Abstract
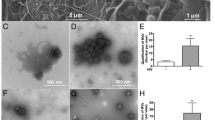
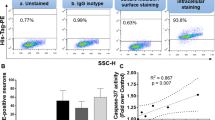
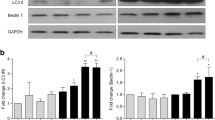
HIV-1-infected mononuclear phagocytes release soluble factors that affect the homeostasis in tissue. HIV-1 can prompt metabolic encephalopathy with the addition of neuronal dysfunction and apoptosis. Recently, we reported that HIV-1 enhances the expression and secretion of bioactive cathepsin B in monocyte-derived macrophages, ultimately contributing to neuronal apoptosis. In this research, we asked if microglia respond to HIV infection similarly by modifying the expression, secretion, and neurotoxic potential of cathepsin B and determined the in vivo relevance of these findings. HIV-1ADA-infected human primary microglia and CHME-5 microglia cell line were assessed for expression and activity of cathepsin B, its inhibitors, cystatins B and C, and the neurotoxicity associated with these changes. Human primary neurons were exposed to supernatants from HIV-infected and uninfected microglia in the presence of cathepsin B inhibitors and apoptosis was assessed by TUNEL. Microglial expression of cathepsin B was validated in brain tissue from HIV encephalitis (HIVE) patients. HIV-infected microglia secreted significantly greater levels of cathepsin B, cystatin B, and cystatin C compared to uninfected cells. Increased apoptosis was observed in neurons exposed to supernatants from HIV-1 infected microglia at day 12 post-infection. The cathepsin B inhibitor CA-074 and cathepsin B antibody prevented neuronal apoptosis. Increased microglia-derived cathepsin B, cystatin B, and cystatin C and caspase-3+ neurons were detected in HIVE brains compared to controls. Our results suggest that HIV-1-induced cathepsin B production in microglia contributes to neuronal apoptosis and may be an important factor in neuronal death associated with HIVE.







Similar content being viewed by others
References
Albright A (2002) Winning at losing: a guide to healthy weight loss. Best and worst exercises for weight loss. What works and what does not for shedding pounds through physical activity. Diabetes Forecast 55:69–70, 72, 74.
Albright AV, Shieh JT, Itoh T et al (1999) Microglia express CCR5, CXCR4, and CCR3, but of these, CCR5 is the principal coreceptor for human immunodeficiency virus type 1 dementia isolates. J Virol 73:205–13
Alexaki A, Liu Y, Wigdahl B (2008) Cellular reservoirs of HIV-1 and their role in viral persistence. Curr HIV Res 6:388–400
Arbour N, Ekandé S, Côté G et al (1999) Persistent infection of human oligodendrocytic and neuroglial cell lines by human coronavirus 229E. J Virol 73:3326–37
Atanassov CL, Muller CD, Dumont S et al (1995) Effect of ammonia on endocytosis and cytokine production by immortalized human microglia and astroglia cells. Neurochem Int 27:417–24
Baratchi S, Kanwar RK, Kanwar JR (2011) Survivin mutant protects differentiated dopaminergic SK-N-SH cells against oxidative stress. PLoS One 6:e15865. doi:10.1371/journal.pone.0015865
Barré-Sinoussi F, Chermann JC, Rey F et al (1983) Isolation of a T-lymphotropic retrovirus from a patient at risk for acquired immune deficiency syndrome (AIDS). Science 220:868–71
Barrett AJ, Kirschke H (1981) Cathepsin B, cathepsin H, and cathepsin L. Methods Enzymol 80 Pt C:535–61
Biedler JL, Helson L, Spengler BA (1973) Morphology and growth, tumorigenicity, and cytogenetics of human neuroblastoma cells in continuous culture morphology and growth, tumorigenicity, and cytogenetics of human neuroblastoma cells in continuous culture1. 2643–2652.
Biedler JL, Roffler-tarlov S, Schachner M, Freedman LS (1978) Multiple neurotransmitter synthesis by human neuroblastoma cell lines and clones multiple neurotransmitter synthesis by human neuroblastoma cell lines and clones. 3751–3757.
