Abstract
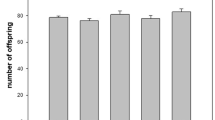
Bisphenol-A(BPA) classified as an endocrine disruptor that can interfere endocrine system, leading to adverse developmental, reproductive, neurological, and immunological effects in both humans and wildlife. To study the reproductive and geno-toxicological effects of BPA in aquatic ecosystem, we employed Daphnia magna (D. magna) as a test animal model. D. magna in one of the most widely used water species employed in ecotoxicology testing all over the world. As a result, the reproductive activity based on the number of offspring production is significantly reduced toward 21 day exposure to 10 mg/L of BPA, compared with control. Gene expression profiles in response to BPA exposure using DNA microarray analysis was subsequently carried out. Our data showed that expression levels of numerous putative genes with nucleotide sequences homology to animal models including cuticular protein, vitellogenin, protease, ribosomal protein family were altered. These genes which are critically reported in other animal systems could be considered as novel biomarkers in response to BPA exposure.
Similar content being viewed by others
References
Watanabe, H. et al. Development of a daphnia magna DNA microarray for evaluating the toxicity of environmental chemicals. Environ Toxicol Chem 4:669–676 (2007).
Colborn, T., Dumanoski, D. & Myers, J. P. Our stolen future. New York: Dutton signet (1996).
Ikezuki, Y., Tsutsumi, O., Takai, Y., Kamei, Y. & Taketani, Y. Determination of bisphenol A concentrations in human biological fluids reveals significant early prenatal exposure. Hum Reprod 17:2839–2841 (2002).
Schonfelder, G. et al. Parental Bisphenol-A accumulation in the human-maternal-fetal-placental unit. Environ Health Perspect 11:703–707 (2002).
Paulozzi, L. J., Erickson, J. D. & Jackson, R. J. Hypospadias trends in two US surveillance systems. Pediatrics 100:831–834 (1997).
Herman-Giddens, M. E. et al. Secondary sexual characteristics and menses in young girls seen in office practice: a study from the pediatric research in office settings network. Pediatrics 99:505–512 (1997).
Howdeshell, K. L., Hotchkiss, A. K., Thayer, K. A., Vandenbergh, J. G. & vom Saal, F. S. Exposure to bisphenol A advances puberty. Nature 401:763–764 (1999).
Dorn, P. B., Chou, C. S. & Gentempo, J. J. Degradation of bisphenol-A in natural waters. J Chemosphere 16:1501–1507 (1987).
Alexander, H. C., Dill, D. C., Smith, L. W., Guiney, P. D. & Dorn, P. Bisphenol-A: Acute aquatic toxicity. Environ Toxicol Chem 7:19–26 (1998).
Lilius, H., Hastbacka, T. & Isomaa, B. A comparison of the toxicity of 30 reference chemicals to Daphnia magna and Daphnia pulex. Environ Toxicol Chem 14:2085–2088 (1995).
U.S. EPA. Quality criteria for water. EPA 440/5-86-001 (1986).
U.S. EPA. Methods for measuring the acute toxicity of effluents and receiving waters to freshwater and marine organisms. EPA/600/4-89/001 (1993).
OECD (Organization for Economic Co-operation and Development). OECD guidelines for testing of chemicals. 202. Daphnia sp. Acute immobilization and reproduction test. OECD, Paris, France (1981).
OECD. OECD guidelines for testing of chemicals. 211. Daphnia magna reproduction test. OECD, Paris, France (1998).
Tatarazako, N. & Oda, S. The water flea Daphnia magna (Crustacea, Cladocera) as a test species for screening and evaluation of chemicals with endocrine disrupting effects on crustaceans. Ecotoxicology 16:197–203 (2007).
Seo, Y. R., Kim, B. J. & Ryu, J. C. Molecular and genomic approaches on nickel toxicity and carcinogenicity. Mol Cell Toxicol 1:73–77 (2005).
