Abstract
Purpose. Development of reliable mathematical models to predict skin permeability remains a challenging objective. This article examines some of the existing algorithms and critically evaluates their statistical relevance.
Methods. Complete statistics were recalculated for a number of published models using a stepwise multiple regression procedure. The predictivity of the models was obtained by cross-validation using a “leave-one-out” deletion pattern. The relative contribution of each independent variable to the models was calculated by a standardization procedure.
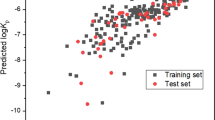
Results. The heterogeneity of the data in terms of skin origin and experimental conditions has been shown to contribute to the residual variance in existing models. Furthermore, rigorous statistics demonstrate that some published models are based on nonsignificant parameters. As such, they afford misleading mechanistic insight and will lead to over-interpretation of the data.
Conclusions. The large number of published models reflects the need for predictive tools in cutaneous drug delivery and toxicology. However, such models are more reliable when confined within well-defined chemical classes, and their applicability is often limited by the narrow property space of the set of permeants under study.
Similar content being viewed by others
REFERENCES
D. Howes, R. H. Guy, J. Hadgraft, J. Heylings, U. Hoeck, F. Kemper, H. I. Maibach, J. P. Marty, H. Merk, J. Parra, D. Rekkas, I. Rondelli, H. Schaefer, U. Täuber, and N. Verbiese. Methods for assessing percutaneous absorption. ATLA 24:81-106 (1996).
E. L. Cussler. Diffusion: Mass Transfer in Fluid Systems, 2nd ed. Cambridge University Press, New York, 1997.
J. C. Shah. Analysis of permeation data: evaluation of the lag time method. Int. J. Pharm. 90:161-169 (1993).
B. W. Barry. Dermatological Formulations. Dekker, New York, 1983.
S. Wold. Validation of QSAR's. Quant. Struct. Act. Relat. 10:191-193 (1991).
H. Kubinyi. QSAR and 3D QSAR in drug design. Part 1: methodology. Drug Discovery Today 2:457-467 (1997).
A. Golbraikh and A. Tropsha. Beware of q2! J. Mol. Graphics Model. 20:269-276 (2002).
H. Mager and A. Barth. Problems involved in the specification and interpretation of quantitative structure-activity relationships. Pharmazie 34:557-559 (1979).
P. P. Mager. Non-least-squares regression analysis applied to organic and medicinal chemistry. Med. Res. Rev. 14:553-588 (1994).
A. L. Bunge and R. L. Cleek. A new method for estimating dermal absorption from chemical exposure. 2. Effect of molecular weight and octanol-water partitioning. Pharm. Res. 12:88-95 (1995).
T. X. Xiang and B. D. Anderson. The relationshipo between permeant size and permeability in lipid bilayer membranes. J. Membrane Biol. 140:111-122 (1994).
A. N. Martin. Physical Pharmacy. Physical Chemical Principle in the Pharmaceutical Sciences. Les & Febiger, Philadelphia, 1993.
G. B. Kasting, R. L. Smith, and E. R. Cooper. Effect of lipid solubilities and molecular size on percutaneous absorption. In B. Shroot and H. Schaefer (eds.), Skin Pharmacokinetics. Karger, Basel, 1987, pp. 138-153.
N. El Tayar, R. S. Tsai, B. Testa, P. A. Carrupt, C. Hansch, and A. Leo. Percutaneous penetration of drugs: a quantitative structure-permeability relationship study. J. Pharm. Sci. 80:744-749 (1991).
M. S. Roberts, W. J. Pugh, and J. Hadgraft. Epidermal permeability: penetrant structure relationships. 2. The effect of H-bonding groups in penetrants on their diffusion through the stratum corneum. Int. J. Pharm. 132:23-32 (1996).
M. J. Kamlet and R. W. Taft. The solvatochromic comparison method. I. The β-scale of solvent hydrogen-bond acceptor (HBA) basicities. J. Am. Chem. Soc. 98:377-383 (1976).
R. W. Taft and M. J. Kamlet. The solvatochromic comparison method. 2. The α-scale of solvent hydrogen-bond donor (HBD) acidities. J. Am. Chem. Soc. 98:2886-2894 (1976).
D. E. Leahy. Intrinsic molecular volume as a measure of the cavity term in linear solvation energy relationships: octanol-water partition coefficients and aqueous solubilities. J. Pharm. Sci. 75:629-636 (1986).
