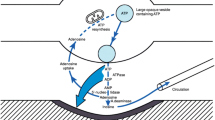
Abstract
Adenosine triphosphate (ATP) has a fundamental intracellular role as the universal source of energy for all living cells. The demonstration of its release into the extracellular space and the identification and localisation of specific receptors on target cells have been essential in establishing, after considerable resistance, its extracellular physiological roles. It is now generally accepted that ATP is a genuine neurotransmitter both in the central and peripheral nervous systems. As such, there are numerous arguments which prove that the release of ATP by nerve terminals is by exocytosis. In some non-neuronal cells, however, recent evidence suggests that ATP release could also be carrier-mediated and would involve ATP-binding cassette proteins (ABC), an ubiquitous family of transport ATPases.
Similar content being viewed by others
REFERENCES
Fiske, C. H. and Subbarow, Y. 1929. Phosphorus compounds of muscle and liver. Science 70:381–382.
Lohmann, K. 1929. Uber die Pyrophosphatfraktion im Muskel. Naturwissenschaften 17:624–625.
Sperlágh, B. and Vizi, E. S. 1996. Neuronal synthesis, storage and release of ATP. Semin. Neurosci. 8:175–186.
Kalckar, H. M. 1969. Biological Phosphorylations: Development of Concepts. Englewood Cliffs, NJ: Prentice Hall.
Lippman, F. 1941. Metabolic generation and utilization of phosphate bond energy. Enzymology 1:99.
Glynn, I. M. 1968. Membrane adenosine triphosphatase and cation transport. Br. Med. Bull. 24:165–169.
Chaudry, I. H. 1982. Does ATP cross the cell plasma membrane. Yale J. Biol. Med. 55:1–10.
Drury, A. N. and Szent-Györgyi, A. 1929. The physiological activity of adenine compounds with special reference to their action upon the mammalian heart. J. Physiol. (Lond) 68:213–237.
Bennett, D. W. and Drury, A. N. 1931. Further observations relating to the physiological activity of adenine compounds. J. Physiol. (Lond) 72:288–320.
Burnstock, G. 1972. Purinergic nerves. Pharmacol. Rev. 24:509–581.
Burnstock, G. 1997. The past, present and future of purine nucleotides as signalling molecules. Neuropharmacol. 36:1127–1139.
Lindler, F. and Rigler, R. 1931. Uber die Beeinflussung der Weite Der Herzkranzgefasse durch Produkte des Zellkernstoffwechsels. Pflugers Arch. 226:697–708.
Hoffman, W. S. 1930. The isolation of crystalline adenine nucleotide from blood. J. Biol. Chem. 63:675.
Drury, A. N. 1936. The physiological activity of nucleic acid and its derivatives. Physiol. Rev. 16:292–325.
Moir, T. W. and Downs, T. D. 1972. Myocardial reactive hyperemia: Comparative effects of adenosine, ATP, ADP, and AMP. Am. J. Physiol. 222:1386–1390.
Burnstock, G. 1978. A basis for distinguishing two types of purinergic receptor. In: Straub, R. W., Bolis, L., eds. Cell Membrane Receptors for Drugs and Hormones: A Multidisciplinary Approach. New York: Raven Press. p. 107–118.
Abbracchio, M. P. and Burnstock, G. 1994. Purinoceptors: Are there families of P2X and P2Y purinoceptors? Pharmacol. Ther. 64:445–475.
Burnstock, G. and Kennedy, C. 1985. Is there a basis for distinguishing two types of P2-purinoceptor? Gen. Pharmacol. 16:433–440.
Ralevic, V. and Burnstock, G. 1998. Receptors for purines and pyrimidines. Pharmacol. Rev. 50:413–492.
Green, H. N. and Stoner, H. B. 1950. Biological Actions of the Adenine Nucleotides. London: Lewis.
Trams, E. J., Kauffman, H., and Burnstock, G. 1980. A proposal for the role of ecto-enzymes and adenylates in traumatic shock. J. Theor. Biol. 87:609–621.
Katsuragi, T., Tokunaga, T., Ogawa, S., Soejima, O., Sato C., and Furukawa T. 1991. Existence of ATP-evoked ATP release system in smooth muscles. J. Pharmacol. Exp. Ther. 259:513–518.
Bodin, P. and Burnstock, G. 1996. ATP-stimulated release of ATP by human endothelial cells. J. Cardiovasc. Pharmacol. 27:872–875.
