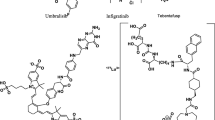
Abstract
In this review, we discuss all sulfur-containing FDA-approved drugs and their structures. The second section of the review is dedicated to structural analysis and is divided into 14 subsections, each focusing on one type of sulfur-containing moiety. A concise graphical representation of each class features drugs that are organized on the basis of structural similarity, evolutionary relevance, and medical indication. This review offers a unique and comprehensive overview of the structural features of all sulfur-containing FDA-approved drugs to date.


























Similar content being viewed by others
Abbreviations
- 6-APA:
-
6-Aminopenicillanic acid
- ADP:
-
Adenosine diphosphate
- cGMP:
-
Cyclic guanosine monophosphate
- COX-2:
-
Cyclooxygenase-2
- GERD:
-
Gastroesophageal reflux disease
- GPCR:
-
G protein-coupled receptor
- HIV:
-
Human immunodeficiency virus
- mRNA:
-
Messenger RNA
- NSAID:
-
Nonsteroidal anti-inflammatory drugs
- SMN1:
-
Survival motor neuron 1
- SMN2:
-
Survival motor neuron 2
- PBP-3:
-
Penicillin binding protein 3
- P2Y12 :
-
A type of purinergic receptor
- PDE5:
-
Phosphodiesterase type 5
- PET:
-
Positron emission tomography
- PPI:
-
Proton pump inhibitor
- US FDA:
-
United States Food and Drug Administration
References
Ilardi EA, Vitaku E, Njardarson JT (2014) J Med Chem 57:2832–2842
Sprague JM (1958) Ann NY Acad Sci 71:328–342
Maren TH (1976) Annu Rev Pharmacol Toxicol 16:309–327
Fitzgerald D (2005) Dialogues Cardiovasc Med 10:175–182
Mulder H, Shopman W Sr, van der Lely AJ (1991) Eur J Clin Pharmacol 1991(40):379–381
Meyer M, Chudziak F, Schwanstecher C et al (1999) Br J Pharmacol 128:27–34
Inagaki N, Gonoi T, Clement JP et al (1996) Neuron 16:1011–1017
Tréfouël JT, Nitti F, Bovet D (1935) CR Soc Biol 120:756
Seydel JK (1968) J Pharm Sci 57:1455–1478
Lemke TL, Williams DA, Roche VF, Zito SW (2013) Foye’s medicinal chemistry, 7th edn. Lippincott Williams & Wilkins, Baltimore
Corbin JD, Beasley A, Blount MA, Francis SH (2004) Neurochem Int 45:859–863
Nagashima H (2002) J Biol Chem 277:50439–50449
Abraham EP, Newton GGF (1961) Biochem J 79:377–393
Long KS, Hansen LH, Jakobsen L, Vester B (2006) Antimicrob Agents Chemother 50:1458–1462
Oliphant CS, Doby JB, Das K (2010) Curr Vasc Pharmacol 8:93–101
Elion G (1989) Science 244:4900
Nag K, Singh DR, Shetti AN, Kumar H, Sivashanmugam T, Parthasarathy S (2013) Anesth Essays Res 7:302–306
Corey EJ, Gin DY, Kania RS (1996) J Am Chem Soc 38:9202–9203
Blanchet PJ (1999) Can J Neurol Sci 26(Suppl 2):S21–S26
Horvath J, Fross RD, Kleiner-Fisman G et al (2004) Mov Disord 19:656–662
Chang W, Weber M, Breier MR, Saint Marie RL, Hines SR, Swerdlow NR (2012) Brain Res 1437:69–76
Markowitz M, Saag M, Powderly WG et al (1995) N Eng J Med 333:1534–1539
Humphries TJ, Merritt GJ (1999) Aliment Pharmacol Ther 13:18–26
Miller RG, Mitchell JD, Moore DH (2012) Cochrane Database Syst Rev 3:CD001447
Ding Z, Kim S, Dorsam RT et al (2003) Blood 101:3908–3914
Wallentin L (2009) Eur Heart J 30:1964–1977
Horn AS, Snyder SH (1971) Proc Natl Acad Sci 68:2325–2328
