexes with the second lightest or one of the heavier donor atoms of each group, and only a very little, mainly semi quantitative, to atoms on the borderline between the classes, such as boron2 and thallium3.
Similar content being viewed by others
References
For example, (a) Davidson and Brown, J. Amer. Chem. Soc., 64, 316 (1942). (b) Coates, J. Chem. Soc., 2003 (1951).
Graham and Stone, J. Inorg. Nucl. Chem., 3, 164 (1956).
Coates and Whitcombe, J. Chem. Soc., 3351 (1956).
Bjerrum, “Metal Ammine Formation in Aqueous Solution”, 130 (Haas and Son, Copenhagen, 1941).
Bruehlman and Verhoek, J. Amer. Chem. Soc., 70, 1401 (1948).
Berne and Leden, Sven. Kem. Tid., 65, 88 (1953).
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
AHRLAND, S., CHATT, J., DAVIES, N. et al. Relative Tendencies of Similar Organic Derivatives of Nitrogen(III), Phosphorus(III), Arsenic(III), Oxygen(II), Sulphur(II) and Selenium(II) to form Complexes with Silver Ions. Nature 179, 1187–1188 (1957). https://doi.org/10.1038/1791187c0
Issue Date:
DOI: https://doi.org/10.1038/1791187c0
- Springer Nature Limited