Abstract
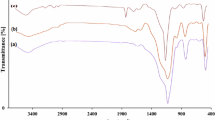
In this paper, we adopt a facile method to prepare a type of porous silica nanoparticles (n-SiO2) from tetraethyl orthosilicate as the source of silica. Then, dendritic polymer supported on nanosilica with surface amino groups was fabricated. Finally, H3PW12O40 nanoparticles (PWAn) were synthesized by the treatment of H3PW12O40 powder with n-Octane as solvent by a solvothermal method and then immobilized onto the dendrimer polymer functionalized nanosilica. The synthesized nanostructures were characterized by fourier transform infrared (FT-IR), X-ray diffraction (XRD), thermogravimetric analysis (TGA), dynamic light scattering (DLS), N2 adsorption–desorption isotherm analysis, UV–Vis and elemental analysis. The morphology of the catalyst was characterized using transmission electron microscopes (TEM). The acidity of the catalyst was determined by FTIR pyridine adsorption spectroscopy. Then, this catalytic system was used as an efficient catalyst for the synthesis of 2H-indazolo[2,1-b]phthalazine-triones via multi-component and one-pot reactions of various aldehydes, phthalic anhydride, hydrazinium hydroxide, and dimedone under thermal solvent-free conditions or ultrasound irradiation at room temperature. Also, the catalyst can be easily recovered and reused for six consecutive reaction cycles without significant loss of activity.







Similar content being viewed by others
References
P. Li, S. Regati, R.J. Butcher, H.D. Arman, Z. Chen, S. Xiang, B. Chen, C.G. Zhao, Tetrahedron Lett. 52, 6220 (2011)
B. Willy, T.J. Muller, Eur. J. Org. Chem. 24, 4157 (2008)
V.P. Litvinov, Russ. Chem. Rev. 72, 69 (2003)
Q.R. Zhang, D.Q. Xue, P. He, K.P. Shao, P.J. Chen, Y.F. Gu, J.L. Ren, L.H. Shan, H.M. Liu, Bioorg. Med. Chem. Lett. 24, 1236 (2014)
L. Zhang, L.P. Guan, X.Y. Sun, C.X. Wei, K.Y. Chai, Z.S. Quan, Chem. Bio. Drug Des. 73, 313 (2009)
R. Ghorbani-Vaghei, R. Karimi-Nami, Z. Toghraei-Semiromi, M. Amiri, M. Ghavidel, Tetrahedron 67, 1930 (2011)
G. Shukla, R.K. Verma, G.K. Verma, M.S. Singh, Tetrahedron Lett. 52, 7195 (2011)
M.V. Reddy, G.C.S. Reddy, Y.T. Jeong, Tetrahedron 68, 6820 (2012)
R. Fazaeli, H. Aliyan, N. Fazaeli, Open Cat. J. 3, 14 (2010)
H.R. Shaterian, E. Mollashahi, A. Biabangard, J. Chem. Soc. Pak. 35, 329 (2013)
A. Khalafi-Nezhad, M. Divara, F. Panahi, RSC Adv. 5, 2223 (2015)
B. Karami, V. Ghashghaee, S. Khodabakhshi, Catal. Commun. 20, 71 (2012)
Varsha Brahmkhatri, Anjali Patel, Ind. Eng. Chem. Res. 50, 13693 (2011)
M. Esmaeilpour, J. Javidi, F. Dehghani, F. Nowroozi, Dodeji. New J. Chem. 38, 5453 (2014)
M. Esmaeilpour, J. Javidi, M. Zandi, Mater. Res. Bull. 55, 78 (2014)
A. Khalafi-Nezhad, F. Panahi, R. Yousefi, S. Sarrafi, Y. Gholamalipour, J. Iran. Chem. Soc. 11, 1311 (2014)
M. Esmaeilpour, J. Javidi, M. Zandi, New J. Chem. 39, 3388 (2015)
J. Javidi, M. Esmaeilpour, Z. Rahiminezhad, F. Nowroozi, Dodeji. J. Clust. Sci. 25, 1511 (2014)
J. Javidi, M. Esmaeilpour, F. Nowroozi Dodeji, RSC Adv. 5, 308 (2015)
