Abstract
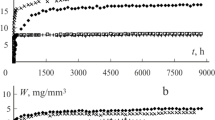
A study is reported in which a series of dental cements of varying types (zinc phosphate, zinc polycarboxylate, glass-ionomer and resin-modified glass-ionomer) was exposed to aqueous solutions of differing pH for time intervals of a week, after which the pH of the storage solutions was determined. The results showed that all of the acid-base cements altered the pH of their storage solution, regardless of whether that initial solution was weakly acidic, weakly alkaline or close to neutral. All cements were found to act as buffers, because they not only increased the pH of the weakly acidic lactic acid solution, but they also decreased the pH of the weakly alkaline artificial saliva. In deionized water, the zinc polycarboxylate generally increased pH, while all other cements reduced it. In all cases, these results were shown to be repeatable on exposure to fresh-aqueous solutions of the appropriate pH for a further week, such experiments being carried out for up to six weeks. In terms of mass change, in most solutions, there was a modest increase during the first week, after which the mass remained steady. In lactic acid, zinc phosphate and zinc polycarboxylate cements showed a gradual reduction in mass throughout the six weeks, whereas the glass-ionomers showed an initial increase, followed by a much slower decrease in mass. These results confirm that glass-ionomers are the most resistant of the cements towards acid erosion.
Similar content being viewed by others
References
A. D. Wilson and J. W. Nicholson, “Acid-base cements” (Cambridge University Press, 1993).
J. W. McLean, J. W. Nicholson and A. D. Wilson, Quintessence Int. 25 (1994) 587.
B. G. N. Smith, P. S. Wright and D. Brown, “The clinical handling of materials”, 2nd Edn (Butterworth-Heinemann, Oxford, 1994).
L. Forsten, Scand. J. Dent. Res. 85 (1977) 503.
S. B. Mitra, J. Dent. Res. 70 (1991) 72.
M. Tyas, Aust. Dent. J. 36 (1991) 236.
L. Forsten, Acta Odontol. Scand. 51 (1993) 195.
J. W. Nicholson, B. Czarnecka and H. Limanowska-Shaw, Biomaterials submitted.
International Organisation for Standardisation, ISO 9917 (ISO, London, 1991).
S. Hojo, N. Takahashi and T. Yamada, J. Dent. Res. 70 (1991) 182.
S. Hojo, M. Komatsu, R. Okuda, N. Takahashi and T. Yamada, ibid. 73 (1994) 1853.
British Standards Institution, BS 7115, Part 2 (BSI, London, 1988).
S. Crisp, B. G. Lewis and A. D. Wilson, J. Dent. Res. 55 (1976) 1032.
S. Matsuya, M. Koda, Y. Matsuya, Y. Yamamoto and M. Yamane, Dent. Mater. J. 2 (1983) 76.
R. D. Harrison (ed.), “Book of data” (Longman, London, 1972).
J. W. Nicholson, P. J. Brookman, O. M. Lacy and A. D. Wilson, J. Dent. Res. 67 (1988) 145.
J. W. Nicholson, P. J. Brookman, O. M. Lacy, G. S. Sayers and A. D. Wilson, J. Biomed. Mater. Res. 22 (1988) 623.
W. D. Cook, ibid. 17 (1983) 1015.
A. D. Wilson, D. M. Groffman, D. R. Powis and R. P. Scott, Biomaterials 7 (1986) 217.
N. E. Hersek and S. Canay, Quintessence Int. 27 (1996) 211.
S. Crisp, B. G. Lewis and A. D. Wilson, J. Dent. Res. 55 (1976) 299.
E. A. Wasson and J. W. Nicholson, ibid. 72 (1993) 481.
S. Matsuya, T. Maeda and M. Ohta, ibid. 75 (1996) 1920.
J. W. Nicholson, H. M. Anstice and J. W. McLean, Brit. Dent. J. 176 (1992) 98.
H. M. Anstice and J. W. Nicholson, J. Mater. Sci. Mater. Med. 3 (1992) 447.
W. Kanchanavasita, G. J. Pearson and H. M. Anstice, Biomaterials 18 (1997) 343.
J. W. Nicholson, J. Mater. Sci. Mater. Med. 8 (1997) 691.
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Nicholson, J., Amiri, M. The interaction of dental cements with aqueous solutions of varying pH. Journal of Materials Science: Materials in Medicine 9, 549–554 (1998). https://doi.org/10.1023/A:1008908523969
Issue Date:
DOI: https://doi.org/10.1023/A:1008908523969