Abstract
Knowledge on reproductive success is vital for successful management of large ungulates and is often measured by means of observing surviving offspring. In harvested ungulates, postmortem investigations of reproductive organs are used to estimate reproductive potential by obtaining ovulation rates and fetus numbers. However, there are differences in numbers of offspring observed, fetal/embryo counts, and ovulation rates. We hypothesize that the discrepancy between estimated reproductive potential and reproductive outcome in large ungulates is not only due to ova loss but also due to embryonic mortality. We investigated reproductive status in early pregnancy by sampling hunter-harvested moose (Alces alces) in southern Sweden from 2007 to 2011. In all, 213 reproductive organs were examined postmortem, and in confirmed pregnant moose (n = 53), 25 % (19 of 76) embryos were nonviable and 6 % of ova was unfertilized. The discrepancy between the ovulation rate of all pregnant moose (1.49) and the number of expected offspring per pregnant female, when embryonic mortality and unfertilized oocytes were accounted for (1.08), was 27.5 %. An association between inflammation of the inner mucous membrane (endometritis) of the moose's uterus and embryonic mortality was observed. This is the first comprehensive report of embryonic mortality and endometritis in moose. The observed discrepancy between ovulation rates and early embryonic development/survival shows that ovulation rates are indicative but not accurate estimates of moose reproductive rate. The use of ovulation rates as a sole estimator of future offspring rates may lead to an overharvest of a managed moose population.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
For intensively managed large ungulates, knowledge about reproductive performance (individual and population fecundity) is vital for successful management and population sustainability. In many wild, large ungulate species, knowledge of reproduction is to a large extent based on research in captive animals, kept under restricted conditions, which may not reflect the situation of a natural habitat. To obtain further knowledge about reproduction in wild animals, there is a need to study specimens in the wild. In this respect, obtaining large amounts of samples is feasible in species subjected to harvest. In moose (Alces alces), reproductive performance is usually measured by observing numbers of surviving offspring after the summer period (calf per adult female ratio, Ericsson and Wallin 1999). Reproductive potential as well as individual and population fecundity (population production of offspring) among female moose can also be obtained by performing postmortem examinations of reproductive organs. In general, ovulation rates and/or embryo/fetal counts are used as the standard measurement. Some reports indicate that embryonic mortality and unfertilized oocytes (ova loss) account for a discrepancy between the number of fetuses and ovulation rates in harvested females (Markgren 1969; Schwartz and Hundertmark 1993). Though rarely reported, disorders of the reproductive system in wild, large ungulates could affect reproductive success and thus population viability. Such disorders may include embryonic mortality (of various origin), diseases of the reproductive tract (infectious and noninfectious), and loss of ova/oocytes. The aim of this study was to determine the discrepancy between ovulation rates and early pregnancy loss (due to embryonic mortality and unfertilized oocytes) in free-ranging Scandinavian moose by postmortem examination of reproductive organs from hunter-harvested females. We hypothesize that loss of ova is not only attributed to non-fertilization of oocytes (ova loss) but also dependent on embryonic mortality and survival. Also, we hypothesize that there is a discrepancy between ovulation rates and embryo survival rates.
Materials and methods
Sampling of moose
From 2007 to 2011, during the moose-hunting season (opening in the second week of October), reproductive organs and mandibles were collected from harvested female moose in three areas of the southern part of Sweden (Fig. 1). The sampling areas consisted of local moose management units located from 56°55.450′ N, 14°45.056′ E to 59°5.323′ N, 17°22.600′ E. Field laboratories for macroscopic examination of the reproductive organs were set up in the vicinity of the hunting areas, and hunters were asked to directly contact trained field personnel when a female moose was harvested. Collected samples were stored in coolers (at approximately +8 °C) during transport from the collection site to the field laboratory. Some samples (n = 35), that were collected without access to a field laboratory (from animals harvested later during the hunting season), were stored at −20 °C and transported frozen to the laboratory at the Department of Clinical Sciences in Uppsala, Sweden, where they were subsequently thawed and examined.