Vizi, E. S. and Burnstock, G. 1988. Origin of ATP release in the rat vas deferens: Concomitant measurement of [3H]noradrenaline and [14C]ATP. Eur. J. Pharmacol. 158:69–77.
Sperlágh, B. and Vizi, E. S. 1991. Effect of presynaptic P2 receptor stimulation on transmitter release. J. Neurochem. 56:1466–1470.
Vizi, E. S., Liang, S. D., Sperlágh, B., Kittel, A., and Juranyi, Z. 1997. Studies on the release and extracellular metabolism of endogenous ATP in rat superior cervical ganglion: Support for neurotransmitter role of ATP. Neuroscience 79:893–903.
Vizi, E. S., Sperlágh, B., and Baranyi, M. 1992. Evidence that ATP released from the postsynaptic site by noradrenaline, is involved in mechanical responses of guinea-pig vas deferens: Cascade transmission. Neuroscience 50:455–465.
Holton, F. A. and Holton, P. 1954. The capillary dilator substances in dry powders of spinal roots: A possible role of adenosine triphosphate in chemical transmission from nerve endings. J. Physiol. (Lond.) 126:124–140.
Holton, P. 1959. The liberation of adenosine triphosphate on antidromic stimulation of sensory nerves. J. Physiol. (Lond.) 145:494–504.
Abood, L. G., Koketsu, K., and Miyamoto, S. 1962. Outflux of various phosphates during membranedepolarisation of excitable tissues. Am. J. Physiol. 202:469–474.
Burnstock, G., Campbell, G., Bennett, M., and Holman, M. E. 1963. The effects of drugs on the transmission of inhibition from autonomic nerves to the smooth muscle of the guinea pig taenia coli. Biochem. Pharmacol. 12 (Suppl.):134–135.
Burnstock, G., Campbell, G., Satchell, D., and Smythe, A. 1970. Evidence that adenosine triphosphate or a related nucleotide is the transmitter substance released by nonadrenergic inhibitory nerves in the gut. Br. J. Pharmacol. 40:668–688.
Burnstock, G. 1971. Neural nomenclature. Nature 229:282–283.
Burnstock, G. 1976. Do some nerve cells release more than one transmitter? Neuroscience 1:239–248.
Su, C., Bevan, J. A., and Burnstock, G. 1971. [3H]adenosine triphosphate: Release during stimulation of enteric nerves. Science 173:337–339.
Burnstock, G., Cocks, T., Kasakov, L., and Wong, H. K. 1978. Direct evidence for ATP release from non-adrenergic, noncholinergic (“purinergic”) nerves in the guinea-pig taenia coli and bladder. Eur. J. Pharmacol. 49:145–149.
Westfall, D. P., Stitzel, R. E., and Rowe, J. N. 1978. The postjunctional effects and neural release of purine compounds in the guinea-pig vas deferens. Eur. J. Pharmacol. 50:27–38.
Sneddon, P. and Burnstock, G. 1984. ATP as a co-transmitter in rat tail artery. Eur. J. Pharmacol. 106:149–152.
Sedaa, K. O., Bjur, R. A., Shinozuka, K., and Westfall, D. P. 1990. Nerve and drug-induced release of adenine nucleosides and nucleotides from rabbit aorta. J. Pharmacol. Exp. Ther. 252:1060–1067.
Burnstock, G. 1975. Purinergic transmission. In: Iversen, L. I., Iversen, S. D., and Snyder, S. H., eds. Handbook of Psychopharmacology, Vol. 5. New York: Plenum Press. p 131–194.
Westfall, D. P., Hogaboom, G. K., Colby, J., O'Donnell, J. P., and Fedan, J. S. 1982. Direct evidence against a role of ATP as the nonadrenergic, noncholinergic inhibitory neurotransmitter in guinea pig taenia coli. Proc. Natl. Acad. Sci. USA 79:7041–7045.
Burnstock, G. 1997. History of extracellular nucleotides and their receptors. In: Turner, J. T., Weisman, G., and Fedan, J. S., eds. The P2 Nucleotide Receptors. Totowa, NJ: Humana Press Inc. pp. 3–40.
Silinsky, E. M., Gerzanich, V., and Vanner, S. M. 1992. ATP mediates excitatory synaptic transmission in mammalian neurones. Br. J. Pharmacol. 106:762–763.
Burnstock, G. 1999. Purinergic cotransmission. Brain. Res. Bull. 50:355–357.
