Abstract
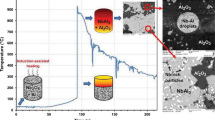
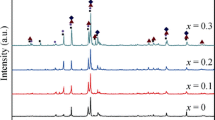
Sol-gel technology was employed to synthesize nanosized precursors of La-Mg hydrogen storage alloy at different pH values (0.5, 1.5, 8.0 and 9.0) of reaction solution. The effect of pH value on microstructure of the nano precursors of La-Mg hydrogen storage alloy was studied by infrared radiation (IR), thermo-gravimetric and differential thermal analysis (TG/DTA), X-ray diffraction analyzer (XRD) and transmission electron microscopy (TEM). IR results indicate that the chelating agent, citric acid, is not fully ionized, and carboxyl groups are not entirely used to complex metal ions in acidic solutions. The efficiency of complexing metal ions is enhanced in basic solutions. TG/DTA results show that the combustion may take place with low rate of the flame propagation that causes the longer combustion time when pH<1.5. On the contrary, the dry gel synthesized in basic solution combusts at low ignition temperature and combustion reaction is violent; it is easy to form fine particles. XRD and TEM results reveal that the precursor powders are mainly two-phase mixture of La2O3 and MgO. The morphology of the particles is almost flake with the size of ∼30 nm when pH is 8.0.
Similar content being viewed by others
References
Wu M.S., Wu H.R., Wang Y.Y., and Wan C.C., Effects of the stoichiometric ratio of aluminium and manganese on electrochemical properties of hydrogen storage alloys, Journal of Applied Electrochemistry, 2003, 33(7): 619.
Tetsuo S., Ituki U., and Hiroshi I., R&D on metal hydride materials and Ni-MH batteries in Japan, Journal of Alloys and Compounds, 1999, 293–295: 762.
Kim D.M., Jang K.J., and Lee J.Y., A review on the development of AB2-type Zr-based laves phase hydrogen storage alloys for Ni-MH rechargeable batteries in the Korea advanced of science and technology, Journal of Alloys and Compounds, 1999, 293–295: 583.
Zhu M., Progress on Mg base hydrogen storage alloy and its application in Ni-MH battery, Bulletin of National Science Foundation of China, 2001, 15(2): 83.
Zhang P.L., Song X.P., Wang X.L., Pei P., Luo G.P., Tu J.P., and Chen G.L.., Effect of rapid quenching on the microstructure and electrochemical characteristics of La0.6Ce0.4Ni3.6Co0.65Mn0.4Al0.2Ti0.05(FeB)0.1 hydrogen storage alloy, Rare Metals, 2010, 29(6): 593
Li J.H., Liu B.Z., Han S.M., Hu L., Zhao X., and Wang M.Z., Phase structure and hydrogen storage properties of LaMg3.70-Ni1.18 alloy, Rare Metals, 2011, 30(5): 458
Darriet B., Pezat M., Hbika A., and Hagenmuller P., Application of magnesium rich rare-earth alloys to hydrogen storage, International Journal of hydrogen Energy, 1980, 5(2): 173.
Chen Y., Cesar S., Chen C., and Wang Q.D., Electrochemical properties of the ball-milled La1.8Ca0.2Mg14Ni3+xwt%Ni composites (x=0,50,100,200), Journal of Alloys and Compounds, 2003, 354(1–2): 120.
Pal K., Synthesis, characterization and dehydriding behavior of the intermetallic compound LaMg12, International Journal of hydrogen Energy, 1997, 22(8): 799.
Zhang Y., Luo Y.C., Wang D.H., Zhang F.L., Kang J.H., and Yan R.X., Investigation on crystal structure and electrochemical properties of LaMgNi3.7M0.3(M=Ni,Al,Mn,Co,Sn,Cu)hydrogen storage alloys, Journal of Functional Materials, 2005, 36(9): 1372.
Qiao Y.Q., Zhao M.S., Tian B., Zhu X.J., and Cao G.Y., Study on electrochemical performances and structure of nanocrystallite Mg2Ni hydrogen storage alloy, Journal of Functional Materials, 2005, 36(12): 1875.
Murugan A.V., and Kale B.B., Comparison of different chemical routes synthesis of nanocrystalline LiMn2O4 and their influence on its physicochemical properties, Journal of Solid State Electrochemistry, 2006, 10(2): 104.
Li F.F., Zhang M.X., Hou G.Q., Shen Y., Liu Z.G., and Li H.S., Sol-gel preparation and phosphorescence property of Mn2+-doped zinc borosilicate glass thin films, Rare Metals, 2011, 30(3): 298.
Zhang J.X., Tang X.M., Liu Y.L., and Chen W.X., The soft chemistry synthesis of LaNi5 storage hydrogen alloys and their electrochemical properties, Chinese Journal of Inorganic Chemistry, 2002, 18(2): 181.
Zhang J.X., Chen W.X., Yi S.J., and Liu Y.L., Preparation of AB5 hydrogen storage alloy by reduction-diffusion method, Chinese Rare Earths, 2004, 25(1): 73.
Qi B.Y., Han X.L., and Zhu R., Influence of partial substitution of Ni by Mn on the electrochemical characteristic of LaMg2Ni9 series hydrogen storage alloys, Chinese Rare Earths, 2008, 29(5): 53.
Wang D.H., Xia J.H., Zhang S., and Xia Y., Influence of citric acid and calcination temperature on microstructure of the nano precursors of La-Mg hydrogen storage alloy, Advanced Materials Research, 2012, 339–401: 1496.
Huang M., and Gu X.Y., Influence of pH value on synthesizing anorthite ultrafine powders by sol-gel method, China Ceramic Industry, 2010, 17(3): 5.
Waqas H., and Qureshi A.H., Influence of pH on nanosized Mn-Zn ferrite synthesized by sol-gel auto combustion process, Journal of Thermal Analysis and Calorimetry, 2009, 98(2): 355.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Wang, D., Xia, J. & Zhang, S. Microstructure of nano precursors of La-Mg hydrogen storage alloy synthesized by sol-gel technology at different pH values. Rare Metals 31, 466–469 (2012). https://doi.org/10.1007/s12598-012-0540-5
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12598-012-0540-5