Massie SP (1954) Chem Rev 54:797–833
Shen WW (1999) Compr Psychiatry 40:407
Binchi MT (2010) Med Hypotheses 74(2):297–300
Bovet D, Stauo AM (1937) CR Biol 124:547–549
Delay J, Deniker P, Harl J (1952) Ann Med Psychol 110:112–117
Zhang P, Li J, Han Y et al (2010) Rheumatol Int 30:713–718
Andersson T, Weidolf L (2008) Clin Drug Investig 28:263–279
Agranat I, Caner H, Caldwell J (2002) Nat Rev Drug Discov 1:753–768
Shin JM, Cho YM, Sachs G (2004) J Am Chem Soc 12:7800–7811
Besancon M, Simon A, Sachs G, Shin JM (1997) J Biol Chem 272:22438–22446
Coulthard SA, Hogarth LA, Little M et al (2002) Mol Pharmacol 62:1
Cresciolo C, Cosmi L, Borgogni E et al (2007) J Endocrinol 195:145–155
Kim TH, Jeong JW, Lee KR, Ahn S, Kim S, Koo TS (2015) Arch Pharm Res 38:2076–2082
Taurog A (1976) Endocrinology 98:1031–1046
Jacobsen E, Larsen V (1949) Acta Pharmacol Toxicol 5:285–291
Bieder A, Brunel P, Mazeau L (1966) Ann Pharm France 24:493–500
Caceres NE, Harris NB, Wellehan JF et al (1997) J Bacteriol 179:5046–5055
Ijaz N, Fitzgerald D (2017) Br J Hosp Med 2:C88–C91
Steudel R, Laitinen R (1982) Top Curr Chem 102:177–197
Karr S (2017) Am J Manag Care 23:S139–S1478
Ottesen EW (2017) Transl Neurosci 8:1–6
Seeling A, Oelschläger H, Rothley D (2000) Pharmazie 55:293–296
Diener HC, Limmroth V (1999) Opin Neurol 12:261–267
Medeiros GF, Mendes A, Castro RAB, Baú EC, Nader B, Dietrich CP (2000) Biochim Biophys Acta Gen Subj 1475:287–294
Ueda H, Nakajima H, Horim Y et al (1994) J Antibiot 3:301–310
Li KW, Wu J, Simon JA (1994) J Am Chem Soc 118:7237–7238
Kvols L, Woltering E (2006) Anticancer Drugs 17:601–608
Plosker GL (2015) Drugs 75:1255–1268
Aoyagi Y, Katano K, Suguna H, Prmeau J, Chang LH, Hecht SM (1982) J Am Chem Soc 104:5537–5538
Bonadonna G, De Lena M, Monfardini S, Bartoli C, Bajetta E, Beretta G, Fossati-Bellani F (1972) Eur J Cancer 8:205–215
Koukourakis MI, Maltezos E (2006) Anticancer Drugs 17:2
Gahl WA, Thoene JG, Schneider JA (2002) N Eng J Med 347:111–121
Imada A, Kitano K, Kintaka K et al (1981) Nature 289:590–591
Hilton GF, Das T, Majji S (1996) Indian J Ophthamol 44:131–143
Sidharta PN, van Giersbergen PL, Halabi A, Dingemanse J (2011) Eur J Clin Pharmacol 67:977–984
Bang LM, Scott LJ (2003) Drugs 63:2413–2424
Busatto GF, Kerwin RW (1997) J Psychopharmachol 11:3–12
Hulin B, McCarthy PA, Gibbs EM (1996) Curr Pharm Des 2:85–102
Strohmaier K, Snyder E, Adamsons I (1998) J Am Optom Assoc 69:441–451
Kamen L, Henney HR, Runyan JD (2008) Curr Med Res Opin 24:425–439
Acknowledgements
The funding was provided by National Science Foundation (Grant no. CHE-1565500).
Author information
Authors and Affiliations
Corresponding author
Additional information
This article is part of the Topical Collection “Sulfur Chemistry”, edited by Xuefeng Jiang.
Rights and permissions
About this article
Cite this article
Scott, K.A., Njardarson, J.T. Analysis of US FDA-Approved Drugs Containing Sulfur Atoms. Top Curr Chem (Z) 376, 5 (2018). https://doi.org/10.1007/s41061-018-0184-5
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s41061-018-0184-5