Abstract
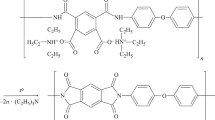
Tubular grafts based on nanofibers of copolymer of ε-caprolactam and hexamethylendiaminadipate were obtained by the electrospinning method. The strength of materials based on the dry nanofibers was 6.2 MPa with elongation at break of 133%, or 7.5 MPa and 299% in saline, respectively. The pressure value at which liquid started seeping through the tube wall was P = 10 kPa. Absence of cytotoxicity was proved, as well as adhesion and proliferation of mesenchymal stem cells on the surface. Tubes with inner diameter of 1 mm were tested in vivo in rat abdominal aorta. A layer of endothelial cells was shown to form on the inner side of the prosthesis after 30 days. There was no evidence of stenosis or dilatation of the prosthesis after 14 months with observation of endothelial and subendothelial layers.








Similar content being viewed by others
References
Armentano, I., M. Dottori, E. Fortunati, S. Mattioli, and J. M. Kenny. Biodegradable polymer matrix nanocomposites for tissue engineering: a review. Polym. Degrad. Stab. 95:2126–2146, 2010.
Battiston, K. G., B. Ouyang, R. S. Labow, C. A. Simmons, and J. P. Santerre. Monocyte/macrophage cytokine activity regulates vascular smooth muscle cell function within a degradable polyurethane scaffold. Acta Biomater. 10(3):1146–1155, 2014.
Baumgartner, P. K. Electrostatic spinning of acrylic microfibers. J. Colloid Interface Sci. 36:71–79, 1971.
Begovac, P. C., R. C. Thomson, J. L. Fisher, A. Hughson, and A. Gällhagen. Improvements in GORE-TEX vascular graft performance by Carmeda Bio Active surface heparin immobilization. Eur. J. Vasc. Endovasc. Surg. 25(5):432–437, 2003.
Bernard, M. C., and O. Brien. Microvascular Reconstructive Surgery. Edinburgh: Churchill Livingstone, p. 91, 1977.
Boccafoschi, F., J. Habermehl, S. Vesentini, and D. Mantovani. Biological performances of collagen-based scaffolds for vascular tissue engineering. Biomaterials 26:7410–7417, 2005.
Butson, R., and V. Sattinrar. Nonreversed and in situ vein grafts. Ann. Surg. 51:771–779, 1985.
Bykova, I., V. Weinhardt, A. Kashkarova, S. Lebedev, T. Baumbach, V. Pichugin, K. Zaitsev, and I. Khlusov. Physical properties and biocompatibility of UHMWPE-derived materials modified by synchrotron radiation. J. Mater. Sci. Mater. Med. 25(8):1843–1852, 2014.
Chung, S., N. P. Ingle, G. A. Montero, S. H. Kim, and M. W. King. Bioresorbable elastomeric vascular tissue engineering scaffolds via melt spinning and electrospinning. Acta Biomater. 6:1958–1967, 2010.
Davies, M. G., and P. O. Hagen. Pathophysiology of vein graft failure: a review. Eur. J. Vasc. Endovasc. Surg. 9:7–18, 1995.
Dobrovolskaya, I. P., P. V. Popryadukhin, V. E. Yudin, et al. Structure and properties of porous films based on aliphatic copolyamide developed for cellular technologies. J. Mater. Sci. Mater. Med. 26(1):1–10, 2015.
Geiger, G. Vascular grafts in below-knee femoro-popliteal bypass. J. Cardiovasc. Surg. 25:523–529, 1984.
Harskamp, R. E., R. D. Lopes, C. E. Baisden, R. J. Winter, and J. H. Alexander. Saphenous vein graft failure after coronary artery bypass surgery: pathophysiology, management, and future directions. Ann. Surg. 257(5):824–833, 2013.
Hassantash, S. A., B. Bikdeli, S. Kalantarian, M. Sadeghian, and H. Afshar. Pathophysiology of aortocoronary saphenous vein bypass graft disease. Asian Cardiovasc. Thorac. Ann. 16:331–338, 2008.
