Abstract
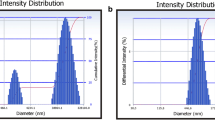
The objective of the present work was to develop polymeric nanoparticles of uricase and aceclofenac (NSAID) and to incorporate them into gel, for delivering drugs to synovial joints, for effective treatment of Gout. Nanoparticles containing uricase and aceclofenac were prepared by double emulsion solvent evaporation method and emulsion solvent evaporation, using PLGA (50:50) as carrier, respectively. Process parameters were optimized using Taguchi L4 orthogonal array and L9 array, respectively. The formulations were characterized for particle size, entrapment efficiency, surface charge, in vitro drug release, ex vivo drug permeation, and urate crystal degradation activity. The particle size and entrapment efficiency for optimized batch was found to be 228.8 nm and 81.26 % for uricase nanoparticles and 288.5 nm and 85.36 % for aceclofenac nanoparticles, respectively. The developed nanoparticles formulations displayed zero order and Higuchi release kinetics with non-Fickian diffusion, respectively. The in vivo studies were performed in rabbit model. Topical application of gel containing polymeric uricase nanoparticles alone and a combination of both, uricase nanoparticles and aceclofenac nanoparticles in rabbit model test groups, provided complete removal of urate crystals and inflammation within 40 and 25 days of treatment, respectively. The combination treatment therapy resulted in effective treatment of gout due to degradation of crystals and anti-inflammatory response.



Similar content being viewed by others
References
Christopher MB, Robert LW. Gout therapeutics: new drugs for an old disease. Lancet. 2010;377:165–77.
Merry RS, Mark GPS, Fernando PR. PEG-uricase in the management of treatment-resistant gout and hyperuricemia. Adv Drug Deliv Rev. 2008;60:59–68.
Tarl WP. Nanoparticles and microparticles for skin drug delivery. Adv Drug Deliv Rev. 2011;63:470–91.
Zhang J, Tan Q, Wang N, Xiaoling L, Huarong X, Teng Y, et al. Uricase from Bacillus fastidious loaded in alkaline enzymosomes: enhanced biochemical and pharmacological characteristics in hypouricemic rats. Eur J Pharm Biopharm. 2012;82:43–8.
Tan QY, Wang N, Yang H, Zhang LK. Characterization, stabilization and activity of uricase loaded in lipid vesicles. Int J Pharm. 2010;384:165–72.
Seegmiller JE, Howell RR, Malawista SE. The inflammatory reaction to sodium urate. J Am Med Assoc. 1962;180:469–75.
Chittasupho C, Xie S, Baoum A, Yakovleva T, Siahaan JT, Berkland C. ICAM-1 targeting of doxorubicin-loaded PLGA nanoparticles to lung epithelial cells. Eur J Pharm Sci. 2009;37:141–50.
Sabina EP, Rasool MK, Mathew L, Ezilrani P, Indu H. 6-Shogaol inhibits monosodium urate crystal-induced inflammation—an in vivo and in vitro study. Food Chem Toxicol. 2010;48:229–35.
Ansari SA, Husain Q. Potential applications of enzymes immobilized on/in nanomaterials: a review. Biotechnol Adv. 2012;30:512–23.
Mohanraj VJ, Chen Y. Review on nanoparticles. Trop J Pharm Res. 2006;5:561–73.
Abdel-Mottaleb MM, Moulari B, Beduneau A, Pellequer Y, Lamprecht A. Nanoparticles enhance therapeutic outcome in inflamed skin therapy. Eur J Pharm Biopharm. 2012;82:151–7.
Abdullah D, Ping Q, Liu G. Studies on the interaction between eucalyptus oil and liquid crystals of skin lipids. J Chin Pharm Sci. 1999;8:135–41.
Oliviero F, Scanu A, Punzi L. Metabolism of crystals within the joint. Reumatismo. 2011;63:221–9.
Bonnete F. Macromolecular crystallization controlled by colloidal interactions: the case of urate oxidase. In: Marcello RBA, editor. Crystallization – science and technology. France: InTech-Open Access Company; 2012. p. 349–78.
Acknowledgments
The authors are thankful to Evonik Degussa India Pvt. Ltd., Mumbai, for providing us the gift sample of PLGA (50:50) for this research work.
Conflict of interest
The authors Sanjay Tiwari, Harinath Dwivedi, Koshy M Kymonil, and Shubhini A Saraf declare that they have no conflict of interest.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Tiwari, S., Dwivedi, H., Kymonil, K.M. et al. Urate crystal degradation for treatment of gout: a nanoparticulate combination therapy approach. Drug Deliv. and Transl. Res. 5, 219–230 (2015). https://doi.org/10.1007/s13346-015-0219-1
Published:
Issue Date:
DOI: https://doi.org/10.1007/s13346-015-0219-1