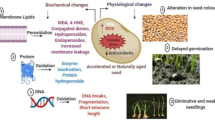
Abstract
To elucidate the mechanism responsible for magnetic field induced seed invigoration in aged seeds an experiment was conducted on six year old garden pea seeds stored under controlled (20 °C and 40% RH) condition. Aged seeds were magnetoprimed by exposing to pulsed magnetic field (PMF) of 100 mT for 1 h in three pulsed modes. The 6 min on and off PMF showed significant improvement in germination (7.6%) and vigor (84.8%) over aged seeds. Superoxide and hydrogen peroxide production increased in germinating primed seeds by 27 and 52%, respectively, over aged seeds. Nicotinamide adenine dinucleotide (reduced) (NADH) peroxidase and superoxide dismutase involved in generation of hydrogen peroxide showed increased activity in PMF primed seeds. Increase in catalase, ascorbate peroxidase and glutathione reductase activity after 36 h of imbibition in primed seeds demonstrated its involvement in seed recovery during magnetopriming. An increase in total antioxidants also helped in maintaining the level of free radicals for promoting germination of magnetoprimed seeds. A 44% increase in level of protein carbonyls after 36 h indicated involvement of protein oxidation for counteracting and/or utilizing the production of ROS and faster mobilization of reserve proteins. Higher production of free radicals in primed seeds did not cause lipid peroxidation as malondialdehyde content was low. Lipoxygenase was involved in the germination associated events as the magnitude of activity was higher in primed aged seeds compared to aged seeds. Our study elucidated that PMF mediated improvement in seed quality of aged pea seeds was facilitated by fine tuning of free radicals by the antioxidant defense system and protein oxidation.





Similar content being viewed by others
References
Abdul-Baki AA, Anderson JD (1973) Vigour determination in soybean by multiple criteria. Crop Sci 10:31–34
Aebi H (1984) Catalase in vitro. Method Enzymol 105:121–126
Apak R, Guclu K, Ozyurek M, Celik SE (2008) Mechanism of antioxidant capacity assays and the CUPRAC (cupric ion reducing antioxidant capacity) assay. Microchim Acta 160:413–419
Bailly C, Benamar A, Corbineau F, Côme D (1998) Free radical scavenging as affected by accelerated ageing and subsequent priming in sunflower seeds. Physiol Plant 104:646–652
Bailly C, Benamar A, Corbineau C, Come D (2000) Antioxidant systems in sunflower (Helianthus annuus L.) seeds as affected by priming. Seed Sci Res 10:35–42
Bailly C, Bogatek-Leszczynska R, Côme D, Corbineau F (2002) Changes in activities of antioxidant enzymes and lipoxygenase during growth of sunflower seedlings from seeds of different vigour. Seed Sci Res 12:47–55
Bailly C, El-Maarouf-Bouteau H, Corbineau F (2008) From intracellular signaling networks to cell death: the dual role of reactive oxygen species in seed physiology. C R Biol 331:806–814
Barba-Espin G, Diaz-Vivancos P, Job D, Belghazi M, Job C, Hernandez JA (2011) Understanding the role of H2O2 during pea seeds germination: a combined proteomic and hormone profiling approach. Plant Cell Environ 34:1907–1919
Barba-Espin G, Antonio Hernandez J, Diaz-Vivancos P (2012) Role of H2O2 in pea seed germination. Plant Signal Behav 7:193–195
Bhardwaj J, Anand A, Nagarajan S (2012) Biochemical and biophysical changes associated with magnetopriming in germinating cucumber seeds. Plant Physiol Biochem 57:67–73
Bolwell GP, Wojtaszek P (1997) Mechanisms for the generation of reactive oxygen species in plant defence—a broad perspective. Physiol Mol Plant Pathol 51:347–366
Bradford MM (1976) A rapid and sensitive for the quantitation of microgram quantities of protein utilizing the principle of protein-dye-binding. Anal Biochem 72:248–254
De Marco A, Roubelakis-Angelakis KA (1996) The complexity of enzymatic control of hydrogen peroxide concentration may affect the regeneration potential of plant protoplast. Plant Physiol 110:137–145
Dell’Aquila A, Taranto G (1986) Cell division and DNA-synthesis during osmopriming treatment and following germination in aged wheat embryos. Seed Sci Technol 14:333–341
Dhindsa RS, Plumb-Dhindsa P, Thorpe TA (1981) Leaf senescence: correlated with increased levels of membrane permeability and lipid peroxidation, and increased levels of superoxide dismutase and catalase. J Exp Bot 32:93–101