Kannan, R. Y., H. J. Salacinski, P. E. Butler, G. Hamilton, and A. M. Seifalian. Current status of prosthetic bypass grafts: a review. J. Biomed. Mater. Res. B Appl. Biomater. 74(1):570–581, 2005.
Kim, F. Y., G. Marhefka, N. J. Ruggiero, S. Adams, and D. J. Whellan. Saphenous vein graft disease: review of pathophysiology, prevention, and treatment. Cardiol. Rev. 21:101–109, 2013.
Konig, G., et al. Mechanical properties of completely autologous human tissue engineered blood vessels compared to human saphenous vein and mammary artery. Biomaterials 30:1542–1550, 2009.
Lamm, P., G. Juchem, S. Milz, M. Schuffenhauer, and B. Reichart. Autologous endothelialized vein allograft: a solution in the search for small-caliber grafts in coronary artery bypass graft operations. Circulation 104:108–114, 2001.
Larsen, C. C., F. Kligman, C. Tang, K. Kottke-Marchant, and R. E. Marchant. A biomimetic peptide fluorosurfactant polymer for endothelialization of ePTFE with limited platelet adhesion. Biomaterials 28(24):3537–3548, 2007.
Leon, L., and H. P. Greisler. Vascular grafts. Expert Rev. Cardiovasc. Ther. 1:581–594, 2003.
Leon, L., and H. P. Greisler. Vascular grafts. Expert Rev. Cardiovasc. Ther. 1(4):581–594, 2003.
Nerem, R. M., and D. Seliktar. Vascular tissue engineering. Annu. Rev. Biomed. Eng. 3:225–243, 2001.
Park, K. M., Y. W. Kim, S. S. Yang, and D. I. Kim. Comparisons between prosthetic vascular graft and saphenous vein graft in femoro-popliteal bypass. Ann. Surg. Treat. Res. 87:35–40, 2014.
Roh, J. D., G. N. Nelson, M. P. Brennan, T. L. Mirensky, T. Yi, T. Hazlett, et al. Small-diameter biodegradable scaffolds for functional vascular tissue engineering in the mouse model. Biomaterials 29(10):1454–1463, 2008.
Sabiston, A. The Biological Basis of Modern Surgical Practice. Philadelphia: W.B. Saunders, pp. 203–218, 1997.
Taylor, L. M., J. M. Edwards, and J. M. Porter. Present status of reversed vein bypass grafting: five-year results of a modern series. J. Vasc. Surg. 11:193–205, 1990.
Tschoeke, B., T. C. Flanagan, S. Koch, M. S. Harwoko, T. Deichmann, V. Ella, S. Jorg, S. M. Kellomaki, T. Gries, T. Schmitz-Rode, and S. Jockenhoevel. Tissue-engineered small-caliber vascular graft based on a novel biodegradable composite fibrin-polylactide scaffold. Tissue Eng. A 15(8):326–331, 2009.
Acknowledgments
The authors are grateful to the Russian Science Foundation under contract grant #14-03-00003. The authors would like to thank Prof. Textor Marcus for discussions and assistance in preparing the manuscript.
Conflict of interest
We declare that there is no conflict of interest with any organization regarding the material discussed in this manuscript.
Statement of Human Studies
No human studies in this study.
Statement of Animal Studies
We declare that the study submitted to Cardiovascular Engineering and Technology complies with the principles laid down in the Declaration of Helsinki. Animal research approved by the Animal Care and Use Committee (IACUC) of Gateway Medical Innovation Center (Shanghai, China).
Author information
Authors and Affiliations
Corresponding author
Additional information
Associate Editor Ajit P. Yoganathan oversaw the review of this article.
Rights and permissions
About this article
Cite this article
Popryadukhin, P.V., Popov, G.I., Dobrovolskaya, I.P. et al. Vascular Prostheses Based on Nanofibers from Aliphatic Copolyamide. Cardiovasc Eng Tech 7, 78–86 (2016). https://doi.org/10.1007/s13239-015-0234-x
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s13239-015-0234-x