Abstract
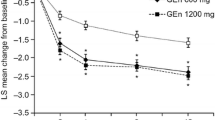
Gabapentin enacarbil is an extended-release prodrug of gabapentin that is approved in the USA (Horizant®) and Japan (Regnite®) for the treatment of moderate to severe primary restless legs syndrome (RLS) in adults [featured indication]. This article summarizes pharmacological, efficacy and tolerability data relevant to the use of oral gabapentin enacarbil in this indication. In double-blind, multicentre trials, treatment with gabapentin enacarbil 600 mg/day for 12 weeks significantly improved the symptoms of moderate to severe primary RLS in adults. Gabapentin enacarbil also significantly improved RLS pain scores and generally improved sleep and mood outcomes. These data are supported by retrospective pooled analyses of three of these trials (XP081, PIVOT RLS I and PIVOT RLS II), with gabapentin enacarbil generally improving symptoms irrespective of disease severity, associated sleep disturbance or prior dopamine agonist use. Responses to gabapentin enacarbil were sustained in longer-term trials, with lower relapse rates in gabapentin enacarbil than placebo recipients in a longer-term maintenance study. Overall, in short and longer-term trials, relatively few patients discontinued treatment, adverse events were mostly mild to moderate in severity, and somnolence/sedation and dizziness were the most commonly reported adverse events. Notably, there were no reports of augmentation or QT-interval prolongation. Gabapentin enacarbil is an important agent for the treatment of adults with moderate to severe primary RLS.
Similar content being viewed by others
References
Allen RP, Picchietti DL, Garcia-Borreguero D, et al. Restless legs syndrome/Willis-Ekbom disease diagnostic criteria: updated International Restless Legs Syndrome Study Group (IRLSSG) consensus criteria-history, rationale, description, and significance. Sleep Med. 2014;15(8):860–73.
Nagandla K, De S. Restless legs syndrome: pathophysiology and modern management. Postgrad Med J. 1053;2013(89):402–10.
Ohayon MM, O’Hara R, Vitiello MV. Epidemiology of restless legs syndrome: a synthesis of the literature. Sleep Med Rev. 2012;16(4):283–95.
Comella CL. Treatment of restless legs syndrome. NeuroTherapeutics. 2014;11(1):177–87.
Ondo WG. Restless legs syndrome: pathophysiology and treatment. Curr Treat Options Neurol. 2014;16(11):317.
Trenkwalder C, Winkelmann J, Inoue Y, et al. Restless legs syndrome-current therapies and management of augmentation. Nat Rev Neurol. 2015;11(8):434–45.
Rios Romenets S, Postuma RB. Treatment of restless legs syndrome. Curr Treat Options Neurol. 2013;15(4):396–409.
Garcia-Borreguero D, Silber MH, Winkelman JW, et al. Guidelines for the first-line treatment of restless legs syndrome/Willis-Ekbom disease, prevention and treatment of dopaminergic augmentation: a combined task force of the IRLSSG, EURLSSG, and the RLS-foundation. Sleep Med. 2016;21:1–11.
Scott LJ. Gabapentin enacarbil: in patients with restless legs syndrome. CNS Drugs. 2012;26(12):1073–83.
US FDA. Horizant (gabapentin enacarbil) extended-release tablets: US prescribing information. 2013. http://www.fda.gov. Accessed 26 Apr 2016.
Lal R, Sukbuntherng J, Luo W, et al. Population pharmacokinetics and pharmacodynamics of gabapentin after administration of gabapentin enacarbil. J Clin Pharmacol. 2013;53(1):29–40.
Davy M, Upward J, Arumugham T, et al. Cardiac repolarization with gabapentin enacarbil in a randomized, double-blind, placebo- and active-controlled, crossover thorough QT/QTc study in healthy adults. Clin Ther. 2013;35(12):1964–74.
Chen D, Lal R, Zomorodi K, et al. Evaluation of gabapentin enacarbil on cardiac repolarization: a randomized, double-blind, placebo- and active-controlled, crossover thorough QT/QTc study in healthy adults. Clin Ther. 2012;34(2):351–62.e3.
Lal R, Sukbuntherng J, Luo W, et al. Pharmacokinetics and tolerability of single escalating doses of gabapentin enacarbil: a randomized-sequence, double-blind, placebo-controlled crossover study in healthy volunteers. Clin Ther. 2009;31(8):1776–86.