Conesa, A. et al. Blast2GO: a universal tool for annotation, visualization and analysis in functional genomics research. Bioinformatics 18:3674–3676 (2005).
Poynton, H. C. et al. Gene expression profiling in daphnia magna Part l: Concentration-dependent profiles provide support for the no observed transcriptional effect level. Environ Sci Technol 42:6250–6256 (2008).
Merzendorfer, H. & Zimoch, L. Chitin metabolism in insect: structure, function and regulation of chitin synthases and chitinase. J Exp Biol 206:4393–4412 (2003).
Havemann, J. et al. Cuticle differentiation in the embryo of the amphipod crustacean Parhyale hawaiensis. Cell Tissue Res 332:359–370 (2008).
Locke, M. The Wigglesworth lecture: Insects for studying fundamental problems in biology. J Insect Physiol 47:495–507 (2001).
Connon, R. et al. Linking molecular and population stress responses in Daphnia magna exposed to cadmium. Environ Sci Technol 42:2181–2188 (2008).
Soetaert, A. et al. Molecular responses during cadmium-induced stress in Daphnia magna: Integration of differential gene expression with higher-level effects. Aquat Toxicol 83:212–222 (2007).
Depledge, M. H., Galloway, T. S. & Billinghurst, Z. Effects of endocrine disrupting chemicals in invertebrates. Issue Environ Sci Technol 12:49–60 (1999).
Smida, A. D. et al. Cadmium stimulates transcription of the cytochrome P450 side chain cleavage gene in genetically modified stable Porcine granulose cells. Biol Reprod 70:25–31 (2004).
Kato, Y., Tokishita, S., Ohta, T. & Yamagata, H. A vitellogenin chain containing a superoxide dismutaselike domain is the major component of yolk proteins in cladoceran crustacean Daphnia magna. Gene 334: 157–165 (2004).
Tokishita, S. et al. Organization and repression by juvenile hormone of vitellogenin gene cluster in the crustacean, Daphnia magna. Biochem Biophys Res Commun 345:362–370 (2006).
Huang, H. F. et al. Function of aquaporins in female and male reproductive systems. Human Reproduction Update 6:785–795 (2006).
Soetaert, A. et al. Daphnia magna and ecotoxicogenomics: Gene expression profiles of the anti-ecdysteroidal fungicide fenarimol using energy-, molting- and life stage-related cDNA libraries. Chemosphere 67:60–71 (2007).
Ote, M. et al. Indentification of molting fluid carboxypeptidase A (MFCPA) in Bombyx mori. Comp Biochem Physiol B 141:314–322 (2005).
Tatarazako, N. et al. Juvenile hormone agonists affect the occurrence of male Daphnia. Chemosphere 53:827–833 (2003).
Van Gelder, R. N. et al. Amplified RNA synthesized from limited quantities of heterogeneous cDNA. Proc Natl Acad Sci U S A 87:1663–1667 (1990).
Baugh, L. R., Hill, A. A., Brown, E. L. & Hunter, C. P. Quantitative analysis of mRNA amplification by in vitro transcription. Nucleic Acids Res 29:E29 (2001).
Peixoto, B. R. et al. Evaluation of reference-based two-color methods for measurement of gene expression ratios using spotted cDNA microarrays. BMC Genomics 24:7–35 (2006).
Peng, X. et al. Statistical implications of pooling RNA samples for microarray experiments. BMC Bioinformatics 4:26 (2003).
Altschul, S. F. et al. Basic local alignment search tool. J Mol Biol 215:403–410 (1990).
Author information
Authors and Affiliations
Corresponding authors
Rights and permissions
About this article
Cite this article
Jeong, S.W., Lee, S.M., Yum, S.S. et al. Genomic expression responses toward bisphenol-A toxicity in Daphnia magna in terms of reproductive activity. Mol. Cell. Toxicol. 9, 149–158 (2013). https://doi.org/10.1007/s13273-013-0019-y
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s13273-013-0019-y