Age determination
Moose age was determined by sectioning the first molar and counting of cementum layers (Wolfe 1969). In 22 of the 213 moose, age determination was not possible because hunters decapitated the animals before transport.
Examination of reproductive organs
Ovarian structures were examined and the size and numbers of follicles (≥5 mm) and corpora lutea recorded. The presence of one or more corpora lutea was the criteria for ovulation. Uteri were weighed (ligaments and cervix not included) and cut open to macroscopically investigate mucous membranes and content, including embryos. Signs of early pregnancy (i.e., embryonic structures such as allantochorionic membranes, proper embryos, or remnants of embryos), as well as any abnormalities such as signs of uterus inflammation (discolored mucous membrane, presence of pus), were noted.
From uteri with signs of inflammation, samples for bacteriological culture were taken with a sterile swab and transported in Amies medium (Copan S.P.A., Brescia, Italy) to the National Veterinary Institute, Uppsala for routine aerobic culture on blood agar at 37 °C for 24 h. Tissue samples (0.5 × 1 cm) were taken from at least two sites of the uterus for microscopic examination to verify the macroscopic signs of inflammation (increased infiltration of inflammatory cells). The tissue samples were fixed in formalin solution (10 %), dehydrated, embedded in paraffin, and stained with hematoxylin–eosin. In order to determine reproductive status, ovarian and uterine findings were combined. Confirmation that conception had occurred included the presence of one or more corpora lutea, allantochorionic membranes, a proper embryo (Fig. 3), and/or remnants of embryos. The ratio between numbers of corpora lutea and embryos was calculated in order to attain the proportion of fertilized oocytes. To be classified as embryonic mortality, at least one of the following criteria had to be fulfilled:
-
(a)
Malformed embryo with failure of organogenesis (uneven edges, no visible hind or forelimbs, misshaped head, absence of visible liver; Fig. 2)
-
(b)
Judged to be older than 2 weeks of pregnancy, i.e., longer and wider but not containing embryo proper or containing remnants of allantochorionic membranes (Fig. 3)
Data and statistical analyses
In all animals found to be pregnant, the total number of corpora lutea (CLtot) was calculated to assess the ovulation rate. To determine the rate of fertilized oocytes (conception rate), the total number of embryo-related structures (Etot) was divided by the total number of corpora lutea. Viable embryos without observed disorders were noted (Enorm). The rate of embryonic mortality (EM) was determined by dividing all embryonic structures judged to be nonviable (Emalf) with Etot. To determine the rate of unfertilized oocytes, the total number of embryo-related structures (Etot) was subtracted from the total number of corpora lutea (CLtot). Pregnancy outcome (mean number of embryos per pregnant female) was determined by subtracting the rate of embryonic mortality and the rate of unfertilized oocytes from the ovulation rate.
An exploratory analysis of multiple risk factors affecting the presence of embryonic mortality was performed using a logistic regression in R (R Core Team 2013, Table 4). Crude and adjusted odds ratios (OR) with 95 % confidence intervals were calculated to identify which factors (age of female, sampling area, sampling year, presence of twin embryos, presence of endometritis, and number of corpora lutea) were associated with embryonic mortality. Crude OR is an estimation of one specific factor without consideration and possible confounding effects of other factors. The adjusted OR is estimated with consideration of other factors (stated previously) included in the model. In addition, statistical significance calculations (P values) between the different risk factors were performed (Table 4).
Results
Sampled moose
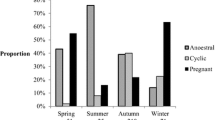
In total, 213 female moose were sampled. The majority of the samples (91.1 %, n = 194) were collected from Oct 10 to Nov 11, and the remaining samples (n = 19) were collected from Nov 12 to Dec 12. The mean age of sampled female moose was 4.2 years (range, 1.5–18.5).
