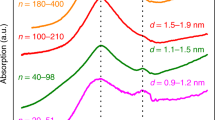
Abstract
In this paper we overview our recent studies of anisotropic noble metal (e.g. gold and silver) nanoparticles, in which a combination of theory and experiment has been used to elucidate the extinction spectra of the particles, as well as information related to their surface enhanced Raman spectroscopy. We used wet-chemical methods to generate several structurally well-defined nanostructures other than solid spheres, including silver nanodisks and triangular nanoprisms, and gold nanoshells and multipods. When solid spheres are transformed into one of these shapes, the surface plasmon resonances in these particles are strongly affected, typically red-shifting and even splitting into distinctive dipole and quadrupole plasmon modes. In parallel, we have developed computational electrodynamics methods based on the discrete dipole approximation (DDA) method to determine the origins of these intriguing optical features. This has resulted in considerable insight concerning the variation of plasmon wavelength with nanoparticle size, shape and dielectric environment, as well as the use of these particles for optical sensing applications.
Similar content being viewed by others
REFERENCES
A. P. Alivisatos (1996). Perspectives on the physical chemistry of semiconductor nanocrystals. J. Phys. Chem. 100, 13226-13239.
B. O. Dabbousi, J. Rodriguez-Viejo, F. V. Mikulec, J. R. Heine, H. Mattoussi, R. Ober, K. F. Jensen, and M. G. Bawendi (1997). J. Phys. Chem. B 101, 9463-9475.
M. A. Hines and P. Guyot-Sionnest (1996). J. Phys. Chem. 100, 468-471.
R. Jin, Y. Cao, C. A. Mirkin, K. L. Kelly, G. C. Schatz, and J. G. Zheng (2001). Science 294, 1901-1903.
K. L. Kelly, E. Coronado, L. L. Zhao, and G. C. Schatz (2003). J. Phys. Chem. B 107, 668-677.
S. Link and M. A. El-Sayed (1999). J. Phys. Chem. B 103, 8410-8426.
C. B. Murray, D. J. Norris, and M. G. Bawendi (1993). J. Am. Chem. Soc. 115, 8706-8715.
X. Peng, M. C. Schlamp, A. V. Kadavanich, and A. P. Alivisatos (1997). J. Am. Chem. Soc. 119, 7019-7029.
M. Gao, S. Kirstein, H. Möhwald, A. L. Rogach, A. Kornowski, A. Eychmuller, and H. Weller (1998). J. Phys. Chem. B 102, 8360-8363.
A. C. Templeton, W. P. Wuelfing, and R. W. Murray (2000). Acc. Chem. Res. 33, 27-36.
B. D. Busbee, S. O. Obare, and C. J. Murphy (2003). Adv. Mater. 15, 414-416.
S.-S. Chang, C.-W. Shih, C.-D. Chen, W.-C. Lai, and C. R. C. Wang (1999). Langmuir 15, 701-709.
A. Chemseddine and T. Moritz (1999). Eur. Inorg. Chem. 235-245.
N. R. Jana, L. Gearheart, and C. J. Murphy (2001). Chem. Commun., 617-618.
F. Kim, J. H. Song, and P. Yang (2002). J. Am. Chem. Soc. 124, 14316-14317.
Z. Tang, N. A. Kotov, and M. Giersig (2002). Science 297, 237-240.
Y. N. Xia, P. D. Yang, Y. G. Sun, Y. Y. Wu, B. Mayers, B. Gates, Y. D. Yin, F. Kim, and Y. Q. Yan (2003). Adv. Mater. 15, 353-389.
Y.-Y. Yu, S.-S. Chang, C.-L. Lee, and C. R. C. Wang (1997). J. Phys. Chem. B 101, 6661-6664.
S. Chen, Z. Fan, and D. L. Carroll (2002). J. Phys. Chem. B 106, 10777-10781.
E. Hao, K. L. Kelly, J. T. Hupp, and G. C. Schatz (2002). J. Am. Chem. Soc. 124, 15182-15183.
M. Maillard, S. Giorgio, and M.-P. Pileni (2002). Adv. Mater. 14, 1084-1086.
V. F. Puntes, D. Zanchet, C. K. Erdonmez, and A. P. Alivisatos (2002). J. Am. Chem. Soc. 124, 12874-12880.
M. Maillard, P. Huang, and L. Brus (2003). Nano Lett. 3, 1611-1615.
S. Chen and D. L. Carroll (2002). Nano Lett. 2, 1003-1007.
I. Pastoriza-Santos and L. M. Liz-Marzán (2002). Nano Lett. 2, 903-905.
Y. Sun, B. Mayers, and Y. Xia (2003). Nano Lett. 3, 675-679.
J. Yang and J. H. Fendler (1995). J. Phys. Chem 99, 5505-5511.
R. Jin, Y. Cao, E. Hao, G. Metraux, G. C. Schatz, and C. A. Mirkin (2003). Nature 425, 487-490.
S.-M. Lee, Y.-W. Jun, S. N. Cho, and J. Cheon (2002). J. Am. Chem. Soc. 124, 11244-11245