Cundy KC, Branch R, Chernov-Rogan T, et al. XP13512 [(±)-1-([(α-isobutanoyloxyethoxy)carbonyl] aminomethyl)-1-cyclohexane acetic acid], a novel gabapentin prodrug: I. Design, synthesis, enzymatic conversion to gabapentin, and transport by intestinal solute transporters. J Pharmacol Exp Ther. 2004;311(1):315–23.
Cundy KC, Sastry S, Luo W, et al. Clinical pharmacokinetics of XP13512, a novel transported prodrug of gabapentin. J Clin Pharmacol. 2008;48(12):1378–88.
Lal R, Sukbuntherng J, Ho J, et al. A phase I, single-dose study of the disposition of 14C-radiolabeled gabapentin enacarbil in healthy male volunteers. Int J Clin Pharmacol Ther. 2011;49(2):109–15.
Lal R, Sukbuntherng J, Luo W, et al. Clinical pharmacokinetic drug interaction studies of gabapentin enacarbil, a novel transported prodrug of gabapentin, with naproxen and cimetidine. Br J Clin Pharmacol. 2010;69(5):498–507.
Chen C, Upward J, Arumugham T, et al. Gabapentin enacarbil and morphine administered in combination versus alone: a double-blind, randomized, pharmacokinetic, and tolerability comparison. Clin Ther. 2015;37(2):349–57.
Lal R, Ellenbogen A, Chen D, et al. A randomized, double-blind, placebo-controlled, dose-response study to assess the pharmacokinetics, efficacy, and safety of gabapentin enacarbil in subjects with restless legs syndrome. Clin Neuropharmacol. 2012;35(4):165–73.
Inoue Y, Hirata K, Uchimura N, et al. Gabapentin enacarbil in Japanese patients with restless legs syndrome: a 12-week, randomized, double-blind, placebo-controlled, parallel-group study. Curr Med Res Opin. 2013;29(1):13–21.
Lee DO, Ziman RB, Perkins AT, et al. A randomized, double-blind, placebo-controlled study to assess the efficacy and tolerability of gabapentin enacarbil in subjects with restless legs syndrome. J Clin Sleep Med. 2011;7(3):282–92.
Winkelman JW, Bogan RK, Schmidt MH, et al. Randomized polysomnography study of gabapentin enacarbil in subjects with restless legs syndrome. Mov Disord. 2011;26(11):2065–72.
Kushida CA, Becker PM, Ellenbogen AL, et al. Randomized, double-blind, placebo-controlled study of XP13512/GSK1838262 in patients with RLS. Neurology. 2009;72(5):439–46.
GlaxoSmithKline. Result summary: study 114025. 2014. http://www.gsk-clinicalstudyregister.com/files2/114025-Clinical-Study-Result-Summary.pdf. Accessed 26 Apr 2016.
Lee DO, Buchfuhrer MJ, Garcia-Borreguero D, et al. Efficacy of gabapentin enacarbil in adult patients with severe primary restless legs syndrome. Sleep Med. 2015;19:50–6.
VanMeter SA, Kavanagh ST, Warren S, et al. Dose response of gabapentin enacarbil versus placebo in subjects with moderate-to-severe primary restless legs syndrome: an integrated analysis of three 12-week studies. CNS Drugs. 2012;26(9):773–80.
Ondo WG, Hermanowicz N, Garcia Borreguero D, et al. Effect of prior exposure to dopamine agonists on treatment with gabapentin enacarbil in adults with moderate-to-severe primary restless legs syndrome: pooled analyses from 3 randomized trials. J Clin Mov Disord. 2015. doi:10.1186/s40734-015-0018-3.
Hermanowicz N, Buchfuhrer M, Wynn D, et al. The effect of gabapentin enacarbil (GEn) on pain outcomes in adults with moderate-to-severe and severe primary restless legs syndrome (RLS): pooled analyses from 3 randomized controlled trials [abstract no. P7.294 plus poster]. In: 67th Annual Meeting of the American Academy of Neurology; 2015.
Hermanowicz N, Ellenbogen A, Irving G, et al. The effect of gabapentin enacarbil on pain associated with moderate-to-severe primary restless legs syndrome in adults: pooled analyses from three randomized controlled trials. CNS Drugs. 2016. doi:10.1007/s40263-016-0333-8.
Ahmed M, Hays R, Poceta J, et al. The effect of gabapentin enacarbil on individual items of the international restless legs scale and post-sleep questionnaire in patients with moderate-to-severe primary restless legs syndrome: pooled analyses from 3 randomized trials [abstract no. 0634]. Sleep. 2014;37(abstract supplement):A221.
