Abstract
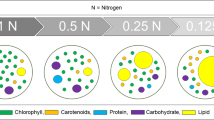
Microalgal lipids can be enhanced through varying nitrogen (N) content, and limited supply of nitrogen source seems to be valuable approach for increased lipid accumulation in microalgae. In this study, Chlorella sp. IM-02 was observed under fluorescence microscope for increased number of lipid bodies under nitrogen scarcity. Fourier transform infrared spectroscopy was used to determine spectral changes due to varying lipid content under nitrogen-starved (N0, without sodium nitrate), nitrogen-limited (N0.1, N0.25, N0.5 and N1.0 representing 0.1, 0.25, 0.5 and 1.0 g/L of sodium nitrate, respectively) and nitrogen-sufficient (N1.5, i.e., 1.5 g/L sodium nitrate) setting. Chlorophyll content was also monitored under these conditions as growth indicator. Various biochemical components viz. total carbohydrates, total proteins and total lipids were also estimated under varying nitrogen levels spectrophotometrically. On fourth day itself, maximum lipid productivity was observed in case of N0.5, which is having one-third of nitrogen concentration present in original growth media, BG-11. This concludes N0.5 as suitable nitrogen provision for better production of lipids in Chlorella sp. IM-02 without much compromising the biomass production as both growth and lipid quantity are key parameters affecting the lipid productivity of any microalgal strain.








Similar content being viewed by others
References
Beardall J, Berman T, Heraud P, Kadiri MO, Light BR, Patterson G, Roberts S, Sulzberger B, Sahan E, Uehlinger U, Wood B (2012) A comparison of methods for detection of phosphate limitation in microalgae. Aquat Sci 63:107–121
Belotti G, Barvi M, Caprariis DB, Filippis DP, Scarsella M (2013) Effect of nitrogen and phosphorous starvations on Chlorella vulgaris lipid productivity and quantity under different tropic regimens for biodiesel production. Am J Plant Sci 4:44–51
Botham PA, Ratledge C (1979) A biochemical explanation for lipid accumulation in Candida 107 and other oleaginous micro-organisms. J Microbiol 114:361–375
Breuer G, Lamers PP, Martens DE, Draaisma RB, Wijffels RH (2012) The impact of nitrogen starvation on the dynamics of triacylglycerol accumulation in nine microalgae strains. Bioresour Technol 124:217–226
Chen Y, Vaidyanathan S (2012) A simple, reproducible and sensitive spectrophotometric method to estimate microalgal lipids. Anal Chim Acta 724:67–72
Chen W, Zhang C, Song L, Sommerfeld M, Hu Q (2011) Microwave-assisted Nile red method for in vivo quantification of neutral lipids in microalgae. Bioresour Technol 102:134–141
Converti A, Casazza AA, Ortiz EY, Perego P, Borghi MD (2009) Effect of temperature and nitrogen concentration on the growth and lipid content of Nannochloropsis and Chlorella vulgaris for biodiesel production. Chem Eng Process 48:1146–1151
Da Silva AF, Lourenco SO, Chalou RM (2009) Effect of nitrogen starvation on the photosynthetic physiology of a tropical marine Rhodomonas sp. (Cryptophyceae). Aquat Bot 91:279–291
Dayananda C, Sarada R, Shamala TR, Ravishankar GA (2006) Influence of nitrogen sources on growth, hydrocarbon and fatty acid production by Botryococcus braunii. Asian J Plant Sci 5:799–804
Dean AP, Sigee DC, Estrada B, Pittman JK (2010) Using FTIR spectroscopy for rapid determination of lipid accumulation in response to nitrogen limitation in freshwater microalgae. Bioresour Technol 101:4499–4507
Dubois M, Gillies KA, Hamilton JK, Hemers PA, Smith F (1956) Colorimetric method for the determination of sugars and related compound. Anal Chem 28:350–356
Gouveia L, Oliveira AC (2009) Microalgae as a raw material for biofuels production. J Ind Microbiol Biotechnol 36:269–274
Griffiths MJ, Harrisons STL (2009) Lipid productivity is a key characteristic for choosing algal species for biodiesel production. J Appl Phycol 21:493–507
James GO, Hocart CH, Hiller W, Chen H, Kordbacheh F, Price GD, Djordjevic MA (2011) Fatty acid profiling of Chlamydomonas reinhardtii under nitrogen deprivation. Bioresour Technol 102:3343–3351
John RP, Anisha GS, Nampoothiri KM, Pandey A (2011) Micro and macroalgal biomass: a renewable source for bioethanol. Bioresour Technol 102:186–193
Kiran B, Kumar R, Deshmukh D (2014a) Perspectives of microalgal biofuels as a renewable source of energy. Energy Conserv Manag 88:1228–1244