Cantres-Rosario Y, Plaud-Valentín M, Gerena Y et al (2013) Cathepsin B and cystatin B in HIV-seropositive women are associated with infection and HIV-1-associated neurocognitive disorders. AIDS 27:347–56. doi:10.1097/QAD.0b013e32835b3e47
Chugh P, Bradel-Tretheway B, Monteiro-Filho CMR et al (2008) Akt inhibitors as an HIV-1 infected macrophage-specific anti-viral therapy. Retrovirology 5:11. doi:10.1186/1742-4690-5-11
Gan L, Ye S, Chu A et al (2004) Identification of cathepsin B as a mediator of neuronal death induced by Abeta-activated microglial cells using a functional genomics approach. J Biol Chem 279:5565–72. doi:10.1074/jbc.M306183200
Gendelman HE, Baca LM, Husayni H et al (1990) Macrophage-HIV interaction: viral isolation and target cell tropism. AIDS 4:221–8
Gendelman HE, Orenstein JM, Martin MA et al (1988) Efficient isolation and propagation of human immunodeficiency virus on recombinant colony-stimulating factor 1-treated monocytes. J Exp Med 167:1428–41
Ghafouri M, Amini S, Khalili K, Sawaya BE (2006) HIV-1 associated dementia: symptoms and causes. Retrovirology 3:28. doi:10.1186/1742-4690-3-28
Ghorpade A, Xia MQ, Hyman BT et al (1998a) Role of the beta-chemokine receptors CCR3 and CCR5 in human immunodeficiency virus type 1 infection of monocytes and microglia. J Virol 72:3351–61
Ghorpade A, Nukuna A, Che M et al (1998b) Human immunodeficiency virus neurotropism: an analysis of viral replication and cytopathicity for divergent strains in monocytes and microglia. J Virol 72:3340–50
Glass JD, Fedor H, Wesselingh SL, McArthur JC (1995) Immunocytochemical quantitation of human immunodeficiency virus in the brain: correlations with dementia. Ann Neurol 38:755–62. doi:10.1002/ana.410380510
González-Scarano F, Baltuch G (1999) Microglia as mediators of inflammatory and degenerative diseases. Annu Rev Neurosci 22:219–40. doi:10.1146/annurev.neuro.22.1.219
Guicciardi ME, Deussing J, Miyoshi H et al (2000) Cathepsin B contributes to TNF-alpha-mediated hepatocyte apoptosis by promoting mitochondrial release of cytochrome c. J Clin Invest 106:1127–37. doi:10.1172/JCI9914
He J, Chen Y, Farzan M et al (1997) CCR3 and CCR5 are co-receptors for HIV-1 infection of microglia. Nature 385:645–9. doi:10.1038/385645a0
Honey K, Rudensky AY (2003) Lysosomal cysteine proteases regulate antigen presentation. Nat Rev Immunol 3:472–82. doi:10.1038/nri1110
Huang Y, Zhao L, Jia B et al (2011) Glutaminase dysregulation in HIV-1-infected human microglia mediates neurotoxicity: relevant to HIV-1-associated neurocognitive disorders. J Neurosci 31:15195–204. doi:10.1523/JNEUROSCI.2051-11.2011
Ioannidis JP, Reichlin S, Skolnik PR (1995) Long-term productive human immunodeficiency virus-1 infection in human infant microglia. Am J Pathol 147:1200–6
Janabi N, Peudenier S, Héron B et al (1995) Establishment of human microglial cell lines after transfection of primary cultures of embryonic microglial cells with the SV40 large T antigen. Neurosci Lett 195:105–8
Jayadev S, Garden GA (2009) Host and viral factors influencing the pathogenesis of HIV-associated neurocognitive disorders. J Neuroimmune Pharmacol 4:175–89. doi:10.1007/s11481-009-9154-6
Kingham PJ, Pocock JM (2001) Microglial secreted cathepsin B induces neuronal apoptosis. J Neurochem 76:1475–84
Kopitar-Jerala N (2006) The role of cystatins in cells of the immune system. FEBS Lett 580:6295–301. doi:10.1016/j.febslet.2006.10.055
Lachance C, Arbour N, Cashman NR, Talbot PJ (1998) Involvement of aminopeptidase N (CD13) in infection of human neural cells by human coronavirus 229E. J Virol 72:6511–9
Laspiur JP, Anderson ER, Ciborowski P et al (2007) CSF proteomic fingerprints for HIV-associated cognitive impairment. J Neuroimmunol 192:157–170. doi:10.1016/j.jneuroim.2007.08.004
Lecointe D, Dugas N, Leclerc P et al (2002) Human cytomegalovirus infection reduces surface CCR5 expression in human microglial cells, astrocytes and monocyte-derived macrophages. Microbes Infect 4:1401–8