Reproductive organ examinations
The majority of the investigated moose (67.3 %, n = 142) had ovulated (corpora lutea present, Table 1), and these animals were then included in the further analyses. A total of 53 (37.3 %) females that had ovulated fulfilled the criteria for being pregnant. The mean ovulation rate for the pregnant females was 1.49, and the mean rate of fertilized oocytes was 1.43 per capita. The proportion of unfertilized oocytes (ova loss) in pregnant moose was 6.3 %, the twinning rate was 43.4 %, and one triple ovulation (0.7 %) was noted. Embryonic mortality was observed in 16 individuals (30.9 % of pregnant moose), carrying a total of 19 (25 % of all embryos) malformed embryos, or remnants thereof (Table 2). The total loss of potential offspring due to unfertilized oocytes and embryonic mortality was 27.5 %, i.e., the discrepancy between the ovulation rate of pregnant females (1.49) and the early pregnancy rate per female (1.08).
Among the pregnant females, 12 (23.1 %) had macroscopical signs of inflammation of the inner mucous membrane of the uterus (endometritis), which was confirmed by microscopical examination, and eight of those animals also had embryonic mortality (Table 3). Hence, 50 % of the females with embryonic mortality also had endometritis (Table 3). Aerobic cultivation of bacteriological samples (n = 8) from uteri was negative. The risk of embryonic mortality increased with presence of endometritis (P < 0.001). Also, the risk of endometritis increased with female age (P = 0.003; Table 4). In addition, a temporal effect on the presence of embryonic mortality (P = 0.009) was observed.
Discussion
Ova loss and embryonic mortality
Here, we show that reproductive failure in early pregnancy, commonly described as ova loss, is common in Scandinavian moose. We also show that the major part of the loss was attributed to the embryonic mortality and not to the actual loss of an oocyte. In accordance with domestic animal reproductive terminology, we also suggest that ova loss is to be defined as when an oocyte fails to become fertilized by a sperm (conception failure), or when a fertilized oocyte due to impaired quality is terminated in very early pregnancy (approx. <12 days since conception) (Schillo 2009). Embryonic mortality (or embryo loss) is defined as mortality after this period, and can occur until the embryo becomes a fetus, which occurs when the fetus has identifiable features of a given species (Schillo 2009). In moose, embryonic mortality has been discussed as a plausible cause of discrepancies between ovulation rate and embryo/fetal rate (Edwards and Ritcey 1958; Markgren 1969; Pimlott 1959; Schwartz and Hundertmark 1993; Simkin 1965). Markgren (1969) noted a 19 % ova loss in Scandinavian moose, and Schwartz and Hundertmark (1993) estimated a 9.3 % loss in Alaskan moose (Alces alces gigas). Few reports on the detection of one or more nonviable embryos exist to date, most likely due to practical difficulties in repeated testing (by rectal ultrasonography) of female moose in the wild. Another cause, which may contribute to an underreporting of the event, could be difficulties in differentiating a nonviable from a viable embryo when the embryonic membranes are intact. Hence, previous reports on ova loss may include undetected events of embryonic mortality. Also, thorough examination of an embryo in order to detect failure of organogenesis may not have been performed, and the embryo has been labeled as normal/viable instead of the opposite. Testa and Adams (1998) did however detect a presumed dead embryo using rectal ultrasonography in immobilized Alaskan moose, and one case of fetal mortality was reported by Schwartz and Hundertmark (1993). In elk (Cervus canadensis), Trainer (1971) described embryonic mortality on two occasions (2.5 % of 79 examined females), and Woolf and Harder (1979) also reported a low (1.6 % of 60 examined animals) presence of embryonic mortality in white-tailed deer (Odocoileus virginianus). Furthermore, embryonic mortality has been diagnosed in captive fallow deer (Dama dama), where Willard et al. (1998) used repeated hormone analyses (progesterone, estrone sulfate, pregnancy-specific protein) and transrectal ultrasonography to detect embryonic mortality in 2 out of 10 pregnant females. In free-ranging caribou (Rangifer