M. H. Abraham and H. S. Chadha. Application of a solvation equation to drug transport properties. In V. Pliska, B. Testa, and H. van de Waterbeemd (eds.), Lipophilicity in Drug Action and Toxicology. VCH Publishers, Weinheim, 1996, pp. 311-337.
M. H. Abraham. Scales of solute hydrogen-bonding: their construction and application to physicochemical and biochemical processes. Chem. Soc. Rev. 22:73-83 (1993).
A. Pagliara, G. Caron, G. Lisa, W. Fan, P. Gaillard, P. A. Carrupt, B. Testa, and M. H. Abraham. Solvatochromic analysis of di-n-butyl ether/water partition coefficients as compared to other solvent systems. J. Chem. Soc. Perkin Trans. 2:2639-2643 (1997).
G. Steyaert, G. Lisa, P. Gaillard, G. Boss, F. Reymond, H. H. Girault, P. A. Carrupt, and B. Testa. Intermolecular forces expressed in 1,2-dichloroethane/water partition coefficient: a solvatochromic analysis. J. Chem. Soc. Faraday Trans. 93:401-406 (1997).
A. Pagliara, E. Khamis, A. Trinh, P. A. Carrupt, R. S. Tsai, and B. Testa. Structural properties governing retention mechanisms on RP-HPLC stationary phase used for lipophilicity measurements. J. Liquid Chromatogr. 18:1721-1745 (1995).
M. H. Abraham, H. S. Chadha, and R. C. Mitchell. Hydrogen bonding. 33. Factors that influence the distribution of solutes between blood and brain. J. Pharm. Sci. 83:1257-1268 (1994).
G. R. Famini and L. Y. Wilson. Using theoretical descriptors in quantitative structure activity relationships and linear free energy relationships. Network Science Co., 1116 Miller Mountain Road, Saluda, NC 28773. www.netsci.org/Science/Compchem/feature08.html (1996).
J. S. Murray, P. Politzer, and G. R. Famini. Theoretical alternatives to linear solvation energy relationships. J. Mol. Struct. 454:299-306 (1998).
C. Hansch and A. Leo. Substituent Constants for Correlation Analysis in Chemistry and Biology. John Wiley & Sons, New York, 1979.
W. T. Nauta and R. F. Rekker (eds.), The Hydrophobic Fragmental Constant. Elsevier, Amsterdam, 1977.
B. Testa, L. B. Kier, and P. A. Carrupt. A systems approach to molecular structure, intermolecular recognition, and emergence-dissolvence in medicinal research. Med. Res. Rev. 17:303-327 (1997).
B. Testa, P. A. Carrupt, P. Gaillard, F. Billois, and P. Weber. Lipophilicity in molecular modeling. Pharm. Res. 13:335-343 (1996).
V. Pliska, B. Testa, and H. van de Waterbeemd (eds.). Lipophilicity in Drug Action and Toxicology. VCH, Weinheim, 1996.
B. Testa, H. van de Waterbeemd, G. Folkers, and R. H. Guy (eds.). Pharmacokinetic Optimization in Drug Research: Biological, Physicochemical and Computational Strategies. Wiley-VHCA, Zurich, 2001.
N. El Tayar, B. Testa, and P. A. Carrupt. Polar intermolecular interactions encoded in partition coefficients: an indirect estimation of hydrogen-bond parameters of polyfunctional solutes. J. Phys. Chem. 96:1455-1459 (1992).
G. Caron, F. Reymond, P. A. Carrupt, H. H. Girault, and B. Testa. Combined molecular lipophilicity descriptors and their role in understanding intramolecular effects. Pharm. Sci. Technol. Today 2:327-335 (1999).
R. C. Young, R. C. Mitchell, T. H. Brown, C. R. Ganellin, R. Griffiths, M. Jones, K. K. Rana, D. Saunders, I. R. Smith, N. E. Sore, and T. J. Wilks. Development of a new physicochemical model for brain penetration and its application to the design of centrally acting H2 receptor histamine antagonists. J. Med. Chem. 31:656-671 (1988).
C. R. Ganellin, A. Fkyerat, S. K. Hosseini, Y. S. Khalaf, A. Piripitsi, W. Tertiuk, J. M. Arrang, M. Garbarg, X. Ligneau, and J. C. Schwartz. Structure-activity studies with histamine H3-receptor ligands. J. Pharm. Belg. 50:179-187 (1995).