Lee SC, Hatch WC, Liu W et al (1993) Productive infection of human fetal microglia in vitro by HIV-1. Ann N Y Acad Sci 693:314–6
Li JH, Pober JS (2005) The cathepsin B death pathway contributes to TNF plus IFN-gamma-mediated human endothelial injury. J Immunol 175:1858–1866
Lucas A, Kim Y, Rivera-Pabon O et al (2010) Targeting the PI3K/Akt cell survival pathway to induce cell death of HIV-1 infected macrophages with alkylphospholipid compounds. PLoS One. doi:10.1371/journal.pone.0013121
Luciano-Montalvo C, Ciborowski P, Duan F et al (2008) Proteomic analyses associate cystatin B with restricted HIV-1 replication in placental macrophages. Placenta 29:1016–1023. doi:10.1016/j.placenta.2008.09.005
Luciano-Montalvo C, Melendez LM (2009) Cystatin B associates with signal transducer and activator of transcription 1 in monocyte-derived and placental macrophages. Placenta 30:464–467. doi:10.1016/j.placenta.2009.03.003
Melendez LM, Colon K, Rivera L et al (2011) Proteomic analysis of HIV-infected macrophages. J Neuroimmune Pharmacol 6:89–106. doi:10.1007/s11481-010-9253-4
Nagai A, Murakawa Y, Terashima M et al (2000) Cystatin C and cathepsin B in CSF from patients with inflammatory neurologic diseases. Neurology 55:1828–32
Nagaraj NS, Vigneswaran N, Zacharias W (2006) Cathepsin B mediates TRAIL-induced apoptosis in oral cancer cells. J Cancer Res Clin Oncol 132:171–83. doi:10.1007/s00432-005-0053-9
Nguyen MD, Julien J-P, Rivest S (2002) Innate immunity: the missing link in neuroprotection and neurodegeneration? Nat Rev Neurosci 3:216–27. doi:10.1038/nrn752
Nishida Y, Adati N, Ozawa R et al (2008) Identification and classification of genes regulated by phosphatidylinositol 3-kinase- and TRKB-mediated signalling pathways during neuronal differentiation in two subtypes of the human neuroblastoma cell line SH-SY5Y. BMC Res Notes 1:95. doi:10.1186/1756-0500-1-95
Nixon RA, Cataldo AM, Mathews PM (2000) The endosomal-lysosomal system of neurons in Alzheimer’s disease pathogenesis: a review. Neurochem Res 25:1161–72
O’Donnell LA, Agrawal A, Jordan-Sciutto KL et al (2006) Human immunodeficiency virus (HIV)-induced neurotoxicity: roles for the NMDA receptor subtypes. J Neurosci 26:981–990. doi:10.1523/JNEUROSCI.4617-05.2006
Pennacchio LA, Lehesjoki AE, Stone NE et al (1996) Mutations in the gene encoding cystatin B in progressive myoclonus epilepsy (EPM1). Science 271:1731–4
Peudenier S, Hery C, Montagnier L, Tardieu M (1991) Human microglial cells: characterization in cerebral tissue and in primary culture, and study of their susceptibility to HIV-1 infection. Ann Neurol 29:152–61. doi:10.1002/ana.410290207
Rivera-Rivera L, Perez-Laspiur J, Colón K, Meléndez LMM (2012) Inhibition of interferon response by cystatin B: implication in HIV replication of macrophage reservoirs. J Neurovirol 18:20–9. doi:10.1007/s13365-011-0061-2
Rock RB, Gekker G, Hu S et al (2004) Role of microglia in central nervous system infections. Clin Microbiol Rev 17:942–64. doi:10.1128/CMR.17.4.942-964.2004, table of contents
Rodriguez-Franco EJ, Cantres-Rosario YM, Plaud-Valentin M et al (2012) Dysregulation of macrophage-secreted cathepsin B contributes to HIV-1-linked neuronal apoptosis. PLoS One 7:e36571. doi:10.1371/journal.pone.0036571
Schurigt U, Stopfel N, Hückel M et al (2005) Local expression of matrix metalloproteinases, cathepsins, and their inhibitors during the development of murine antigen-induced arthritis. Arthritis Res Ther 7:R174–88. doi:10.1186/ar1466
Shieh JT, Albright AV, Sharron M et al (1998) Chemokine receptor utilization by human immunodeficiency virus type 1 isolates that replicate in microglia. J Virol 72:4243–9
Stoka V, Turk B, Schendel SL et al (2001) Lysosomal protease pathways to apoptosis. Cleavage of bid, not pro-caspases, is the most likely route. J Biol Chem 276:3149–57. doi:10.1074/jbc.M008944200
Tambuyzer BR, Ponsaerts P, Nouwen EJ (2009) Microglia: gatekeepers of central nervous system immunology. J Leukoc Biol 85:352–70. doi:10.1189/jlb.0608385