tarandus), Russell et al. (1998) reported embryonic mortality in 8.5 % (12/141) of the investigated radio-collared females, using repeated hormone analyses (progesterone, pregnancy-specific protein). Using transrectal ultrasonography after mating, and studying subsequent parturition rates, Gómez-Nieto et al. (2011) stated that 13.0 % (6/46) of captive red deer (Cervus elaphus) most likely had experienced embryonic mortality. Embryonic mortality and lack of fertilization of oocytes are well-known occurrences in domestic species, e.g., cattle (Diskin et al. 2011; Hanly 1961; King 1991), sheep (Cognie et al. 1975; Dutt and Simpson 1957; Vázquez et al. 2009; Viñoles et al. 2012), goats (Armstrong and Evans 1983; Gonz et al. 2004; Shelton 1978), pigs (Dziuk 1968; Geisert et al. 2007; Soede et al. 1994), and horses (Ball et al. 1986; Dippert et al. 1994; Newcombe and Cuervo-Arango 2011), and underlying causes of may be infectious (bacteria, virus, parasites, fungi) or of genetic, endocrine, and environmental origin (Diskin et al. 2011).
Inflammation of the uterus
Endometritis, defined as inflammation of the inner mucous membrane in the uterus, is a condition observed in domestic animals. In dairy cattle, it often occurs postpartum, where bacteria of different species enter the uterus and cause an infection. The association between endometritis and embryonic mortality was considerable, as 50 % (8/16, Table 2) of all cases of embryonic mortality also was accompanied by microscopically confirmed endometritis. In addition, the risk of endometritis (and embryonic mortality) seems to increase with age. It is known from domestic animals that embryos do not survive in uteri with endometritis. In the present study, the aerobic culturing of samples from uteri was negative. Hence, the cause of endometritis in the sampled female moose was not fully elucidated but may include an anaerobic bacterial infection introduced at mating or after mating. In the latter case, bacteria can ascend from the vagina into the uterus in females where the neck of the uterus (cervix uteri) does not function as a proper barrier. To our knowledge, endometritis has never been reported in wild cervids. McShea et al. (1997) reported endometritis in a captive white-tailed deer, which had been given immunocontraceptives. The considerably high presence of endometritis in moose in the present study indicates that this condition most likely is underdiagnosed in other ungulates.
Moose productivity estimation
The use of estimation of productivity (reproductive rates) of moose populations by ovulation rates is common in Scandinavia and North America (Ferguson et al. 2000; Saether and Haagenrud 1985; Sand and Cederlund 1996; Simkin 1965). However, based on the high rate of early reproductive failure seen in the present study, an overestimation of future numbers of offspring is possible. Though rarely reported, (Schwartz and Hundertmark 1993) fetal loss must also be considered, which if occurring, affects future offspring numbers in a population.
Causes of embryonic mortality
The reasons for the embryonic mortality in the present study are most likely multifactorial and could include impaired quality of the oocyte or spermatozoa at fertilization as well as an inadequate maternal recognition of pregnancy. In sheep, the rate of very early losses (until the early embryonic stage) is approximately 5–10 %, which is considered acceptable from a management point of view (Diskin and Morris 2008). However, in lactating dairy cows, the loss rate has been reported to be around 20 %, where the marked increase in milk yield over the past 40 years, has been accompanied by a decrease in fertility (Diskin and Morris 2008). Understandably, this relationship is not applicable for moose in the wild, and hence the causes are of another nature. Uterine malformations as a cause of embryo mortality were not identified in the present study, and chromosomal aberrations, which can lead to early embryonic mortality (Diskin and Morris 2008), were not investigated in the dead embryos of the present study. A temporal effect (year of sampling) of embryonic mortality was shown in the present study, where the proportion of females with embryonic mortality differed between years. This effect is likely due to annual differences in the age structure of sampled moose.