C. R. Ganellin, A. Fkyerat, B. Bang-Andersen, S. Athmani, W. Tertiuk, M. Garbarg, X. Ligneau, and J. C. Schwartz. A novel series of (phenoxyalkyl)imidazoles as potent H3-receptor histamine antagonists. J. Med. Chem. 39:3806-3813 (1996).
B. Testa, P. A. Carrupt, P. Gaillard, and R. S. Tsai. Intramolecular interactions encoded in lipophilicity: their nature and significance. In V. Pliska, B. Testa, and H. van de Waterbeemd (eds.), Lipophilicity in Drug Action and Toxicology. VCH Publishers, Weinheim, 1996, pp. 49-71.
M. S. Roberts, R. A. Anderson, and J. Swarbrick. Permeability of human epidermis to phenolic compounds. J. Pharm. Pharmacol. 29:677-683 (1977).
R. J. Scheuplein and I. H. Blank. Permeability of the skin. Pharmacol. Rev. 51:702-747 (1971).
J. Houk and R. H. Guy. Membrane models for skin penetration studies. Chem. Rev. 88:455-471 (1988).
G. Ridout, J. Houk, R. H. Guy, G. C. Santus, J. Hadgraft, and L. L. Hall. An evaluation of structure-penetration relationships in percutaneous absorption. Farmaco 47:869-892 (1992).
R. H. Guy and J. Hadgraft. Structrure-activity correlations in percutaneous absorption. In R. L. Bronaugh and H. I. Maibach (eds.), Percutaneous Absorption. Mechanisms-Methodology-Drug Delivery. Marcel Dekker, New York, 1989, pp. 95-109.
G. L. Flynn. Physicochemical determinants of skin absorption. In T. R. Gerrity and C. J. Henry (eds.), Principles of Route-to-Route Extrapolation for Risk Assessment. Elsevier, Amsterdam, 1990, pp. 93-127.
R. O. Potts and R. H. Guy. Predicting skin permeability. Pharm. Res. 9:663-669 (1992).
D. Southwell, B. W. Barry, and R. Woodford. Variations in permeability of human skin within and between specimens. Int. J. Pharm. 18:299-309 (1984).
A. Wilshut, W. F. ten Berge, P. J. Robinson, and T. E. McKone. Estimating skin permeation. The validation of five mathematical skin permeation models. Chemosphere 30:1275-1296 (1995).
R. H. Guy and R. O. Potts. Penetration of industrial chemicals across the skin: a predictive model. Am. J. Ind. Med. 23:711-719 (1993).
T. E. McKone and R. A. Howd. Estimating dermal uptake of nonionic organic chemicals from water and soil. I. Unified fugacity-based models for risk assessments. Risk Anal. 12:543-557 (1992).
S. L. Brown and J. E. Rossi. A simple method for estimating dermal absorption of chemicals in water. Chemosphere 19:1989-2001 (1989).
V. Fiserova-Bergerova, J. T. Pierce, and P. O. Droz. Dermal absorption potential of industrial chemicals: criteria for skin notation. Am. J. Ind. Med. 17:617-635 (1990).
P. S. Magee. Some novel approaches to modelling transdermal penetration and reactivity with epidermal proteins. In J. Devillers (ed.), Comparative QSAR. Taylor & Francis, London, 1998, pp. 137-168.
L. A. Kirchner, R. P. Moody, E. Doyle, R. Bose, J. Jeffery, and I. Chu. The prediction of skin permeability by using physicochemical data. ATLA 25:359-370 (1997).
M. T. D. Cronin, J. C. Dearden, G. P. Moss, and G. Murray-Dickson. Investigation of the mechanism of flux across human skin in vitro by quantitative structure-permeability relationships. Eur. J. Pharm. Sci. 7:325-330 (1999).
H. F. Frasch and D. P. Landsittel. Regarding the sources of data analyzed with quantitative structure-skin permeability relationship methods (commentary on “Investigation of the mechanism of flux across human skin in vitro by quantitative structure-permeability relationships”). Eur. J. Med. Chem. 15:399-403 (2002).
R. O. Potts and R. H. Guy. A predictive algorithm for skin permeability: the effects of molecular size and hydrogen bond activity. Pharm. Res. 12:1628-1633 (1995).
M. H. Abraham, H. S. Chadha, and R. C. Mitchell. The factors that influence skin penetration of solutes. J. Pharm. Pharmacol. 47:8-16 (1995).