Tran TM, Temkin V, Shi B et al (2009) TNFalpha-induced macrophage death via caspase-dependent and independent pathways. Apoptosis 14:320–32. doi:10.1007/s10495-009-0311-4
Tsai H-T, Wang P-H, Tee Y-T et al (2009) Imbalanced serum concentration between cathepsin B and cystatin C in patients with pelvic inflammatory disease. Fertil Steril 91:549–55. doi:10.1016/j.fertnstert.2007.12.076
Velazquez I, Plaud M, Wojna V et al (2009) Antioxidant enzyme dysfunction in monocytes and CSF of Hispanic women with HIV-associated cognitive impairment. J Neuroimmunol 206:106–111. doi:10.1016/j.jneuroim.2008.10.013
Voyvodic JT (1996) Cell death in cortical development: how much? Why? So what? Neuron 16:693–6
Wakasugi K, Nakano T, Morishima I (2004) Association of human neuroglobin with cystatin C, a cysteine proteinase inhibitor. Biochemistry 43:5119–25. doi:10.1021/bi0495782
Wang T, Gong N, Liu J et al (2008) Proteomic modeling for HIV-1 infected microglia-astrocyte crosstalk. PLoS One 3:e2507. doi:10.1371/journal.pone.0002507
Yoshii H, Kamiyama H, Goto K et al (2011) CD4-independent human immunodeficiency virus infection involves participation of endocytosis and cathepsin B. PLoS One 6:e19352. doi:10.1371/journal.pone.0019352
Zenón F, Segarra AC, Gonzalez M, Meléndez LM (2014) Cocaine potentiates cathepsin B secretion and neuronal apoptosis from HIV-infected macrophages. J Neuroimmune Pharmacol 9:703–15. doi:10.1007/s11481-014-9563-z
Acknowledgments
We thank Dr. Randall L. Davis from the Oklahoma State University Center for Health Sciences for kindly providing the CHME-5 microglia cell line. We thank Dr. Howard Gendelman from the University of Nebraska Medical Center for providing HIV-ADA isolates. We would like to thank Rafael Romeu for his help with cell cultures and Dr. Yamil Gerena for the advice in flow cytometry. We would like to acknowledge Eduard Guerrero, Carol Torres from the RCMI Emergent Disease Core Laboratories, and Dr. Juliana Perez Laspiur and Yolanda Rodriguez from the Proteomics Core Laboratory for all their excellent technical support. Part of this work was previously presented as a poster at the ISNV. May 28-June 2, 2012, NY. Part of this work was also presented at the Annual Biomedical Research Conference for Minority Students (ABRCMS) meeting, November 13–16, 2013, Nashville, TN, USA
Funding
This work was supported in part by grants from the National Institutes of Health R01MH083516 (to LMM), U54NS4301 (to LMM), R25GM061838 (to FZ, ER, YC), GM08224, and NIMHHD8G12-MD007600 (Emergent Diseases Core and Translational Proteomics Center), and SC1GM11369-01. We acknowledge the facilities from grants ISI0 RR-13705-01 and DBI-0923132 to establish and upgrade the Confocal Microscopy Facility at the University of Puerto Rico (CIF-UPR). UPR Vice President (M.P.). UPR Medical Sciences Campus Chancellor, and the Associate Deanship of Biomedical Sciences provided additional funding to complete this study. This publication was made possible from NIH funding through the NIMH and NINDS Institutes by the following grants: Manhattan HIV Brain Bank U01MH083501, R24MH59724; Texas NeuroAIDS Research Center U01MH083507, R24 NS45491; National Neurological AIDS Bank 5U01MH083500, NS 38841; California NeuroAIDS Tissue Network U01MH083506, R24MH59745; and Statistics and Data Coordinating Center U01MH083545, N01MH32002. Its contents are solely the responsibility of the authors and do not necessarily represent the official view of the NNTC or NIH.
Conflict of interest
The authors declare that they have no competing interests.
Author information
Authors and Affiliations
Corresponding author
Electronic supplementary material
Below is the link to the electronic supplementary material.
ESM 1
(PDF 77584 kb)
Rights and permissions
About this article
Cite this article
Zenón, F., Cantres-Rosario, Y., Adiga, R. et al. HIV-infected microglia mediate cathepsin B-induced neurotoxicity. J. Neurovirol. 21, 544–558 (2015). https://doi.org/10.1007/s13365-015-0358-7
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s13365-015-0358-7