Kiran B, Pathak K, Kumar R, Deshmukh D (2014b) Cultivation of Chlorella sp. IM-01 in municipal wastewater for simultaneous nutrient removal and energy feedstock production. Ecol Eng J 73:326–330
Kiran B, Pathak K, Kumar R, Deshmukh D (2016a) Growth pattern and biofuel production potential of newly isolated microalga, Chlorococcum sp. IM-03 under nitrogen limited conditions. J Chem Technol Biotechnol 91:1339–1344
Kiran B, Pathak K, Kumar R, Deshmukh D (2016b) Statistical optimization using central composite design for biomass and lipid productivity of microalga: a step towards enhanced biodiesel production. Ecol Eng J 92:73–81
Lowry OH, Rosebrough NJ, Farr LJ, Randall RJ (1951) Protein measurement with the folin phenol reagent. J Biol Chem 193:265–275
Ma YL (2006) Microbial oils and its research advance. Chin J Bioprocess Eng 4(4):7–11
Mackinney G (1941) Absoprtion of light by chlorophyll solution. J Biol Chem 140:315–322
Meng Y, Yao C, Xue S, Yang H (2014) Application of Fourier transform infrared (FT-IR) spectroscopy in determination of microalgae composition. Bioresour Technol 151:347–354
Nigam S, Rai MP, Sharma R (2011) Effect of nitrogen on growth and lipid content of Chlorella pyrenoidosa. Am J Biochem Biotechnol 7:124–129
Ponnuswamy I, Madhavan S, Shabudeen S (2013) Isolation and characterization of green microalgae for carbon sequestration, wastewater treatment and bio-fuel production. Int J Biosci Biotechnol 5:17–26
Pruvost J, Vooren G, Gouic B, Mossion A, Legrand J (2011) Systematic investigation of biomass and lipid productivity by microalgae in photobioreactors for biodiesel application. Bioresour Technol 102:150–158
Rukminasari N (2013) Effect of temperature and nutrient limitation on the growth and lipid content of three selected microalgae (Dunaliella tertiolecta, Nannochloropsis sp. and Scendesmus sp.) for biodiesel production. Int J Mar Sci 3:135–144
Sharma KK, Schuhmann H, Schenk PM (2012) High lipid induction in microalgae for biodiesel production. Energies 5:1532–1555
Sopher HA, Miller WH (1949) The chemical composition of Chlorella; effect of environmental condition. Plant Physiol 24:120–149
Stanier RY, Kunisawa R, Mandel M, Cohen-Bazire G (1971) Purification and properties of unicellular blue-green algae (order Chloroococcales). Bacteriol Rev 35:171–205
Wang L, Min M, Li Y, Chen P, Chen Y, Liu Y, Wang Y, Ruan R (2009) Cultivation of green algae Chlorella sp. in different wastewaters from municipal wastewater treatment plant. Appl Biochem Biotechnol 162:1174–1186
Xin L, Ying HH, Ke G, Xue XY (2010) Effect of different nitrogen and phosphorous concentration on the growth, nutrient uptake, and lipid accumulation of a freshwater microalga Scenedesmus sp. Bioresour Technol 101:5494–5500
Xu H, Miao X, Wu Q (2006) High quality biodiesel production from microalga Chlorella protothecoides by heterotrophic growth in fermenters. J Biotechnol 126:499–507
Yeesang C, Cheirsilp B (2011) Effect of nitrogen, salt, and iron content in the growth medium and light intensity on lipid production by microalgae isolated from freshwater sources in Thailand. Bioresour Technol 103:3034–3040
Yeh KL, Chang JS (2011) Nitrogen starvation strategies and photobioreactor design for enhancing lipid content and lipid production of a newly isolated microalga Chlorella vulgaris ESP-31: implication for biofuels. Biotechnol J 6:1358–1366
Zhu CJ, Lee KL, Chao TM (2009) Effect of temperature and growth phase on lipid and biochemical composition of Isochrysis galbana TK1. J Appl Phycol 9:451–457
Acknowledgments
The authors acknowledge financial assistance provided by Department of Science and Technology, Govt. of India under the INSPIRE Faculty Scheme (IFA12–EAS-01). The funding organization has not played any role in study design, decision to publish or preparation of the manuscript. Authors also acknowledge Dr. Radha Prasanna, IARI, Delhi, for her support in the identification of this strain and Dr. Shrikant Joshi, Veterinary College, Mhow, for giving permission regarding usage of fluorescence microscope in the present study.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors have declared no conflict of interest.
Rights and permissions
About this article
Cite this article
Kiran, B., Pathak, K., Kumar, R. et al. Influence of varying nitrogen levels on lipid accumulation in Chlorella sp.. Int. J. Environ. Sci. Technol. 13, 1823–1832 (2016). https://doi.org/10.1007/s13762-016-1021-4
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s13762-016-1021-4