Abstract
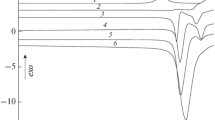
IN the course of a calorimetric investigation of the thermodynamic properties at low temperatures of the tetra-alkyl compounds of Group 4 elements, we have found that tin tetra-ethyl has an unusually large number of melting points. All these lie in the range 137–147° K. When this substance is cooled, it almost always supercools considerably before crystallizing, and it can, in fact, be kept as a glass at liquid air temperatures for at least two or three days. On warming the crystalline solid, the temperature – time curve may show the normal behaviour of a single arrest while melting is taking place, or it may display two, three or even more arrests. The temperatures at which these occur are reproducible, and in some twenty experiments (series I), in which the solid was melted in a low-temperature calorimeter1, the existence of at least five such temperatures was established. Six of the curves obtained, which between them indicate five melting points, are shown. In some of the calorimetric experiments the sample was deliberately heated continuously in order to minimize the chance of any recrystallization occurring after melting had started. The arrests in these runs are inclined and not horizontal.
Similar content being viewed by others
References
Staveley, L. A. K., and Gupta, A. K., Trans. Farad. Soc., 45, 50 (1949).
Mair, B. J., Glasgow, A. R., and Rossini, F. D., J. Res. Nat. Bur. Stand., 26, 591 (1941).
French, F. A., and Rasmussen, R. S., J. Chem. Phys., 14, 389 (1946).
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
STAVELEY, L., PAGET, H., GOALBY, B. et al. Polymorphism in Tin Tetra-ethyl and Related Compounds. Nature 164, 787–788 (1949). https://doi.org/10.1038/164787b0
Issue Date:
DOI: https://doi.org/10.1038/164787b0
- Springer Nature Limited