Hoyle, C. H. V. 1996. Purinergic cotransmission: Parasympathetic and enteric nerves. Semin. Neuroscience 8:207–215.
McConalogue, K. and Furness, J. B. 1994. Gastrointestinal neurotransmitters. Baillieres Clin. Endocrinol. Metab. 8:51–76.
White, T. D. 1977. Direct detection of depolarisation-induced release of ATP from a synaptosomal preparation. Nature 267:67–68.
Potter, P. and White, T. D. 1980. Release of adenosine 5′-triphosphate from synaptosomes from different regions of rat brain. Neuroscience 5:1351–1356.
Edwards, F. A., Gibb, A. J., and Colquhoun, D. 1992. ATP receptor-mediated synaptic currents in the central nervous system. Nature 359:144–147.
Sperlágh, B., Kittel, A., Lajtha, A., and Vizi, E. S. 1995. ATP acts as fast neurotransmitter in rat habenula: Neurochemical and enzymecytochemical evidence. Neuroscience 66:915–920.
Burnstock, G. 1999. Current status of purinergic signalling in the nervous system. Prog. Brain. Res. 120:3–10.
Wu, P. H. and Phillis, J. W. 1978. Distribution and release of adenosine triphosphate in rat brain. Neurochem. Res. 3:563–571.
Phillis, J. W. and Wu, P. H. 1981. The role of adenosine and its nucleotides in central synaptic transmission. Prog. Neurobiol. 16:187–239.
Sweeney, M. I., White, T. D., and Sawynok, J. 1989. Morphine, capsaicin and K+ release purines from capsaicin-sensitive primary afferent nerve terminals in the spinal cord. J. Pharmacol. Exp. Ther. 248:447–454.
Burnstock, G. 1996. A unifying purinergic hypothesis for the initiation of pain. Lancet 347:1604–1605.
Silinsky, E. M. and Hubbard, J. I. 1973. Release of ATP from rat motor nerve terminals. Nature 243:404–405.
Forrester, T. and Lind, A. R. 1969. Identification of adenosine triphosphate in human plasma and the concentration in the venous effluent of forearm muscles before, during and after sustained contractions. J. Physiol. 204:347–364.
Paddle, B. M. and Burnstock, G. 1974. Release of ATP from perfused heart during coronary vasodilatation. Blood Vessels 11:110–119.
Detwiler, T. C. and Feinman, R. D. 1973. Kinetics of the thrombin-induced release of adenosine triphosphate by platelets. Comparison with release of calcium. Biochemistry 12:2462–2468.
Bergfeld, G. R. and Forrester, T. 1989. Efflux of adenosine triphosphate from human erythrocytes in response to a brief pulse of hypoxia. Proc. Physiol. Soc. 418:88.
Maugeri, N., Bermejo, E., and Lazzari, M. A. 1990. Adenosine triphosphate released from human mononuclear cells. Thromb. Res. 59:887–890.
Pearson, J. D. and Gordon, J. L. 1979. Vascular endothelial and smooth muscle cells in culture selectively release adenine nucleotides. Nature 281:384–386.
Bodin, P., Bailey, D. J., and Burnstock, G. 1991. Increased flow-induced ATP release from isolated vascular endothelial but not smooth muscle cells. Br. J. Pharmacol. 103:1203–1205.
Hazama, A., Hayashi, S., and Okada, Y. 1998. Cell surface measurements of ATP release from single pancreatic β cells using a novel biosensor technique. Pflugers Arch. 437:31–35.
Cotrina, M. L., Lin, J. H., Alves-Rodrigues, A., Liu, S., Li, J., Azmi-Ghadimi, H., Kang, J., Naus, C. C., and Nedergaard, M. 1998. Connexins regulate calcium signaling by controlling ATP release. Proc. Natl. Acad. Sci. USA 95:15735–15740.
Queiroz, G., Meyer, D. K., Meyer, A., Starke, K., and von Kügelgen, I. 1999. A study of the mechanism of the release of ATP from rat cortical astroglial cells evoked by activation of glutamate receptors. Neuroscience 91:1171–1181.
Lalevée, N., Rogier, C., Becq, F., and Joffre, M. 1999. Acute effects of adenosine triphosphates, cyclic 3′,5′-adenosine monophosphates, and follicle-stimulating hormone on cytosolic calcium level in cultured immature rat Sertoli cells. Biol. Reprod. 61:343–352.