M. H. Abraham and J. C. McGowan. The use of characteristic volumes to measure cavity terms in reversed phase liquid chromatography. Chromatographia 23:243-246 (1987).
M. H. Abraham, F. Martins, and R. C. Mitchell. Algorithms for skin permeability using hydrogen bond descriptors: the problem of steroids. J. Pharm. Pharmacol. 49:858-865 (1997).
M. H. Abraham, H. S. Chadha, F. Martins, R. C. Mitchell, M. W. Bradbury, and J. A. Gratton. Hydrogen bonding part. 46. A review of the correlation and prediction of transport properties by an LFER method: physicochemical properties, brain penetration and skin permeability. Pestic. Sci. 55:75-88 (1999).
S. Rey. Hydrogen-bonds and Other Recognition Forces in Molecular Modeling. Ph.D. Thesis, University of Lausanne (2002).
W. J. Pugh, M. S. Roberts, and J. Hadgraft. Epidermal permeability-penetrant structure relationships. 3. The effect of hydrogen bonding interactions and molecular size on diffusion across the stratum corneum. Int. J. Pharm. 138:149-165 (1996).
W. J. Pugh and J. Hadgraft. Ab initio prediction of human skin permeability coefficients. Int. J. Pharm. 103:163-178 (1994).
E. J. Lien and H. Gao. QSAR analysis of skin permeability of various drugs in man as compared to in vitro and in vitro studies in rodents. Pharm. Res. 12:583-587 (1995).
W. J. Pugh, I. T. Degim, and J. Hadgraft. Epidermal permeability-penetrant structure relationships. 4. QSAR of permeant diffucion across human stratum corneum in terms of molecular weight, H-bonding and electronic charge. Int. J. Pharm. 197:203-211 (2000).
M. D. Barratt, Quantitative structure-activity relationships for skin permeability. Toxicol. in Vitro 9:27-37 (1995).
J. C. Dearden, M. T. D. Cronin, H. Patel, and O. A. Raevsky. QSAR prediction of human skin permeability coefficients. J. Pharm. Pharmacol. 52(Suppl):221(2000).
D. F. Veber, S. R. Johnson, H. Y. Cheng, B. R. Smith, K. W. Ward, and K. D. Kopple. Molecular properties that influence the oral bioavailability of drug candidates. J. Med. Chem. 45:2615-2623 (2002).
T. Ghafourian and S. Fooladi. The effect of structural QSAR parameters on skin penetration. Int. J. Pharm. 217:1-11 (2001).
P. Buchwald and N. Bodor. A simple, predictive, structure-based skin permeability model. J. Pharm. Pharmacol. 53:1087-1098 (2001).
J. E. Johnson, D. Blankstein, and R. Langer. Evaluation of solute permeation through the stratum corneum: lateral bilayer diffusion as the primary transport mechanism. J. Pharm. Sci. 86:1162-1172 (1997).
J. J. Hostynek and P. S. Magee. Modelling in vivo human skin absorption. Quant. Struct. Act. Relat. 16:473-479 (1997).
M. E. Johnson, D. Blankschtein, and R. Langer. Permeation of steroids through human skin. J. Pharm. Sci. 84:1144-1146 (1995).
I. T. Degim, W. J. Pugh, and J. Hadgraft. Skin permeability data: anomalous results. Int. J. Pharm. 170:129-133 (1998).
R. J. Scheuplein, I. H. Blank, G. J. Baruner, and D. J. MacFarlane. Percutaneous absorption of steroids. J. Invest. Dermatol. 52:63-70 (1969).
B. E. Vecchia and A. Bunge. Evaluating the transdermal permeability of chemicals. In R. H. Guy and J. Hadgraft (eds.), Transdermal Drug Delivery. Marcel Dekker, New York, 2003, pp. 25-55.
B. E. Vecchia and A. Bunge. Skin absorption databases and predictive equations. In R. H. Guy and J. Hadgraft (eds.), Transdermal Drug Delivery. Marcel Dekker, New York, 2003, pp. 57-141.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Geinoz, S., Guy, R.H., Testa, B. et al. Quantitative Structure-Permeation Relationships (QSPeRs) to Predict Skin Permeation: A Critical Evaluation. Pharm Res 21, 83–92 (2004). https://doi.org/10.1023/B:PHAM.0000012155.27488.2b
Issue Date:
DOI: https://doi.org/10.1023/B:PHAM.0000012155.27488.2b