FAOSTAT (2013) Food and Agriculture Organisation. http://faostat3.fao.org
Florez M, Carbonell MV, Martinez E (2007) Exposure of maize seeds to stationary magnetic fields: effects on germination and early growth. Environ Exp Bot 59:68–75
Fujikura Y, Karssen CM (1992) Effect of controlled deterioration and osmopriming on protein synthesis of cauliflower during early germination. Seed Sci Res 2:23–31
Gallego SM, Benavides MP, Tomaro ML (1996) Effect of heavy metal ion excess on sunflower leaves: evidence for involvement of oxidative stresss. Plant Sci 121:151–159
Heath RL, Packer L (1968) Photoperoxidation in isolated chloroplast. I. Kinetics and stoichiometry of fatty acid peroxidation. Arch Biochem Biophys 125:189–198
International Seed Testing Association (1985) Seed Sci Technol 13:299–513
Ishida N, Kano H, Kobayashi T, Yoshida T (1988) Analysis of physical states of water in soybean seeds by NMR. Agri Biol Chem 52:2777–2781
Job C, Rajjou L, Lovigny Y, Belghazi M, Job D (2005) Patterns of protein oxidation in Arabidopsis seeds and during germination. Plant Physiol 138:790–802
Kibinza S, Bazin J, Bailly C, Farrant JM, Corbineau F (2011) Catalase is a key enzyme in seed recovery from ageing during priming. Plant Sci 181:309–315
Kumar M, Singh B, Ahuja S, Dahuja A, Anand A (2014) Gamma radiation and magnetic field mediated delay in effect of accelerated ageing of soybean. J Food Sci Technol 52:4785–4796
Levine A, Tenhaken R, Dixon R, Lamb CJ (1994) H2O2 from the oxidative burst orchestrates the plant hypersensitive disease resistance response. Cell 79:583–593
Mittler R, Vanderauwera S, Gollery M, Van Breusegem F (2004) Reactive oxygen gene network of plants. Trends Plant Sci 9:490–496
Mukherjee SP, Choudhari MA (1983) Implications of water stressed induced changes in the levels of endogenous ascorbic acid and hydrogen peroxide in Vigna seedlings. Physiol Plant 58:166–170
Nagarajan S, Bhardwaj J, Anand A, Pandita VK, Verma APS (2012) Static magnetic field exposure improves germination and vigour of fresh and carry over seeds of garden pea. Ind J Hort 69:435–438
Nakano Y, Asada K (1981) Hydrogen peroxide is scavenged by ascorbate peroxidase in spinach chloroplasts. Plant Cell Physiol 22:867–880
Radhakrishnan R, Ranjitha Kumari BD (2012) Pulsed magnetic field: a contemporary approach offers to enhance plant growth and yield of soybean. Plant Physiol Biochem 51:139–144
Rodriguez-Rosales MP, Kerkeb L, Ferrol N, Donaire JP (1998) Lipoxygenase activity and lipid composition of cotyledons and oil bodies of two sunflower hybrids. Plant Physiol Biochem 36:285–291
Rosahl S (1996) Lipoxygenase in plants-their role in development and stress response. Zeitschrift fur Naturforschung 51:123–138
Schopfer P (2001) Hydroxyl radical-induced cell-wall loosening in vitro and in vivo: implications for the control of elongation growth. Plant J 28:679–688
Schraufstatter IU, Hinshaw DB, Hyslop PA, Spagg R, Cochrane CG (1985) Glutathione cycle activity and pyridine nucleotide levels in oxidant-induced injury of cells. J Clin Invest 76:1131–1139
Shine MB, Guruprasad KN, Anand A (2011) Enhancement of germination, growth, and photosynthesis in soybean by pre-treatment of seeds with magnetic field. Bioelectromagnetics 32:474–484
Shine MB, Guruprasad KN, Anand A (2012) Effect of stationary magnetic field strengths of 150 and 200 mT on reactive oxygen species production in soybean. Bioelectromagnetics 33:428–437
Siedow JN (1991) Plant lipoxygenase: structure and function. Ann Rev Plant Physiol Plant Mol Biol 42:145–188
Smith IK, Vierheller TL, Thorne C (1998) Assay of glutathione reductase in crude tissue homogenates using 5, 5′-dithiobis (2-nitrobenzoic acid). Anal Biochem 175:408–413
Acknowledgements
The authors thank Indian Council of Agricultural Research, New Delhi, India for financial Grant under National Fund for Basic and Strategic Research in Agriculture, Grant Number: NFBSRA/PCN/AP-09/2006-07.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Bhardwaj, J., Anand, A., Pandita, V.K. et al. Pulsed magnetic field improves seed quality of aged green pea seeds by homeostasis of free radical content. J Food Sci Technol 53, 3969–3977 (2016). https://doi.org/10.1007/s13197-016-2392-8
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s13197-016-2392-8