Graff, R. D., Lazarowski, E. R., Banes, A. J., and Lee, G. M. 2000. ATP release by mechanically loaded porcine chondrons in pellet culture. Arthritis Rheum. 43:1571–1579.
Gabella, G. and Davis, C. 1998. Distribution of afferent axons in the bladder of rats. J. Neurocytol. 27:141–155.
Burnstock, G. 2000. P2X receptors in sensory neurones. Br. J. Anaesth. 84:476–488.
Burnstock, G. 1999. Release of vasoactive substances from endothelial cells by shear stress and purinergic mechanosensory transduction. J. Anat. 194:335–342.
Burnstock, G. 2001. Purine-mediated signalling in pain and visceral perception. Trends. Pharmacol. Sci. (in press).
Ferguson, D. R. 1999. Urothelial function. BJU Int. 84:235–242.
Cockayne, D. A., Hamilton, S. G., Zhu, Q.-M., Dunn, P. M., Zhong, Y., Novakovic, S., Malmberg, A. B., Cain, G., Berson, A., Kassotakis, L., Hedley, L., Lachnit, W. G., Burnstock, G., McMahon, S. B., and Ford A. P. D. W. 2000. Urinary bladder hyporeflexia and reduced pain-related behaviour in P2X3-deficient mice. Nature 407:1011–1015.
Marra, B. 1993. Intestinal occlusion due to a colonic lipoma. Apropos 2 cases [In Italian]. Minerva Chir. 48:1035–1039.
Shellenbarger, T. and Krouse, A. 1994. Treating and preventing kidney stones. Medsurg. Nurs. 3:389–394.
Berkley, K. J., Wood, E., Scofield, S. L., and Little, M. 1995. Behavioral responses to uterine or vaginal distension in the rat. Pain 61:121–131.
Dik, P., Lock, T. M., Schrier, B. P., Zeijlemaker, B. Y., and Boon, T. A. 1996. Transurethral marsupialization of a medial prostatic cyst in patients with prostatitis-like symptoms. J. Urol. 155:1301–1304.
Nicholl, J. P., Ross, B., Milner, P. C., Brazier, J. E., Westlake, L., Kohler, B., Frost, E., Williams, B. T., and Johnson, A. G. 1994. Cost effectiveness of adjuvant bile salt treatment in extracorporeal shock wave lithotripsy for the treatment of gall bladder stones. Gut. 35:1294–1300.
Ferguson, D. R., Kennedy, I., and Burton, T. J. 1997. ATP is released from rabbit urinary bladder epithelial cells by hydrostatic pressure changes—a possible sensory mechanism? J. Physiol. 505:503–511.
Vlaskovska, M., Kasakov, L., Rong, W., Bodin, P., Bardini, M., Cockayne, D. A., Ford, A. P. D. W., and Burnstock, G. 2001. P2X3 knockout mice reveal a major sensory role for urothelially released ATP. J. Neurosci. (in press)
Katz, B. 1969. The Release of Neural Transmitter Substances. Liverpool: University Press.
De Robertis, E. 1964. Histophysiology of Synapses and Neurosecretion. Oxford, UK: Pergammon.
Dowdall, M. J., Boyne, A. F., and Whittaker, V. P. 1974. Adenosine triphosphate. A constituent of cholinergic synaptic vesicles. Biochem. J. 140:1–12.
Zimmermann, H. 1982. Co-existence of adenosine 5′-triphosphate and acetylcholine in the electromotor synapse. In: Cuello, A. C., ed. Co-transmission. London: MacMillan Press. pp. 243–259.
Zimmermann, H. and Denston, C. R. 1976. Adenosine triphosphate in cholinergic vesicles isolated from the electric organ of Electrophorus electricus. Brain Res. 111:365–376.
Chen, T. K., Luo, G., and Ewing, A. G. 1994. Amperometric monitoring of stimulated catecholamine release from rat pheochromocytoma (PC12) cells at the zeptomole level. Anal. Chem. 66:3031–3035.
Nagasawa, J. 1977. Exocytosis: The common release mechanism of secretory granules in glandular cells, neurosecretory cells, neurons and paraneurons. Arch. Histol. Jpn. 40 Suppl:31–47.
Parnas, I. and Parnas, H. 1999. Different mechanisms control the amount and time course of neurotransmitter release. J. Physiol. (Lond.) 517:629.
