Abstract
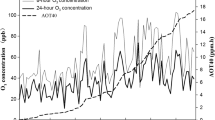
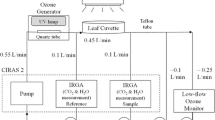
We examined the characteristics of ascorbic acid (ASC) level, dehydroascorbate (DHA) level, and the ASC–DHA redox status in the leaflets of two soybean cultivars grown in a field environment and exposed to elevated ozone (O3) levels. These two cultivars, one that preliminary evidence indicated to be O3-tolerant (cv Essex), and one that was indicated to be O3-sensitive (cv Forrest), were grown in open-top chambers during the summer of 1997. The plants were exposed daily to a controlled, moderately high O3 level (≈58 nl l−1 air) in the light, beginning at the seedling stage and continuing to bean maturity. Concurrently, control plants were exposed to carbon-filtered, ambient air containing a relatively low O3 level (≈24 nl l−1 air) during the same period. Elevated O3 did not affect biomass per plant, mature leaf area accretion, or bean yield per plant of cv Essex. In contrast, elevated O3 level decreased the biomass and bean yield per plant of cv Forrest by approximately 20%. Daily leaflet photosynthesis rate and stomatal conductance per unit area did not decrease in either cultivar as a result of prolonged O3 exposure. A 10% lower mature leaflet area in O3-treated cv Forrest plants contributed to an ultimate limitation in long-term photosynthetic productivity (vegetative and bean yield). Possible factors causing cv Essex to be more O3 tolerant than cv Forrest were: 1) mature leaflets of control and O3-treated cv Essex plants consistently maintained a higher daily ASC level than leaflets of cv Forrest plants, and 2) mature leaflets of cv Essex plants maintained a higher daily ASC–DHA redox status than leaflets of cv Forrest plants.
Similar content being viewed by others
References
Burkey KO (1999) Effects of ozone apoplastic/cytoplasm partitioning of ascorbic acid in snap bean. Physiol Plant 107: 188–193
Castillo FJ and Greppin H (1988) Extracellular ascorbic acid and enzyme activities related to ascorbic acid metabolism in Sedum album L. leaves after ozone exposure. Environ and Exp Bot 28: 231–238
Conklin PL, Williams EH and Last RL (1996) Environmental stress sensitivity of an ascorbic acid-deficient Arabidopsis mutant. Proc Natl Acad Sci USA: 93: 9970–9974
Conklin PL, Norris SR, Wheeler GL, Williams EH, Smirnoff N and Last RL (1999) Genetic evidence for the role of GDP-mannose in plant ascorbic acid (vitamin C) biosynthesis. Proc Natl Acad Sci USA 96: 4198–4203
Davey MW, Gilot C, Persiau G, Østergaard J, Han Y, Bauw GC and Van Montagu MC (1999) Ascorbate biosynthesis in Arabidopsis cell suspension culture. Plant Physiol. 121: 535–543
Feldman DS Jr, Gagnon J, Hofman R and Simpson J (1987) StatView II. The Solution for Data Analysis and Presentation Graphics. Abacus Concepts, Inc., Berkeley, California 278 pp
Heagle AS (1989) Ozone and crop yield. Annu Rev Phytopathol 27: 397–423
Heagle AS, Philbeck RB and Letchworth MB (1979) Injury and yield responses of spinach cultivars to chronic doses of ozone in open-top field chambers. J Environ Qual 8: 368–373
Heagle AS, Miller JE and Booker FL (1998) Influence of ozone stress on soybean responses to carbon dioxide enrichment: I. Foliar properties. Crop Sci 38: 113–121
Heath RL (1994) Possible mechanisms for the inhibition of photosynthesis by ozone. Photosynth Res 39: 439–451
Heath RL (1996) The modification of photosynthetic capacity induced by ozone exposure. In: Baker NR (ed) Photosynthesis and the Environment, pp 409–433.Kluwer Academic Publishers, Dordrecht, The Netherlands
Lee EH, Jersey JA, Gifford C and Bennett J (1984) Differential ozone tolerance in soybeans: Analysis of ascorbic acid in O3-susceptible and O3-resistant cultivars by high-performance liquid chromatography. Exp Environ Bot 24: 331–341
LI-COR Inc. (1987) The LI-6200 Primer. LI-COR, Inc. Lincoln, Nebraska 68504 (1990 printing)