S. Chen, Z. L. Wang, J. Ballato, S. H. Foulger, and D. L., Carroll (2003). J. Am. Chem. Soc. 125, 16186-16187.
L. Manna, D. J. Milliron, A. Meisel, E. C. Scher, and A. P. Alivisatos (2003). Nature Mater. 2, 382-385.
L. Manna, E. C. Scher, and A. P. Alivisatos (2000). J. Am. Chem. Soc. 122, 12700-12706.
E. Hao, R. C. Bailey, G. C. Schatz, J. T. Hupp, and S. Li (2004). Nano Lett. 4, 327-330.
Y. Sun and Y. Xia (2002). Science 298, 2139-2141.
T. S. Ahmadi, Z. L. Wang, T. C. Green, A. Henglein, and M. A. El-Sayed (1996). Science 272, 1924-1926.
S. J. Oldenburg, J. B. Jackson, S. L. Westcott, and N. J. Halas (1999). Appl. Phys. Lett. 75, 2897-2899.
J. B. Jackson and N. J. Halas (2001). J. Phys. Chem. B 105, 2743-2746.
C. Graf and A. v. Blaaderen (2002). Langmuir 18, 524-534.
E. Pordan and P. Nordlander (2003). Nano Lett. 3, 543-547.
S. J. Oldenburg, R. D. Averitt, S. L. Westcott, and N. J. Halas (1998). Chem. Phys. Lett. 288, 243-247.
Y. Jin and S. Dong (2003). J. Phys. Chem. B 107, 12902-12905.
M. B. Mohamed, V. Volkov, S. Link, and M. A. El-Sayed (2000). Chem. Phys. Lett. 317, 517-523.
X. Peng, L. Manna, W. Yang, J. Wickham, E. Scher, A. Kadavanich, and A. P. Alivisatos (2000). Nature 404, 59-61.
W. U. Huynh, J. J. Dittmer, and A. P. Alivisatos (2002). Science 295, 2425-2427.
Y. Sun and Y. Xia (2002). Anal. Chem. 74, 5297-5305.
B. Nikoobakht, J. Wang, and M. A. El-Sayed (2002). Chem. Phys. Lett. 366, 17-23.
E. Hao and G. C. Schatz (2004). J. Chem. Phys. 120, 357-366.
Y. A. Sun and Y. N. Xia (2003). Adv. Mater. 15, 695-699.
G. S. Metraux, Y. C. Cao, R. C. Jin, and C. A. Mirkin (2003). Nano Lett. 3, 519-522.
S. O. Obare, N. R. Jana, and C. J. Murphy (2001). Nano Lett. 1, 601-603.
B. T. Draine and P. J. Flateau. User Guide for the Discrete Dipole Approximation DDSCAT.6.0, http://arxiv.org/abs/astro-ph0309069, with modifications described in W. H. Yang, G. C. 63 Schatz, and R. P. Van Duyne (1995). J. Chem. Phys. 103, 869-875.
T. Jensen, K. L. Kelly, A. Lazarides, and G. C. Schatz (1999). J. Cluster Sci. 10, 295-317.
K. L. Kelly, A. A. Lazarides, and G. C. Schatz (2001). Comput. Sci. Eng. 3, 67-73.
R. X. Bian, R. C. Dunn, X. S. Xie, and P. T. Leung (1995). Phys. Rev. Lett. 75, 4772-4775.
P. D. Cozzoli, A. Kornowski, and H. Weller (2003). J. Am. Chem. Soc. 125, 14539-14548.
S.-J. Park, S. Kim, S. Lee, Z. G. Khim, K. Char, and T. Hyeon (2000). J. Am. Chem. Soc. 122, 8581-8582.
E. Hao, S. Li, R. C. Bailey, G. C. Schatz, and J. T. Hupp (2004). J. Phys. Chem. B 108, 1224-1229.
K. L. Kelly, T. R. Jensen, A. A. Lazarides, and G. C. Schatz (2001). in D. Feldheim and C. Foss (Ed.), Metal Nanoparticles: Synthesis, Characterization and Applications, Marcel-Dekker, New York, pp. 89-118.
E. Coronado and G. C. Schatz (2003). J. Chem. Phys. 119, 3926-3934.
E. D. Palik (1985). Handbook of Optical Constants of Solids, Academic Press, New York.
W. O. Milligan and R. H. Morriss (1964). J. Am. Chem. Soc. 86, 3461-3467.
G. Mie (1908). Ann. Phys. 25, 377-445.
L. Charnary, A. Lee, S. Q. Man, C. E. Moran, C. Radloff, R. K. Bradley, and N. J. Halas (2003). J. Phys. Chem. B 107, 7327-7333.
G. C. Schatz (2001). J. Mol. Struc. (Theochem.) 573, 73-80.
G. C. Schatz (1984). Acc. Chem. Res. 17, 370-376.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Hao, E., Schatz, G.C. & Hupp, J.T. Synthesis and Optical Properties of Anisotropic Metal Nanoparticles. Journal of Fluorescence 14, 331–341 (2004). https://doi.org/10.1023/B:JOFL.0000031815.71450.74
Issue Date:
DOI: https://doi.org/10.1023/B:JOFL.0000031815.71450.74