Sikorski, A. F., Sangerman, J., Goodman, S. R., and Critz, S. D. 2000. Spectrin (βSpllΣ1) is an essential component of synaptic transmission. Brain Res. 852:161–166.
White, T. D. 1978. Release of ATP from a synaptosomal preparation by elevated extracellular K+ and by veratridine. J. Neurochem. 30:329–336.
Salto, C., Calvet, R., Guitart, X., Solsona, C., and Marsal, J. 1990. Opiates depress ACh and ATP release from cholinergic synaptosomes by blocking calcium uptake. Toxicol. Appl. Pharmacol. 106:20–27.
White, T. D. 1984. Characteristics of neuronal release of ATP. Prog. Neuropsychopharmacol. Biol. Psychiatry 8:487–493.
Fredholm, B. B. and Hedqvist, P. 1978. Release of 3H-purines from [3H]-adenine labelled rabbit kidney following sympathetic nerve stimulation, and its inhibition by α-adrenoceptor blockage. Br. J. Pharmacol. 64:239–245.
Irvin, J. L. and Irvin, E. M. 1954. The interaction of quinacrine with adenine nucleotides. J. Biol. Chem. 210:45–56.
Ålund, M. and Olson, L. 1979. Depolarization-induced decreases in fluroescence intensity of gastro-intestinal quinacrine-binding nerves. Brain Res. 166:121–137.
White, P. N., Thorne, P. R., Housley, G. D., Mockett, B., Billett, T. E., and Burnstock, G. 1995. Quinacrine staining of marginal cells in the stria vascularis of the guinea-pig cochlea: A possible source of extracellular ATP? Hear. Res. 90:97–105.
Olson, L., Ålund, M., and Nordberg, K. A. 1976. Fluorescence-microscopical demonstration of a population of gastrointestinal nerve fibres with a selective affinity for quinacrine. Cell Tissue Res. 171:407–423.
Knight, G. E., Hoyle, C. H. V., and Burnstock, G. 1992. Quinacrine-staining of neurones, and activity of purine nucleosides and nucleotides in marine and terrestrial invertebrates from several phyla. Comp. Biochem. Physiol. 102C:305–314.
Mitchell, C. H., Carrè, D. A., McGlinn, A. M., Stone, R. A., and Civan, M. M. 1998. A release mechanism for stored ATP in ocular ciliary epithelial cells. Proc. Natl. Acad. Sci. USA 95:7174–7178.
Douglas, W. W. and Poisner, A. M. 1966. Evidence that the secreting adrenal chromaffin cell releases catecholamines directly from ATP-rich granules. J. Physiol. (Lond.) 183:236–248.
Johnson, R. G. Jr. 1987. Proton pumps and chemiosmotic coupling as a generalized mechanism for neurotransmitter and hormone transport. Ann. NY Acad. Sci. 493:162–177.
Silinsky, E. M. 1975. On the association between transmitter secretion and the release of adenine nucleotides from mammalian motor nerve terminals. J. Physiol. 247:145–162.
Silinsky, E. M. and Redman, R. S. 1996. Synchronous release of ATP and neurotransmitter within milliseconds of a motor nerve impulse in the frog. J. Physiol. (Lond.) 492:815–822.
Lagercrantz, H. and Stjärne, L. 1974. Evidence that most noradrenaline is stored without ATP in sympathetic large dense core nerve vesicles. Nature 249:843–845.
Burnstock, G. 1995. Noradrenaline and ATP: Cotransmitters and neuromodulators. J. Physiol. Pharmacol. 46:365–384.
Hammond, J. R., MacDonald, W. F., and White, T. D. 1988. Evoked secretion of [3H]noradrenaline and ATP from nerve varicosities isolated from the myenteric plexus of the guinea pig ileum. Can. J. Physiol. Pharmacol. 66:369–375.
Kasakov, L., Ellis, J., Kirkpatrick, K., Milner, P., and Burnstock, G. 1988. Direct evidence for concomitant release of noradrenaline, adenosine 5′-triphosphate and neuropeptide Y from sympathetic nerve supplying the guinea-pig vas deferens. J. Auton. Nerv. Syst. 22:75–82.
Santos, P. F., Caramelo, O. L., Carvalho, A. P., and Duarte, C. B. 1999. Characterization of ATP release from cultures enriched in cholinergic amacrine-like neurons. J. Neurobiol. 41:340–348.
White, T. D. and Leslie, R. A. 1982. Depolarization-induced release of adenosine 5′-triphosphate from isolated varicosities derived from the myenteric plexus of the guinea-pig small intestine. J. Neurosci. 2:206–215.