Loewus FA (1988) Ascorbic acid and its metabolic products. In: Preiss J (ed) The Biochemistry of Plants, Vol 14, pp 85–107. Academic Press, New York
Loewus FA (1999) Biosynthesis and metabolism of ascorbic acid in plants and analogs of ascorbic acid in fungi. Phytochemistry 52: 193–210
Luwe M (1996) Antioxidants in the apoplast and symplast of beech (Fagus sylvatica L.) leaves: Seasonal variations and responses to changing ozone concentrations in air. Plant Cell Environ 19: 321–328
Luwe M and Heber U (1995) Ozone detoxification in the apoplasm and symplasm of spinach, broad bean and beech leaves at ambient and elevated concentrations of ozone in air. Planta 197: 448–455
Luwe MWF, Takahawa U and Heber U (1993) Role of ascorbate in detoxifying ozone in the apoplast of spinach (Spinacia oleracea L.) leaves. Plant Physiol 101: 969–976
Lyons T, Ollerenshaw JH and Barnes JD (1999) Impacts of ozone on Plantago major apoplastic and symplastic antioxidant status. New Phytol 141: 253–263
MacKinney G (1941) Absorption of light by chlorophyll solutions. J Biol Chem 140: 315–322
Mulchi CL, Lee E, Tuthill K and Olnick EV (1988) Influence of ozone stress on growth processes, yields and grain quality characteristics among soybean cultivars. Environ Pollut 53: 151–169
Noctor G and Foyer CH (1998) Ascorbate and glutathione: keeping active oxygen under control. Ann Rev Plant Physiol Plant Mol Biol 49: 249–279
Pallanca JE and Smirnoff N (1999) Ascorbic acid metabolism in pea seedlings. A comparison of D-glucosone, L-sorbosone and lgalactono-1,4-lacone as ascorbate precursors. Plant Physiol 120: 453–461
Polle A, Chakrabarti K and Schürmann W (1990) Composition and properties of hydrogen peroxide decomposing systems in extracellular and total extracts from needles of Norway spruce. Plant Physiol 94: 312–319
Polle A, Wieser G and Havranek WM (1995) Quantitification of ozone influx and apoplastic ascorbate content in needles of Norway spruce trees (Picea abies L., Karst) at high altitude. Plant Cell Environ 18: 681–688
Ranieri A, D'Urso G, Nali C, Lorenzini G and Soldatini GF (1996) Ozone stimulates apoplastic antioxidant systems in pumpkin leaves. Physiol Plant 97: 381–387
Rao MV, Hale BA and Ormrod DP (1995) Amelioration of ozoneinduced oxidative damage in wheat plants grown under high carbon dioxide. Plant Physiol 109: 421–432
Robinson JM (1997) The influence of elevated carbohydrate levels on the ascorbate to dehydroascorbate redox ratios in nitrogen limited spinach and soybean plants. Int J Plant Sci 158 (4): 442–450
Robinson JM and Bunce JA (2000) Influence of droughtinduced water stress on soybean and spinach leaf ascorbatedehydroascorbate level and redox status. Int J Plant Sci 161: 271–279
Robinson JM and Britz SJ (2001) Ascorbate-dehydroascorbate level and redox status in leaflets of field grown soybean plants exposed to elevated ozone. Int J Plant Sci 162(1) (in press)
Robinson JM, Britz SJ, Rowland RA and Stracke WF (1998) Influence of chronic ozonation on ascorbate accumulation and the ascorbate/dehydroascorbate redox status in leaflets of field grown soybean cultivars which demonstrate tolerance or sensitivity to ozone. In: De Kok LJ and Stulen I (eds) Responses of Plant Metabolism to Air Pollution and Global Change, pp 439–442. Backhuys Publishers, Leiden, The Netherlands
Smirnoff N (1996) The function and metabolism of ascorbic acid in plants. Ann Bot 78: 661–669
Wheeler GL, Jones MA and Smirnoff N (1998) The biosynthetic pathway of vitamin C in higher plants. Nature 393: 365–369
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Michael Robinson, J., Britz, S.J. Tolerance of a field grown soybean cultivar to elevated ozone level is concurrent with higher leaflet ascorbic acid level, higher ascorbate-dehydroascorbate redox status, and long term photosynthetic productivity. Photosynthesis Research 64, 77–87 (2000). https://doi.org/10.1023/A:1026508227189
Issue Date:
DOI: https://doi.org/10.1023/A:1026508227189