Conclusion
In conclusion, we verified our hypothesis stating that loss of ova is not only attributed to unfertilization of oocytes (ova loss) but also dependent on embryo mortality and survival. Also, we verified the second hypothesis and showed that there is a discrepancy between ovulation rates and embryo survival rates.
Ovulation rates in moose are substantially higher than the actual proportion of normally developed embryos in the uterus due to embryonic mortality and unfertilized oocytes. This should be taken into consideration when trying to estimate moose population productivity for management purposes. Reproductive tract investigations solely focusing on the presence of corpora lutea may lead to misinterpretations of moose productivity.
References
Armstrong DT, Evans G (1983) Factors influencing success of embryo transfer in sheep and goats. Theriogenology 19:31–42
Ball BA, Little TV, Hillman RB, Woods GL (1986) Pregnancy rates at days 2 and 14 and estimated embryonic loss rates prior to day 14 in normal and subfertile mares. Theriogenology 26:611–619
Cognie Y, Hernandez-Barreto M, Saumande J (1975) Low fertility in nursing ewes during the non-breeding season. Ann Biol Anim Biochim Biophys 15:329–343
Dippert KD, Jasko DJ, Seidel GE, Squires EL (1994) Fertilization rates in superovulated and spontaneously ovulating mares. Theriogenology 41:1411–1423
Diskin MG, Morris DG (2008) Embryonic and early foetal losses in cattle and other ruminants. Reprod Dom Anim 43:260–267
Diskin MG, Parr MH, Morris DG (2011) Embryo death in cattle: an update. Reprod Fertil Dev 4:244–251
Dutt R, Simpson EC (1957) Environmental temperature and fertility of Southdown rams early in the breeding season. J Anim Sci 16:136–143
Dziuk PJ (1968) Effect of number of embryos and uterine space on embryo survival in the pig. J Anim Sci 27:673–676
Edwards RY, Ritcey RW (1958) Reproduction in a moose population. J Wildl Manag 22:261–268
Ericsson G, Wallin K (1999) Hunter observations as an index of moose Alces alces population parameters. Wildlife Biol 5:177–185
Ferguson SH, Bisset RB, Messier F (2000) The influences of density of growth and reproduction in moose Alces alces. Wildlife Biol 6:31–39
Geisert RD, Sasakia S, Clutter AC (2007) Embryonic loss in sows with repeated propensity for small litter size. Reprod Domest Anim 42:333–335
Gómez-Nieto JA, Santiago-Moreno J, Landete-Castillejos T, Gallego-Martinez L, Garcia-Diaz AJ (2011) Real-time ultrasonography for early pregnancy diagnosis and incidence of embryonic/foetal mortality in farmed Iberian red deer hinds. Span J Agric Res 9:1182–1185
Gonz A, Baird DT, Campbell BK, Cocero MJ, García-García RM, Inskeep EK, Veiga-López A (2004) Multiple factors affecting the efficiency of multiple ovulation and embryo transfer in sheep and goats. Reprod Fertil Dev 16:421–435
Hanly S (1961) Prenatal mortality in farm animals. J Reprod Fertil 2:182–194
King WA (1991) Embryo-mediated pregnancy failure in cattle. Can Vet J 32:99
Markgren G (1969) Reproduction of moose in Sweden. Viltrevy 6:127–299
McShea WJ, Monfort SL, Hakim S, Kirkpatrick J, Liu I, Turner JW Jr, Munson L (1997) The effect of immunocontraception on the behavior and reproduction of white-tailed deer. J Wildl Manag 61:560–569
Newcombe JR, Cuervo-Arango J (2011) The effect of time of insemination with fresh cooled transported semen and natural mating relative to ovulation on pregnancy and embryo loss rates in the mare. Reprod Dom Anim 46:678–681
Pimlott DH (1959) Reproduction and productivity of Newfoundland moose. J Wildl Manag 23:381–401
R Core Team (2013) R: A Language and Environment for Statistical Computing. R Foundation for Statistical Computing, Vienna, http://www.R-project.org/. ISBN 3-900051-07-0