Burnstock, G. 1976. Purinergic receptors. J. Theor. Biol. 62:491–503.
Hollins, B. and Ikeda, S. R. 1997. Heterologous expression of a P2x-purinoceptor in rat chromaffin cells detects vesicular ATP release. J. Neurophysiol. 78:3069–3076.
Bodin, P. and Burnstock, G. 1998. Increased release of ATP from endothelial cells during acute inflammation. Inflamm. Res. 47:351–354.
Bodin, P. and Burnstock, G. 2001. Evidence that release of ATP from endothelial cells during increased shear stress is vesicular. J. Cardiovasc. Pharmacol. (in press).
Elgavish, A. and Elgavish, G. A. 1985. Evidence for the presence of an ATP transport system in brush-border membrane vesicles isolated from the kidney cortex. Biochim. Biophys. Acta 812:595–599.
Reisin, I. L., Prat, A. G., Abraham, E. H., Amara, J. F., Gregory, R. J., Ausiello, D. A., and Cantiello, H. F. 1994. The cystic fibrosis transmembrane conductance regulator is a dual ATP and chloride channel. J. Biol. Chem. 269:20584–20591.
Schwiebert, E. M. 1999. ABC transporter-facilitated ATP conductive transport. Am. J. Physiol. 276:C1–C8.
Demolombe, S. and Escande, D. 1996. ATP-binding cassette proteins as targets for drug discovery. Trends Pharmacol. Sci. 17:273–275.
Higgins, C. F., Gallagher, M. P., Mimmack, M. L., and Pearce, S. R. 1988. A family of closely related ATP-binding subunits from prokaryotic and eukaryotic cells. Bioessays 8:111–116.
Saurin, W., Hofnung, M., and Dassa, E. 1999. Getting in or out: Early segregation between importers and exporters in the evolution of ATP-binding cassette (ABC) transporters. J. Mol. Evol. 48:22–41.
Abraham, E. H., Prat, A. G., Gerweck, L., Seneveratne, T., Arceci, R. J., Kramer, R., Guidotti, G., and Cantiello, H. F. 1993. The multidrug resistance (mdr1) gene product functions as an ATP channel. Proc. Natl. Acad. Sci. USA 90:312–316.
Schwiebert, E. M., Egan, M. E., Hwang, T. H., Fulmer, S. B., Allen, S. S., Cutting, G. R., and Guggino, W. B. 1995. CFTR regulates outwardly rectifying chloride channels through an autocrine mechanism involving ATP. Cell 81:1063–1073.
Li, C., Ramjeesingh, M., and Bear, C. E. 1996. Purified cystic fibrosis transmembrane conductance regulator (CFTR) does not function as an ATP channel. J. Biol. Chem. 271:11623–11626.
Wang, Y., Roman, R., Lidofsky, S. D., and Fitz, J. G. 1996. Autocrine signaling through ATP release represents a novel mechanism for cell volume regulation. Proc. Natl. Acad. Sci. USA 93:12020–12025.
Hazama, A., Fan, H. T., Abdullaev, I., Maeno, E., Tanaka, S., Ando, A. Y., and Okada, Y. 2000. Swelling-activated, cystic fibrosis transmembrane conductance regulator-augmented ATP release and Cl- conductances in murine C127 cells. J. Physiol. (Lond.) 523:1–11.
Guidotti, G. 1996. ATP transport and ABC proteins. Chem. Biol. 3:703–706.
Sugita, M., Yue, Y., and Foskett, J. K. 1998. CFTR Cl- channel and CFTR-associated ATP channel: Distinct pores regulated by common gates. EMBO J. 17:898–908.
Zimmermann, H. and Braun, N. 1999. Ecto-nucleotidases— molecular structures, catalytic properties, and functional roles in the nervous system. Prog. Brain. Res. 120:371–385.
Yegutkin, G., Bodin, P., and Burnstock, G. 2000. Effect of shear stress on the release of soluble ecto-enzymes ATPase and 5′-nucleotidase along with endogenous ATP from vascular endothelial cells. Br. J. Pharmacol 129:921–926.
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Bodin, P., Burnstock, G. Purinergic Signalling: ATP Release. Neurochem Res 26, 959–969 (2001). https://doi.org/10.1023/A:1012388618693
Issue Date:
DOI: https://doi.org/10.1023/A:1012388618693