Bogan RK, Lee DO, Buchfuhrer MJ, et al. Treatment response to sleep, pain, and mood disturbance and their correlation with sleep disturbance in adult patients with moderate-to-severe primary restless legs syndrome: pooled analyses from 3 trials of gabapentin enacarbil. Ann Med. 2015;47(3):269–77.
Avidan AY, Lee D, Park M, et al. The effect of gabapentin enacarbil on quality of life and mood outcomes in a pooled population of adult patients with moderate-to-severe primary restless legs syndrome. CNS Drugs. 2016. doi:10.1007/s40263-016-0329-4.
Avidan A, Isaacson S, Jaros M, et al. The effect of gabapentin enacarbil (GEn) on quality-of-life (QoL) outcomes in adult patients with moderate-to-severe and severe primary restless legs syndrome (RLS): pooled analyses from two 12-week trials [abstract no. P7.299]. Neurology. 2015;84(14 Suppl).
Ellenbogen AL, Thein SG, Winslow DH, et al. A 52-week study of gabapentin enacarbil in restless legs syndrome. Clin Neuropharmacol. 2011;34(1):8–16.
Inoue Y, Uchimura N, Kuroda K, et al. Long-term efficacy and safety of gabapentin enacarbil in Japanese restless legs syndrome patients. Prog Neuropsychopharmacol Biol Psychiatry. 2012;36(2):251–7.
Bogan RK, Cramer Bornemann MA, Kushida CA, et al. Long-term maintenance treatment of restless legs syndrome with gabapentin enacarbil: a randomized controlled study. Mayo Clin Proc. 2010;85(6):512–21.
US FDA. Horizant (gabapentin enacarbil) medial review. 2011. https://www.fda.gov. Accessed 26 Apr 2016.
Irizarry MC, Webb DJ, Boudiaf N, et al. Risk of cancer in patients exposed to gabapentin in two electronic medical record systems. Pharmacoepidemiol Drug Saf. 2012;21(2):214–25.
Pharmaceuticals and Medical Devices Agency. Regnite® tablets: Japanese prescribing information (in Japanese). 2015. http://www.pmda.go.jp/english/. Accessed 26 Apr 2016.
Aurora RN, Kristo DA, Bista SR, et al. The treatment of restless legs syndrome and periodic limb movement disorder in adults-an update for 2012: practice parameters with an evidence-based systematic review and meta-analyses: an American Academy of Sleep Medicine clinical practice guideline. Sleep. 2012;35(8):1039–62.
Aurora RN, Kristo DA, Bista SR, et al. Update to the AASM clinical practice guideline: “The treatment of restless legs syndrome and periodic limb movement disorder in adults-an update for 2012: practice parameters with an evidence-based systematic review and meta-analyses”. Sleep. 2012;35(8):1037.
Garcia-Borreguero D, Ferini-Strambi L, Kohnen R, et al. European guidelines on management of restless legs syndrome: report of a joint task force by the European Federation of Neurological Societies, the European Neurological Society and the European Sleep Research Society. Eur J Neurol. 2012;19(11):1385–96.
Garcia-Borreguero D, Kohnen R, Silber MH, et al. The long-term treatment of restless legs syndrome/Willis-Ekbom disease: evidence-based guidelines and clinical consensus best practice guidance: a report from the International Restless Legs Syndrome Study Group. Sleep Med. 2013;14(7):675–84.
Sun Y, van Valkenhoef G, Morel T. A mixed treatment comparison of gabapentin enacarbil, pramipexole, ropinirole and rotigotine in moderate-to-severe restless legs syndrome. Curr Med Res Opin. 2014;30(11):2267–78.
Acknowledgments
During the peer review process, the manufacturer of gabapentin enacarbil was also offered an opportunity to review this article. Changes resulting from comments received were made on the basis of scientific and editorial merit.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Funding
The preparation of this review was not supported by any external funding.
Conflict of interest
Esther Kim and Emma Deeks are salaried employees of Adis/Springer, are responsible for the article content and declare no relevant conflicts of interest.
Additional information
The manuscript was reviewed by: R. K. Bogan, SleepMed Inc., School of Medicine, University of South Carolina, Columbia, SC, USA; K. Suzuki, Department of Neurology, Dokkyo Medical University, Mibu, Tochigi, Japan.
Rights and permissions
About this article
Cite this article
Kim, E.S., Deeks, E.D. Gabapentin Enacarbil: A Review in Restless Legs Syndrome. Drugs 76, 879–887 (2016). https://doi.org/10.1007/s40265-016-0584-1
Published:
Issue Date:
DOI: https://doi.org/10.1007/s40265-016-0584-1