Russell DE, Gerhart KL, White RG, Van de Wetering D (1998) Detection of early pregnancy in caribou: evidence for embryonic mortality. J Wildl Manag 62:1066–1075
Saether BE, Haagenrud H (1985) Life history of the moose Alces alces: relationship between growth and reproduction. Ecography 8:100–106
Sand H, Cederlund G (1996) Individual and geographical variation in age at maturity in female moose (Alces alces). Can J Zool 74:954–964
Schillo KK (2009) Sexual differentiation. In: Schillo KK (ed) Reproductive physiology of mammals. From farm to field and beyond. Delmar, Clifton Park, pp 56–57
Schwartz CC, Hundertmark KJ (1993) Reproductive characteristics of Alaskan moose. J Wildl Manag 57:454–468
Shelton M (1978) Reproduction and breeding of goats. J Dairy Sci 61:994–1010
Simkin DW (1965) Reproduction and productivity of moose in northwestern Ontario. J Wildl Manag 29:740–750
Soede NM, Helmond FA, Kemp B (1994) Periovulatory profiles of oestradiol, LH and progesterone in relation to oestrus and embryo mortality in multiparous sows using transrectal ultrasonography to detect ovulation. J Reprod Fertil 101:633–641
Testa JW, Adams GP (1998) Body condition and adjustments to reproductive effort in female moose (Alces alces). J Mammal 79:1345–1354
Trainer CE (1971) The relationship of physical condition and fertility of female Roosevelt elk (Cervus canadensis roosevelti) in Oregon. M.S.thesis, Oregon State Univ., Corvallis, USA
Vázquez MI, Forcada F, Casao A, Sosa C, Palacín I, Abecia JA (2009) Effects of melatonin and undernutrition on the viability of ovine embryos during anestrus and the breeding season. Anim Reprod Sci 112:83–94
Viñoles C, Glover KM, Paganoni BL, Milton JT, Martin GB (2012) Embryo losses in sheep during short-term nutritional supplementation. Reprod Fertil Dev 24:1040–1047
Willard ST, Sasser RG, Jaques JT, White DR, Neuendorff DA, Randel RD (1998) Early pregnancy detection and the hormonal characterization of embryonic-fetal mortality in fallow deer (Dama dama). Theriogenology 49:861–869
Wolfe ML (1969) Age determination in moose from cemental layers of molar teeth. J Wildl Manag 33:428–431
Woolf A, Harder JD (1979) Population dynamics of a captive white-tailed deer herd with emphasis on reproduction and mortality. Wildlife Monogr 67:3–53
Acknowledgments
The authors would like to express their sincere gratitude to the hunters of southern Sweden for assistance in the sample collection, to Gustav Rydevik for advice in statistical analysis, to Carl-Gustaf Thulin and Lennart Söderquist for constructive and valuable comments on the manuscript, and Jane Morrell for improving the language. The study received financial support from the Swedish Environmental Protection Agency, the Environmental monitoring and assessment program at the Swedish University of Agricultural Sciences, and the Wildtech Project (European Union Seventh Framework Programme (FP7/2007-2013) grant agreement no. 222633) and the Swedish National Veterinary Institute (SVA).
Conflict of interest
The authors declare that they have no conflicts of interest.
Author information
Authors and Affiliations
Corresponding author
Additional information
Communicated by: Kris Hundertmark
Rights and permissions
Open Access This article is distributed under the terms of the Creative Commons Attribution License which permits any use, distribution, and reproduction in any medium, provided the original author(s) and the source are credited.
About this article
Cite this article
Malmsten, J., Dalin, AM. Reproductive failure in moose (Alces alces) due to embryonic mortality and unfertilized oocytes. Acta Theriol 59, 449–455 (2014). https://doi.org/10.1007/s13364-013-0173-6
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s13364-013-0173-6