D.H.W. Hubert, M. Jung, A.L. German, Adv. Mater. 12, 1291 (2000)
M. Salavati-Niasari, J. Javidi, M. Dadkhah, Comb. Chem. High Throughput Screening 16, 458 (2013)
C.B. Gorman, J.C. Smith, Acc. Chem. Res. 34, 60 (2001)
R. Heerbeek, P.C.J. Kamer, P.W.N.M. Leeuwen, J.N.H. Reek, Chem. Rev. 102, 3717 (2002)
A.J.L. Villaraza, A. Bumb, M.W. Brechbiel, Chem. Rev. 110, 2921 (2010)
M. Salavati-Niasari, J. Javidi, J. Clust. Sci. 23, 1019 (2012)
R.S. Kalhapure, M.K. Kathiravan, K.G. Akamanchi, T. Govender, Pharm. Dev. Technol. 20, 22 (2015)
M. Esmaeilpour, J. Javidi, F. Nowroozi Dodeji, M. Mokhtari Abarghoui, J. Mol. Catal A Chem. 393, 18 (2014)
A.E. Torkamani, P. Juliano, S. Ajlouni, T.K. Singh, Ultrason. Sonochem. 21, 951 (2014)
A. Kumar, R. A. Maurya, Synlett 6, 883 (2008)
M. Esmaeilpour, J. Javidi, F. Nowroozi Dodeji, M. Mokhtari Abarghoui, Transition Met. Chem. 39, 797 (2014)
L.T. Aany Sofia, K. Asha, M. Sankar, N.K. Kala Raj, P. Manikandan, P.R. Rajamohanan, T.G. Ajithkumar, J. Phys. Chem. C 113, 21114 (2009)
A.S. Ertürk, M. Tülü, A.E. Bozdoğan, T. Parali, Eur. Polym. J. 52, 218 (2014)
M. Esmaeilpour, J. Javidi, M. Mokhtari Abarghoui, F. Nowroozi Dodeji, J. Iran Chem. Soc. 11, 499 (2014)
X.-M. Yan, J.-H. Lei, D. Liu, Y.-C. Wu, W. Liu, Mater. Res. Bull. 42, 1905 (2007)
J. Wang, S. Zheng, Y. Shao, J. Liu, Z. Xu, D. Zhu, J. Colloid. Interf. Sci. 349, 293 (2010)
M. Shekouhy, A. Hasaninejad, Ultrason. Sonochem. 19, 307 (2012)
X.N. Zhao, G.F. Hu, M. Tang, T.T. Shi, X.L. Guo, T.T. Li, Z.H. Zhang, RSC Adv. 4, 51089 (2014)
A. Hasaninejed, M.R. Kazerooni, A. Zare, Catal. Today 196, 148 (2012)
G. Sabitha, C. Srinivas, A. Raghavendar, J.S. Yadav, Helv. Chim. Acta 93, 1375 (2010)
H. Veisi, A.A. Manesh, N. Khankhani, R. Ghorbani-Vaghei, RSC Adv. 4, 25057 (2014)
E. Mosaddegh, A. Hassankhani, Tetrahedron Lett. 52, 488 (2011)
H.R. Shaterian, F. Khorami, A. Amirzadeh, R. Doostmohammadi, M. Ghashang, J. Iran. Chem. Res. 2, 57 (2009)
A.R. Kiasat, S. Noorizadeh, M. Ghahremani, S.J. Saghanejad, J. Mol. Struct. 1036, 216 (2013)
L. Nagarapu, R. Bantu, H.B. Mereyala, J. Heterocycl. Chem. 46, 728 (2009)
B. Maleki, S. Sedigh-Ashrafi, R. Tayebee, RSC Adv. 4, 41521 (2014)
J.M. Khurana, D. Magoo, Tetrahedron Lett. 50, 7300 (2009)
X. Wang, W.W. Ma, L.Q. Wu, F.L. Yan, J. Chin. Chem. Soc. 57, 1341 (2010)
Acknowledgments
Authors are grateful to the council of Iran National Science Foundation and University of Shiraz for their unending effort to provide financial support to undertake this work.
Author information
Authors and Affiliations
Corresponding authors
Rights and permissions
About this article
Cite this article
Esmaeilpour, M., Javidi, J. & Dehghani, F. Preparation, characterization and catalytic activity of dendrimer-encapsulated phosphotungstic acid nanoparticles immobilized on nanosilica for the synthesis of 2H-indazolo[2,1-b]phthalazine-triones under solvent-free or sonochemical conditions. J IRAN CHEM SOC 13, 695–714 (2016). https://doi.org/10.1007/s13738-015-0782-x
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s13738-015-0782-x