Abstract
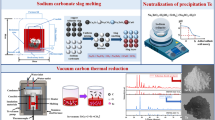
This hydrometallurgical method consists of oxidation leaching, sulfide impurities removing, and sulfur dioxide reduction. The crude tellurium powder was treated by H2O2 oxidation for 2.0 h at pH 2.5 when adding 50 ml H2O2 (30 %) per 100 g raw material, a tellurium recover rate around 91 % is achieved. The tellurium leaching ratio can reach 98.9 % under 3.75 mol·L−1 NaOH concentration in liquid–solid ratio of 5:1 at 80 °C for 1.5 h. The overall separation of tellurium and other heavy metals is optimum at sulfide dosages of about 1.1 times of the theoretical values. The removal rates of Ag, Ni, Pb, and Cu from the solution are greater than 99.8 %, and As and Se removal rates are 98.6 % and 97.2 %, respectively. Over 99.5 % tellurium can be recovered by SO2 reaction when the operation is conducted at 85 °C in 6 mol·L−1 HCl solution. The tellurium powder with size of <5 μm and purity of >99.999 % is obtained.








Similar content being viewed by others
References
Ba LA, Döring M, Jamier V, Jacob C. Tellurium: an element with great biological potency and potential. Org Biomol Chem. 2010;8(19):4203.
Jasinski SM. “Tellurium” from Mineral Commodities Summary, US Bureau of Mines, Minerals Yearbook. Pennsylvania: Bernan Assoc; 1995. 172.
Deng ZT, Zhang Y, Yue JC, Tang FQ, Wei Q. Green and orange CdTe quantum dots as effective pH-sensitive fluorescent probes for dual simultaneous and independent detection of viruses. J Phys Chem B. 2007;111(41):12024.
Bouroushian M. Electrochemistry of Metal Chalcogenides. In: Scholz F, editor. Berlin: Springer; 2010. 964.
Zhang H, Swihart MT. Synthesis of tellurium dioxide nanoparticles by spray pyrolysis. Chem Mater. 2007;19(6):1290.
Brown RD Jr. Selenium and Tellurium, US Geological Survey Minerals Yearbook. Pennsylvania: Bureau Assoc; 1994. 671.
Zhang Y, Li J, Hua YX, Meng GZ, Zhang Z, Wang Z. Thermodynamic analysis of gold telluride chlorination leaching process. Chin J Rare Met. 2001;37(3):461.
Pauporté T, Lincot D. Electrodeposition of semiconductors for optoelectronic devices: results on zinc oxide. Electrochim Acta. 2000;45(20):3345.
Patil PS. Versatility of chemical spray pyrolysis technique. Mater Chem Phys. 1999;59(3):185.
Liu W, Mitzi DB, Yuan M, Kellock AJ, Chey SJ, Gunawan O. 12% Efficiency CuIn(Se, S)2 photovoltaic device prepared using a hydrazine solution process. Chem Mater. 2009;22(3):1010.
Hoffmann JE. Selenium and tellurium—rare but ubiquitous. JOM. 1989;41(7):32.
Ha YC, Sohn HJ, Jeong GJ, Lee CK. RHEE K I. Electrowinning of tellurium from alkaline leach liquor of cemented Te. J Appl Electrochem. 2000;30(3):315.
Lingane JJ, Niedrach LW. Polarography of selenium and tellurium. II. The +4 states. J Am Chem Soc. 1949;71(1):196.
Panson AJ. A study of the telluride ion system. J Phys Chem. 1964;68(7):1721.
Panson AJ. Polarography of the ditelluride ion. J Phys Chem. 1963;67(10):2177.
Jamieson RA, Perone SP. Polarographic, coulometric, and stationary electrode studies of the electroreduction of Te (IV) in alkaline solution. J Electroanal Chem Interfacial Electrochem. 1969;23(3):441.
Mishra KK, Ham D, Rajeshwar K. Anodic oxidation of telluride ions in aqueous base: a rotating ring-disk electrode study. J Electrochem Soc. 1990;137(11):3438.
Sun Z, Zheng Y. Preparation of high pure tellurium from raw tellurium containing Cu and Se by chemical method. Trans Nonferrous Met Soc China. 2011;21(3):665.
Tang Q. Treatment of chromium containing wastewater with sulfur dioxide. Environ Prot Chem Ind. 1998;6:347.
Cheng K, Zheng Y, Sun Z. Recycling Se and Te and capturing Pt and Pd from solution after precipitating gold by SO2 reduction. Chin J Nonferrous Met. 2011;21(9):2258.
Acknowledgments
This work was financially supported by the Science and Technology Department of Guangdong Province (No. 2011B0508000033).
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Liu, Y., Zheng, YJ. & Sun, ZM. Preparation of high-purity tellurium powder by hydrometallurgical method. Rare Met. 33, 479–484 (2014). https://doi.org/10.1007/s12598-014-0338-8
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12598-014-0338-8